Zoonotic Transmission of Intestinal Protozoa: Pathways, Detection, and Control in One Health Context
This article synthesizes current evidence on the zoonotic transmission of intestinal protozoa, focusing on Entamoeba histolytica, Giardia duodenalis, and Cryptosporidium spp.
Zoonotic Transmission of Intestinal Protozoa: Pathways, Detection, and Control in One Health Context
Abstract
This article synthesizes current evidence on the zoonotic transmission of intestinal protozoa, focusing on Entamoeba histolytica, Giardia duodenalis, and Cryptosporidium spp. It explores complex transmission pathways, including waterborne, foodborne, and mechanical vectors. For researchers and drug development professionals, the content details advanced diagnostic methods like immunoassays and molecular techniques, discusses challenges in treatment and control, including drug resistance, and evaluates modeling approaches for outbreak management. The review underscores the critical need for integrated One Health strategies to mitigate the public health and economic burdens of these pervasive pathogens.
Unveiling the Pathways: A Deep Dive into Zoonotic Protozoal Transmission Mechanisms
Species Profiles and Global Burden
Intestinal protozoan parasites represent a significant threat to global health, particularly in low- and middle-income countries. Among these, Entamoeba histolytica, Giardia duodenalis (also known as G. lamblia or G. intestinalis), and Cryptosporidium spp. are of paramount importance due to their zoonotic potential and substantial disease burden. Collectively, these pathogens infect more than a billion people worldwide and are responsible for considerable morbidity and mortality, especially in areas with intense poverty, marginal communities, and rural settings with poor sanitation [1].
Giardia duodenalis is the most common non-viral, non-bacterial diarrheal illness worldwide, leading to significant health issues including weight loss, malnutrition, growth delays in children, delayed puberty, impaired cognitive development, and premature death [2]. In the United States alone, more than 1.2 million cases of giardiasis are diagnosed annually [1]. Cryptosporidium spp. are also major causes of gastroenteritis, with approximately 748,000 cases of cryptosporidiosis reported annually in the USA [1]. Entamoeba histolytica infects approximately 35-50 million people worldwide and causes roughly 55,000 deaths each year [3].
These parasites are particularly problematic in developing countries but remain a concern in developed nations despite high sanitary coverage. Outbreaks are frequently reported in the USA, Canada, Australia, and European countries, often associated with contaminated public water supplies [1]. The massive Milwaukee cryptosporidiosis outbreak in the 1990s affected nearly 400,000 people, demonstrating the potential scale of waterborne transmission [1].
Table 1: Epidemiological Profiles of Major Zoonotic Protozoa
| Parasite | Global Prevalence | Key Animal Reservoirs | Primary Transmission Routes | Major Health Impacts |
|---|---|---|---|---|
| Giardia duodenalis | >1.2 million cases annually (USA) [1] | Livestock, wildlife, domestic pets | Faecal-oral, contaminated water/food [1] | Diarrhoea, malabsorption, growth delays, cognitive impairment [2] |
| Cryptosporidium spp. | 748,000 cases annually (USA) [1] | Cattle, sheep, goats, wildlife [4] | Faecal-oral, waterborne outbreaks [1] | Severe diarrhoea, malnutrition, significant mortality in immunocompromised [4] |
| Entamoeba histolytica | 35-50 million infections worldwide [3] | Primarily human-to-human, potential zoonotic links | Faecal-oral, sexual contact [1] | Dysentery, liver abscesses, ~55,000 deaths/year [3] |
Molecular Epidemiology and Genotype-Specific Characteristics
Understanding the genetic diversity of these protozoan parasites is crucial for tracking transmission dynamics, outbreak investigation, and developing targeted control strategies. Each genus exhibits distinct genetic characteristics that influence their virulence, host range, and zoonotic potential.
Cryptosporidium Species and Subtyping
The genus Cryptosporidium comprises at least 44 species and over 120 genotypes, with C. hominis and C. parvum accounting for the majority of human cryptosporidiosis cases [5]. C. parvum is zoonotic, infecting a wide range of hosts including major domestic livestock species, whereas C. hominis is predominantly anthroponotic [5]. Subtyping using the gp60 (60-kDa glycoprotein) gene has become the standard molecular tool for discriminating within Cryptosporidium species due to its high polymorphism [5].
A recent review of C. hominis and C. parvum gp60 subtypes reported between December 2018 and January 2024 identified 264 gp60 subtypes, highlighting the extensive genetic diversity within these species [5]. The IIa and IId subtype families of C. parvum remain major contributors to infections across various hosts, with recent reports indicating the continued emergence of the IId family [5]. Studies in Algeria have identified the zoonotic C. parvum subtype IIaA17G2R1 in human, animal, and soil samples, providing concrete evidence of zoonotic transmission cycles [4].
Giardia duodenalis Assemblages
Giardia duodenalis is a multispecies complex comprising eight genetically distinct assemblages (genotypes A-H), with assemblages A and B being the most reported zoonotic genotypes [4] [1]. Assemblage A is subdivided into subtypes AI, AII, and AIII: AI is found in both humans and animals, indicating zoonotic transmission; AII is predominantly human-specific; and AIII is mainly isolated from wild animals [2]. Assemblage B includes subtypes BIII and BIV, which are commonly found in humans and display greater genetic variability than assemblage A [2].
Genotype-specific differences have clinical significance. Assemblage B is often associated with more pronounced small intestinal inflammation, including villous shortening/atrophy and lamina propria inflammation, compared to genotype A [2]. Furthermore, genotype B isolates induce more significant alterations in the intestinal mucosa and a reduction in the enzymatic activity of the brush border than genotype A [2]. Assemblage B also demonstrates higher resistance to reactive oxygen species (ROS) and nitric oxide (NO) [2].
Entamoeba Histolytica and Genetic Diversity
While Entamoeba histolytica has traditionally been considered primarily a human pathogen, recent molecular studies have revealed complex genetic diversity. The application of genetic typing methods has expanded understanding of the Entamoeba universe, revealing a multi-branched phylogenetic tree that has grown explosively from the seed planted by the realization that most people excreting cysts of "Entamoeba histolytica" were in fact infected with Entamoeba dispar [6]. Genetic manipulation of E. histolytica has proven challenging but has yielded important insights into virulence factors [7].
Table 2: Molecular Characteristics and Genotype Distribution of Zoonotic Protozoa
| Parasite | Major Genetic Groups | Zoonotic Potential | Key Genetic Markers | Geographic Trends |
|---|---|---|---|---|
| Cryptosporidium spp. | C. parvum, C. hominis, C. bovis, C. ryanae, C. andersoni [4] | High (C. parvum), Low (C. hominis) [5] | gp60, SSU rRNA, cowp, hsp70 [5] | IIa and IId families dominant; IId emerging [5] |
| Giardia duodenalis | Assemblage A (AI, AII, AIII), Assemblage B (BIII, BIV) [2] | High (A & B) [4] | bg, tpi, gdh, SSU rRNA [4] | Assemblage B more common in symptomatic cases [2] |
| Entamoeba histolytica | Primarily clonal population structure | Limited evidence | SSU rRNA, ehcp112, ehadh, ehrabb [7] | Endemic in developing countries; imported cases in industrialized nations [7] |
Zoonotic Transmission Pathways and One Health Approach
Zoonotic transmission of protozoan parasites occurs through complex interfaces involving humans, animals, and their shared environments. The One Health approach recognizes these interconnections and provides a comprehensive framework for understanding and controlling transmission.
Transmission Dynamics
Cryptosporidium spp. and Giardia duodenalis are transmitted via the faecal-oral route, following direct contact with infected hosts or environment, or through consumption of contaminated water and foods [4]. Livestock serve as important reservoirs and sources of human infections by shedding environmentally resilient Cryptosporidium oocysts and Giardia cysts [4]. Both protozoans have been associated with waterborne and foodborne outbreaks worldwide [4].
Recent studies in rural settings demonstrate these transmission patterns clearly. In northeastern Algeria, molecular epidemiological investigations identified several Cryptosporidium species, including C. bovis, C. ryanae, C. andersoni, and C. parvum, in human, animal, and environmental samples [4]. The detection of the identical zoonotic C. parvum subtype IIaA17G2R1 in human, animal, and soil samples provided concrete evidence of cross-species transmission and environmental contamination [4]. Similarly, Giardia duodenalis assemblage B was detected in human samples, while assemblage E was found in cattle and sheep [4].
In rural Iraq, studies investigating the human-animal-environment interface detected Cryptosporidium spp. in 26% of animals, 12% of humans, 5% of soil, and 15% of water samples [8]. Molecular typing identified Cryptosporidium parvum in both humans and sheep, suggesting zoonotic transmission potential [8]. The detection of identical organisms in humans, animals, and the environment underscores the significance of zoonotic and environmental transmission pathways.
Environmental Contamination and Waterborne Spread
Environmental contamination plays a crucial role in protozoan transmission. Cryptosporidium oocysts and Giardia cysts are remarkably resilient in the environment and can survive for extended periods in water, soil, and on surfaces. Waterborne transmission is particularly significant for Cryptosporidium, which has emerged as one of the major causes of outbreaks associated with treated recreational water venues in the United States [1].
Outbreaks of cryptosporidiosis have been related to public drinking water, with failures at water treatment facilities leading to large-scale outbreaks [1]. Recreational, drinking, and fountain waters have been identified as important sources of community infections worldwide [1]. The study in Iraq detected Cryptosporidium spp. in multiple water sources, including filtered drinking water, tap water, well water, and river water [8], highlighting the pervasive nature of environmental contamination.
Pathogenesis and Clinical Manifestations
The clinical spectrum of diseases produced by these protozoa ranges from asymptomatic (up to 50%) to severe disease [1]. Entamoeba histolytica can cause intestinal amoebiasis (including dysentery) and extraintestinal manifestations, most commonly liver abscesses [1]. Giardia duodenalis infections lead to malabsorption syndrome, characterized by diarrhoea, bloating, weight loss, and nutritional deficiencies [1]. Cryptosporidium spp. cause self-limiting diarrhoea in immunocompetent individuals but can lead to severe, persistent diarrhoea in immunocompromised patients [1].
Trichomonas vaginalis, while not intestinal, is worth mentioning as a mucosa-associated protozoan that accounts for one of the most prevalent non-viral sexually transmitted infections [1]. In pregnancy, T. vaginalis is frequently linked with complications such as premature birth and low birth weight babies [1].
The molecular mechanisms of pathogenesis vary among these parasites. For Entamoeba histolytica, virulence factors include the Gal/GalNAc lectin, amoebapores, and cysteine proteases [6] [7]. The recent implementation of CRISPR-Cas9 strategy in E. histolytica has enabled functional studies of virulence genes, revealing that knockout of the ehcp112 gene affected not only the target gene but also the transcription of adjacent genes in the V1 virulence locus [7].
For Giardia, pathogenesis involves disruption of the intestinal epithelial barrier, cytokine production, and microbiome alterations. Recent research has revealed genotype-specific differences in pathogenic potential, with Assemblage B inducing more significant alterations in the intestinal mucosa and greater resistance to reactive oxygen species [2].
Drug Resistance and Treatment Challenges
Current Treatment Options and Limitations
The current pharmacological treatments for amoebiasis, giardiasis, cryptosporidiosis, and trichomoniasis are limited and often associated with adverse side effects and refractory cases due to the development of resistant parasites [1]. The most effective and widely used compound is metronidazole (MTZ), a nitroimidazole derivative that has been the mainstay of protozoan parasite treatment for decades [1].
Metronidazole functions as a prodrug that requires metabolic activation within the cell to become its active form. According to the widely accepted model, nitro compounds are activated by reduction, producing toxic intermediates that cause oxidative stress [2]. The activated form inhibits pathogen DNA synthesis, leading to cell death [1]. However, treatment failure can occur due to noncompliance, resistance, and reinfection [1].
Table 3: Current Treatments and Emerging Resistance Patterns
| Parasite | First-line Treatment | Alternative Treatments | Resistance Status | Geographic Hotspots |
|---|---|---|---|---|
| Giardia duodenalis | Metronidazole/Tinidazole (5-nitroimidazoles) [9] | Quinacrine, Albendazole, Mebendazole, Nitazoxanide [9] [1] | Increasing refractory cases (2.4% overall, 12% in India) [9] | Indian subcontinent (12% refractory rate) [9] |
| Entamoeba histolytica | Metronidazole (tissue amoebiasis) + luminal agent (Paromomycin or Iodoquinol) [1] | Tinidazole, Secnidazole, Ornidazole [1] | No clinical resistance reported to date [6] | Limited evidence |
| Cryptosporidium spp. | Nitazoxanide [1] | Paromomycin, Azithromycin [1] | Limited treatment options, especially in immunocompromised | Global |
| Trichomonas vaginalis | Metronidazole, Tinidazole [1] | Secnidazole, Ornidazole [1] | Documented resistance for several decades [1] | Global |
Emerging Resistance Mechanisms
Treatment-refractory giardiasis is an emerging clinical problem, with a recent study from Sweden (2008-2020) reporting that 2.4% of 4,285 giardiasis cases were nitroimidazole-refractory [9]. The prevalence varied dramatically by geographic region, with cases acquired in India showing a substantially higher percentage (12%) compared to other parts of the world (1.0%) [9]. Even more concerning, the proportion of nitroimidazole-refractory disease among cases acquired in India rose from 8.5% to 17% between the first and second halves of the study period [9].
Recent research has revealed novel mechanisms of drug resistance in Giardia. Small extracellular vesicles (sEVs) derived from metronidazole-resistant clones can modulate drug response in wild-type parasites by altering the expression of enzymes involved in MTZ metabolism and reactive oxygen species production [2]. This vesicle-mediated modulation represents a genotype-specific mechanism for rapid phenotypic adaptation under drug-induced stress [2].
For Entamoeba histolytica, despite over 60 years of metronidazole use, clinical resistance has never been observed, unlike with most other organisms against which metronidazole is employed [6]. However, a decrease in 5-nitroimidazole susceptibility can be induced experimentally [1], suggesting the potential for resistance development remains.
Research Methodologies and Experimental Approaches
Molecular Detection and Characterization
Advanced molecular techniques have revolutionized the detection and characterization of zoonotic protozoa. The standard approach involves DNA extraction from clinical or environmental samples, followed by PCR amplification of genus-specific and species-specific genetic markers.
Sample Collection and DNA Extraction:
- Sample Types: Human and animal feces, water samples, soil samples [4] [8]
- Collection Methods: Sterile tubes for human stools, rectal collection or fresh defecation for animals, filtration or concentration for water samples [4]
- DNA Extraction: Commercial kits such as PureLink Microbiome Genomic DNA Purification Kit, typically using 200 mg of fecal or soil sample [4] [8]
Molecular Detection Protocols:
- Cryptosporidium spp.: Nested PCR targeting the SSU rRNA gene for detection, followed by gp60 gene amplification for subtyping [4] [5]
- Giardia duodenalis: qPCR targeting the SSU rRNA gene, followed by amplification of tpi, bg, and gdh genes for genotyping and subtyping [4]
- Entamoeba histolytica: qPCR targeting specific virulence genes or SSU rRNA [8]
- Blastocystis spp.: PCR amplification of SSU rRNA gene [8]
Sequencing and Analysis:
- Purification of PCR products using commercial kits
- Sanger sequencing of amplified products
- Sequence analysis using BLAST against NCBI database
- Subtype determination using specialized tools like CryptoGenotyper for Cryptosporidium gp60 subtypes [5]
In Vitro Models and Drug Sensitivity Assays
Cryptosporidium research has been hampered by the lack of in vitro models that can recapitulate the complete life cycle of the parasite [10]. Traditional systems employing cancerous cell lines have been unable to support sexual reproduction but have been widely employed for drug screening assays and transcriptome mapping [10].
Cell Culture Systems:
- Traditional Models: Human ileocecal adenocarcinoma cell line (HCT-8), human colon adenocarcinoma cell line (Caco-2) [10]
- Recent Advances: Intestinal organoids (enteroids) grown as 3D structures that more accurately reproduce cell populations in the small intestine [10]
- Key Advantage of Organoids: Ability to fulfill the complete life cycle of Cryptosporidium [10]
Drug Sensitivity Assays:
- Cell Viability Assays: MTT or similar colorimetric assays to assess cytotoxic effects [2]
- IC50 Determination: Dose-response curves to calculate half-maximal inhibitory concentration [2]
- Resistance Induction: Stepwise exposure to increasing sublethal drug concentrations over extended periods (e.g., 730 days for Giardia) [2]
Genetic Manipulation:
- CRISPR-Cas9 Systems: Recently implemented in E. histolytica for gene knockout studies [7]
- Application: Functional studies of virulence genes (e.g., ehcp112 in the V1 virulence locus) [7]
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents and Experimental Resources
| Reagent/Resource | Application | Specific Examples/Protocols | Function in Research |
|---|---|---|---|
| Cell Culture Systems | In vitro parasite propagation | HCT-8, Caco-2 cell lines; Intestinal organoids [10] | Support parasite growth and development for drug testing and pathogenesis studies |
| DNA Extraction Kits | Nucleic acid purification | PureLink Microbiome Genomic DNA Purification Kit [4] | High-quality DNA extraction from complex samples (feces, soil, water) |
| PCR Reagents | Molecular detection and genotyping | qPCR (SSU rRNA), nested PCR (gp60, bg, tpi) [4] | Sensitive and specific detection, species identification, and subtype characterization |
| Sequencing Resources | Genetic characterization | Sanger sequencing; CryptoGenotyper bioinformatic tool [5] | Subtype identification and phylogenetic analysis |
| Antibiotic Selection | Resistance studies | Metronidazole, Paromomycin, Nitazoxanide [1] [2] | Drug sensitivity assays and resistance mechanism investigation |
| CRISPR-Cas9 Systems | Genetic manipulation | ehcp112 gene knockout in E. histolytica [7] | Functional studies of virulence factors and gene essentiality |
| Extracellular Vesicle Isolation | Cell communication studies | Ultracentrifugation of sEVs from culture supernatants [2] | Investigation of novel resistance transmission mechanisms |
The study of major zoonotic protozoa requires integrated approaches that combine advanced molecular techniques with ecological understanding of transmission dynamics. The One Health framework, which recognizes the interconnectedness of human, animal, and environmental health, provides the most comprehensive strategy for addressing these complex parasitic diseases [4].
Future research directions should focus on several key areas: First, the development of improved in vitro models, particularly bioengineered systems with heterogeneous populations of intestinal epithelial and mesenchymal cells, to advance the in vitro field closer to in vivo infection models [10]. Second, enhanced surveillance using molecular tools to track emerging subtypes and resistance patterns, particularly in regions with high disease burden [9] [5]. Third, investigation of novel resistance mechanisms, including the role of extracellular vesicles in mediating phenotypic adaptation and drug tolerance [2].
The continued implementation of advanced genetic tools like CRISPR-Cas9 in E. histolytica [7] and the application of multi-omics approaches will further elucidate molecular mechanisms of pathogenesis and identify new therapeutic targets. As treatment-refractory infections continue to emerge, particularly for giardiasis [9], the development of new chemotherapeutic agents and alternative treatment strategies becomes increasingly urgent.
Understanding the complex interactions between these protozoan pathogens, their hosts, and the environment remains essential for developing effective control measures and reducing the global burden of these significant zoonotic diseases.
Fecal-oral transmission represents a predominant pathway for the spread of numerous infectious agents, including viruses, bacteria, and protozoan parasites. Within the specific context of zoonotic intestinal protozoa research, understanding these transmission routes is fundamental to developing effective public health interventions and pharmaceutical countermeasures. These pathogens traverse from animal reservoirs to human hosts through complex environmental pathways, primarily facilitated by contaminated water, food, and direct contact with contaminated fomites. The persistence of infectious stages in the environment and their resistance to common disinfection methods significantly amplifies transmission potential, particularly in regions with compromised sanitation infrastructure [11] [12]. This whitepaper delineates the core mechanisms of fecal-oral transmission, synthesizes quantitative data on pathogen persistence, and outlines standardized experimental methodologies vital for advancing research in drug and diagnostic development.
Core Transmission Routes and Mechanisms
The transmission of zoonotic intestinal protozoa and other pathogens via the fecal-oral route involves several interconnected pathways. The dynamics of each route are influenced by pathogen characteristics, environmental factors, and human behavioral practices.
Waterborne Transmission
Water serves as a critical vehicle for the dissemination of enteric pathogens. Contamination occurs when human or animal feces containing infectious stages enter water sources used for drinking, recreation, or irrigation. This is particularly prevalent in developing regions, where an estimated 2.2 million deaths annually are attributed to waterborne diseases, with diarrhea being a leading cause of childhood mortality [11]. The severity is exacerbated by limited access to improved water sources and basic sanitation for 2.4 billion people, creating a persistent cycle of contamination and infection [11]. Transmission occurs through:
- Consumption of Contaminated Drinking Water: Inadequate treatment of water derived from fecally contaminated sources is a primary route.
- Recreational Water Exposure: Accidental ingestion during swimming in contaminated pools, lakes, or rivers.
- Indirect Transmission via Irrigated Produce: Consumption of raw fruits and vegetables irrigated with contaminated water [11] [12].
Foodborne Transmission
Food acts as an efficient vehicle for pathogen transmission, with contamination possible at any stage from farm to table. Viruses are often identified as the leading cause of foodborne illnesses when an etiology is ascertained [13]. Key mechanisms include:
- Contamination by Infected Food Handlers: Poor personal hygiene, particularly inadequate handwashing after defecation, can lead to pathogen transfer to ready-to-eat foods [13] [14].
- Use of Contaminated Water in Food Processing: Water used for washing produce or as an ingredient can introduce pathogens [13].
- Zoonotic Contamination from Animal Products: Direct consumption of raw or undercooked meat from infected animals. However, a significant risk also arises from the use of raw animal manure or contaminated water in agriculture, leading to protozoal cysts or oocysts on fresh produce [15] [12]. Norovirus, Hepatitis A and E viruses, rotavirus, and various zoonotic viruses are among the common agents transmitted via food [14].
Fomite and Environmental Transmission
Inanimate objects (fomites) contaminated with fecal material can serve as intermediate points for indirect fecal-oral transmission.
- High-Touch Surfaces: Restroom surfaces (toilet seats, door handles, faucets) are frequently contaminated and can facilitate the transfer of viruses like norovirus and adenovirus to hands [16].
- Persistence on Surfaces: Enteric viruses can remain infectious on hard surfaces for extended periods. For instance, echoviruses can persist on vinyl surfaces with infectivity half-lives ranging from 1.7 to 12.6 hours [17].
- Sequential Contamination: Pathogens like norovirus can spread via fingers in a sequential manner to up to seven clean surfaces, vastly expanding the transmission zone from a single contamination point [16].
Table 1: Quantified Persistence of Enteric Viruses on Fomites (Vinyl Tile)
| Virus | Infectivity Half-Life (Hours) | Relative Frequency of Reported Cases |
|---|---|---|
| Echovirus 1 | 1.7 | <1% |
| Echovirus 2 | 4.1 | <1% |
| Echovirus 3 | 7.3 | Information Not Specified |
| Echovirus 5 | 9.7 | Information Not Specified |
| Echovirus 6 | 12.6 | 6.1% |
| Echovirus 7 | 9.4 | Information Not Specified |
| Poliovirus 1 (Sabin) | 5.8 | (Reference Strain) |
Data derived from stability studies of viruses in a 10% fecal solution at room temperature [17].
Quantitative Data on Pathogen Load and Exposure Risk
Quantitative Microbial Risk Assessment (QMRA) is a mathematical modeling approach used to quantify the public health risks from environmental exposures to pathogens. It is critical for setting standards and evaluating interventions.
Risk from Fomite Contact
QMRA models for restroom use have quantified the infection risk from touching contaminated surfaces like toilet seats and door handles. For norovirus, which has a high infectivity (10-100 viral particles can cause infection), the probability of infection from fomite contact is significant [16] [14]. One study calculated that the annual probability of infection for a one-time contact with a contaminated toilet seat was 1.76 × 10⁻⁴ [16]. A commonly used benchmark for acceptable risk in a single exposure event is 1 × 10⁻⁶ (1 in 1,000,000) [16].
Prevalence in Endemic Areas
Eco-epidemiological studies in marginalized communities provide crucial data on the baseline prevalence of zoonotic intestinal protozoa, informing the scope of the problem and potential intervention targets.
A 2025 study in coastal Ecuador found a high overall prevalence of intestinal parasites in both humans (31.87%) and their domestic dogs (78%) [12]. The most common zoonotic parasites identified were:
- In humans: Entamoeba coli (18.13%) and Entamoeba histolytica (10%)
- In dogs: Ancylostoma caninum (53.6%), Toxocara canis (12.4%) [12]
Significant associations were found between parasitic infections and risk factors such as water source, sanitation infrastructure, barefoot walking, and peridomiciliary habitat of dogs [12].
Table 2: Prevalence of Zoonotic Intestinal Protozoa in a Marginalized Community (Ecuador, 2025)
| Host | Parasite | Prevalence (%) | Zoonotic Potential |
|---|---|---|---|
| Human | Entamoeba coli | 18.13% | Yes |
| Human | Entamoeba histolytica | 10.00% | Yes |
| Human | Hymenolepis nana | 3.75% | Yes |
| Domestic Dog | Ancylostoma caninum | 53.60% | Yes (Cutaneous larva migrans) |
| Domestic Dog | Taenia spp. (Echinococcus granulosus) | 15.20% | Yes (Hydatidosis) |
| Domestic Dog | Toxocara canis | 12.40% | Yes (Visceral larva migrans) |
Data compiled from a study of 160 humans and 500 domestic dogs [12].
Experimental Models and Research Methodologies
Robust, standardized experimental protocols are essential for studying transmission dynamics, evaluating disinfectants, and developing new drugs and diagnostics.
Protocol for Assessing Viral Persistence on Fomites
This methodology is adaptable for studying the environmental stability of protozoan cysts and oocysts on various surfaces.
Objective: To determine the persistence of infectivity of enteric viruses on hard, non-porous surfaces [17].
Materials and Reagents:
- Viral Stocks: Purified viral stocks of target enteroviruses (e.g., Echovirus 6, Poliovirus Sabin) or other pathogens, propagated in appropriate cell lines (e.g., BGM or LLC-MK2 cells).
- Carriers: 1 x 1-inch coupons of the surface material to be tested (e.g., vinyl tile, stainless steel).
- Organic Load: A 10% suspension of human feces in distilled water to simulate realistic environmental conditions.
- Cell Culture Media: Eagle’s Minimum Essential Medium, supplemented with fetal bovine serum (FBS) for growth and without FBS for viral propagation.
- Elution Buffer: Trypticase soy broth or another suitable neutralizer.
- Assay System: Plaque assay using relevant cell lines for quantifying infectious viral titers.
Procedure:
- Inoculation: Mix the viral stock with the 10% fecal suspension. Apply a measured volume (e.g., 1.0 mL) of the virus-fecal mixture onto the surface coupon. The initial titer of the virus applied should be accurately determined (e.g., 4.98 to 5.7 log10 PFU) [17].
- Environmental Holding: Place the inoculated coupons in a controlled environment chamber at desired temperature (e.g., room temperature, ~27°C) and relative humidity (e.g., 40-60%). The choice of conditions should be justified based on the study's objectives [17].
- Sampling and Elution: At predetermined time intervals (e.g., 0, 24, 48 hours), elute the virus from the coupons by adding elution buffer and agitating on a shake table for 15 minutes.
- Titration: Assay the elution fluids for infectious virus using the plaque assay method. Include antibiotics in the assay to suppress bacterial growth.
- Data Analysis: Calculate the log10 reduction in viral titer compared to the initial titer (time zero). Plot log10 reduction versus time. The infectivity decay half-life (t₁/₂) is calculated using the formula: t₁/₂ (h) = 0.301 / slope of the regression line from the stability plot [17].
Protocol for Eco-Epidemiological Field Studies
Objective: To investigate the prevalence and zoonotic transmission of intestinal parasites in human and animal populations within a defined community [12].
Materials and Reagents:
- Sample Collection: Sterile, leak-proof containers for fecal samples.
- Laboratory Reagents: Solutions for coproparasitic techniques (e.g., flotation solutions like saturated sodium chloride or zinc sulfate, sedimentation agents, stains for microscopy).
- Molecular Assay Kits: DNA/RNA extraction kits, primers and probes for specific protozoa (e.g., Giardia, Cryptosporidium, Entamoeba histolytica), and PCR master mixes.
- Data Collection Tools: Standardized questionnaires and environmental assessment sheets.
Procedure:
- Study Design and Ethics: Obtain approval from relevant ethical review boards. Secure informed consent from all human participants and animal owners.
- Population and Sampling: Define the study area and population. Use a calculated sample size (e.g., using WinEpi software) to ensure statistical power. Collect fresh fecal samples from humans and their domestic animals (e.g., dogs).
- Field and Laboratory Analysis:
- Parasitological Examination: Process samples using a panel of techniques: direct smear, formalin-ethyl acetate sedimentation, flotation, and specialized methods like the modified Baermann technique for larvae [12].
- Molecular Characterization: Perform DNA extraction from fecal samples. Use PCR with genus- or species-specific primers for precise identification and genotyping. Conduct phylogenetic analysis to trace zoonotic relationships [15].
- Data Integration: Administer surveys to collect data on sociodemographic factors, hygiene practices, and environmental variables. Correlate parasitological findings with survey data using statistical analyses (e.g., chi-square tests, logistic regression) to identify significant risk factors [12].
Visualization of Transmission and Research Workflows
Zoonotic Fecal-Oral Transmission Pathways
Experimental Workflow for Persistence Studies
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents for Fecal-Oral Pathogen Studies
| Reagent/Material | Function/Application | Example Use Case |
|---|---|---|
| Cell Lines (e.g., BGM, LLC-MK2) | Propagation and titration of cultivable enteric viruses via plaque or TCID₅₀ assays. | Quantifying infectious viral load in persistence and disinfection studies [17]. |
| Plaque Assay Reagents (Agar overlay, cell culture media, neutral red stain) | Enumeration of infectious viral particles forming plaques on a cell monolayer. | Determining the decay kinetics of echoviruses on fomites [17]. |
| PCR/Kits & Reagents | DNA/RNA extraction, amplification, and detection of pathogen-specific genetic material. | Molecular characterization of Giardia and Cryptosporidium in human and dog feces [15] [18]. |
| Coproparasitic Reagents (Flotation solutions, stains, preservatives) | Concentration and microscopic visualization of helminth eggs and protozoan cysts in feces. | Determining prevalence and conducting morphological identification in field studies [12]. |
| Surface Coupons (Vinyl tile, Stainless steel) | Standardized test surfaces for studying pathogen persistence and transfer efficiency. | Evaluating the survival of enteroviruses on hard, non-porous fomites [17]. |
| Organic Load Simulant (e.g., 10% Fecal Suspension) | Mimics the protective effect of bodily fluids during environmental persistence and disinfection tests. | Providing realistic conditions for pathogen stability on surfaces [17]. |
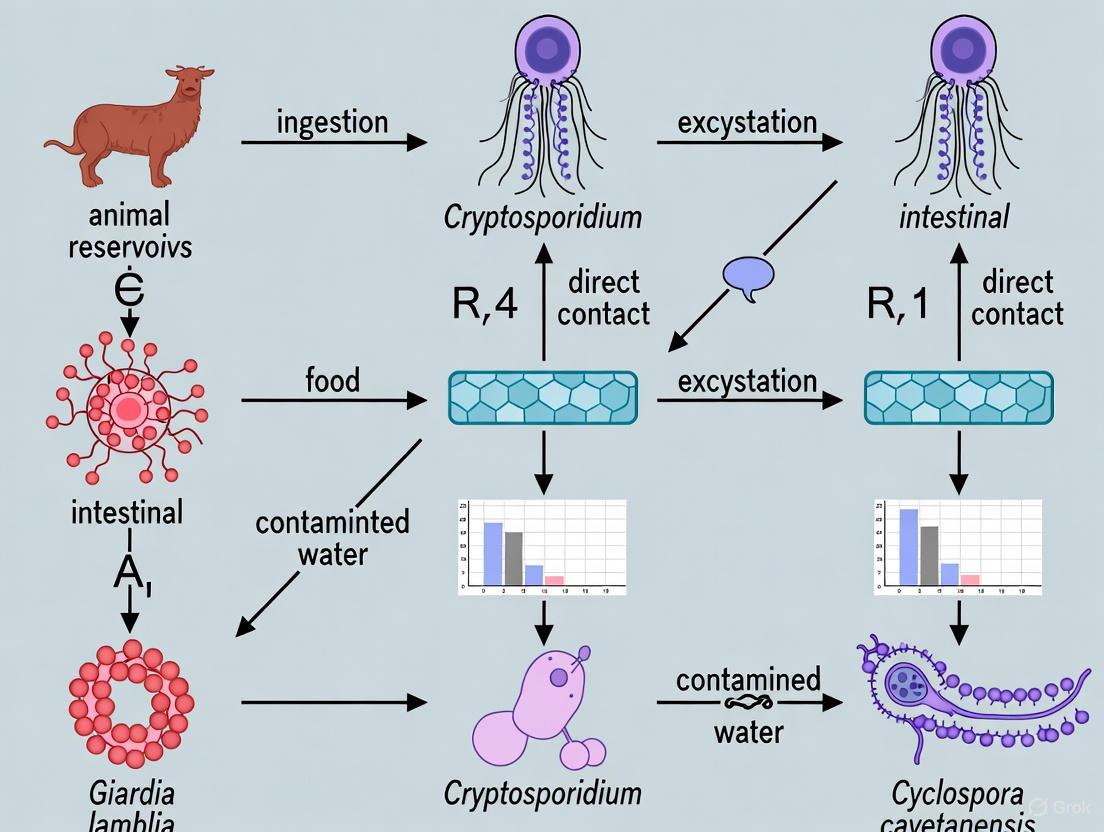
Intestinal protozoan parasites, including Cryptosporidium spp., Giardia duodenalis, Entamoeba histolytica, and Blastocystis sp., represent a significant global health burden, particularly in regions with limited access to sanitation and healthcare resources [18] [19]. The transmission of these parasites occurs primarily via the fecal-oral route, with zoonotic transmission playing a crucial role in their epidemiology [20] [21]. Synanthropic insects—those living in close association with humans—particularly filth flies and cockroaches, have evolved to thrive in human and animal habitats, making them efficient mechanical vectors for these pathogens [22] [23]. Their filthy breeding habits, feeding mechanisms, and indiscriminate movement between filth and food sources enable them to mechanically transmit protozoan cysts and oocysts [22] [24]. Understanding the specific mechanisms and efficiency of this mechanical transmission is fundamental to developing effective control strategies within the broader context of zoonotic disease pathways.
Transmission Mechanisms and Vector Efficiency
Mechanical Transmission Pathways
The mechanical transmission of human protozoan parasites by synanthropic insects occurs via several distinct pathways, which are governed by the insect's morphology and behavior.
- External Adherence: Flies can carry human pathogens on their sponging mouthparts, body and leg hairs (setae), and the sticky pads (pulvilli) of their feet [22]. The fine hairs on a fly's tarsi are coated with a sticky substance that enhances adhesion to surfaces and simultaneously traps particles such as protozoan cysts [22]. Similarly, cockroaches can externally transport parasitic cysts on their cuticle and legs [25] [24]. The electrostatic charge of the insect's exoskeleton further enhances the adhesion of neutrally or differently charged particles [22].
- Regurgitation and Fecal Deposition: During feeding, flies frequently regurgitate digestive enzymes onto food sources (in the form of "vomit drops") before ingesting the liquefied material, a process that can transfer pathogens from their alimentary tract [22]. Furthermore, protozoan parasites can pass through the fly gastrointestinal tract without alteration of their infectivity and are subsequently deposited on visited surfaces in "fecal spots" [22]. Frequent meals on contaminated substrates coupled with alternating regurgitation and ingestion lead to progressive accumulation of human pathogens within the fly's digestive system [22].
- Feces-Enhanced Transmission: The effectiveness of feces in enhancing the transmission of infectious agents by house flies is considerably greater than that of any other substrate, primarily due to fecal viscosity which increases the efficiency of tarsi and bristles in trapping suspended particles [22].
Table 1: Primary Mechanical Transmission Mechanisms of Protozoan Parasites by Insects
| Mechanism | Insect Type | Description | Parasites Commonly Transmitted |
|---|---|---|---|
| External Adherence | Flies, Cockroaches | Pathogens carried on exoskeleton, leg hairs, sticky tarsal pads | Ascaris spp., Trichuris spp., Entamoeba cysts [22] [25] |
| Regurgitation (Vomit Drops) | Primarily Flies | Deposit of alimentary tract contents during feeding | Cryptosporidium spp., Giardia spp., Entamoeba spp. [22] |
| Fecal Spots | Flies, Cockroaches | Defecation of infectious agents onto surfaces | Cryptosporidium oocysts, Giardia cysts, Entamoeba cysts [22] [24] |
| Intermediate Grooming | Cockroaches | Transfer of pathogens from body to antennae and mouthparts during grooming | Various protozoan cysts and helminth eggs [24] |
Vector Competence of Filth Flies and Cockroaches
The efficiency of different insect species as mechanical vectors varies based on their synanthropy, endophily (preference for entering buildings), communicative behavior, and attraction to filth and human food [22].
Filth Flies: Over 50 species of synanthropic flies have been associated with unsanitary conditions and dissemination of human pathogens [22]. Of these, 21 species are listed by regulatory agencies as causative agents of gastrointestinal diseases, including Musca domestica (house fly), Fannia canicularis (lesser house fly), and various Calliphoridae (blow flies) [22]. A recent 2025 study in Upper Egypt collected 12,749 flies and identified Musca domestica as the dominant species, with a remarkably high parasite infestation rate of 96.6% [23]. The study revealed that Cryptosporidium was the most prevalent parasite (64.4–100%), infecting all collected fly species, followed by Entamoeba (22.6–90.1%) and Balantidium (8.9–100%) [23]. Parasitic infections in flies were highest in autumn and spring, with the lowest rates in winter [23].
Cockroaches: Cockroaches frequently feed on human feces, enabling them to disseminate cysts of enteric protozoans [22] [25]. A 2024 study found that 29.73% of collected cockroaches carried medically important parasites, with four types of helminths identified: Ascaris lumbricoides (47.27%), Enterobius vermicularis (30.91%), Trichuris spp. (7.27%), and Hymenolepis nana (14.55%) [25]. The parasites were more frequently found on the external surface of cockroaches (69.09%) compared to the internal surface (30.91%) [25]. Cockroaches captured in toilets carried a higher percentage of parasites (16.75%) compared to those from kitchens (5.94%) and other house areas (7.02%) [25]. An earlier study in Ethiopia examining 6,480 cockroaches found they carried Entamoeba coli, Entamoeba histolytica/dispar cysts, as well as helminth eggs including Enterobius vermicularis, Trichuris trichiura, Taenia spp., and Ascaris lumbricoides [24].
Table 2: Prevalence of Select Protozoan Parasites in Synanthropic Insects Based on Field Studies
| Insect Species | Parasite | Prevalence (%) | Location | Sample Size | Citation |
|---|---|---|---|---|---|
| Musca domestica (House Fly) | Cryptosporidium spp. | 64.4 - 100% | Upper Egypt | 12,749 flies (total study) | [23] |
| Musca domestica (House Fly) | Entamoeba spp. | 22.6 - 90.1% | Upper Egypt | 12,749 flies (total study) | [23] |
| Blattella germanica (German Cockroach) | Entamoeba histolytica/dispar | >25% | South Taiwan | 185 cockroaches | [22] |
| Periplaneta americana (American Cockroach) | Entamoeba histolytica/dispar | >10% | South Taiwan | 185 cockroaches | [22] |
| Cockroaches (Various) | Helminths (Ascaris, Enterobius, etc.) | 29.73% | Duhok, Iraq | 185 cockroaches | [25] |
Experimental Methodologies for Vector Incrimination
Field Collection and Morphological Identification
Establishing insects as mechanical vectors requires robust experimental protocols that demonstrate both the carriage and transmission of viable, infectious parasites.
Insect Collection and Identification: Flies are typically collected using sweep nets from targeted sites such as animal rearing facilities, garbage areas, and human dwellings [23]. Cockroaches are often trapped using sticky traps or manual collection from kitchens, toilets, and food storage areas [25] [24]. Collected specimens are taxonomically identified using standard morphological keys [23] [24]. For example, a 2025 study immobilized collected flies by placing them in a freezer at -20°C before identification [23].
Parasite Isolation and Microscopic Identification: For external parasites, insects are washed in a saturated salt solution, saline, or phosphate-buffered saline (PBS) and vortexed to dislodge eggs and cysts attached to the insect's body [23] [25]. The wash solution is then centrifuged, and the sediment is examined microscopically [23]. For internal parasites, insects are dissected, and their gut contents are examined [24]. Microscopic identification of parasites relies on concentration techniques (e.g., flotation, sedimentation) and staining methods (e.g., Ziehl-Neelsen for Cryptosporidium, trichrome for amoebae) [21] [23]. The Fuelleborn, Heine, and ZnSO4 flotation microscopic techniques are commonly used for parasite identification in field studies [21].
The following workflow diagram illustrates a standard experimental approach for detecting parasites in insect vectors:
Experimental Workflow for Parasite Detection in Insects
Molecular Confirmation and Characterization
While microscopy provides initial evidence, molecular techniques are essential for confirming parasite identity, determining species, and assessing zoonotic potential.
DNA Extraction and Amplification: Parasite DNA is extracted from insect washes, gut contents, or from isolated cysts/oocysts [23]. Nested PCR protocols are frequently employed for their high sensitivity, particularly for detecting low numbers of parasites [23]. For Cryptosporidium, commonly targeted genes include the COWP (oocyst wall protein) and the small subunit ribosomal RNA (SSU rRNA) genes [23]. Similar genetic markers exist for Giardia (e.g., beta-giardin, TPI) and Entamoeba [23].
Genotyping and Sequencing: Amplified PCR products are sequenced, and the resulting sequences are compared to reference strains in genomic databases to determine the species and genotypes present [23]. This is crucial for understanding zoonotic transmission. For instance, a 2025 study in Upper Egypt using PCR and sequencing confirmed the presence of the zoonotic species Cryptosporidium parvum in filth flies, directly implicating them in the transmission of a human-pathogenic strain [23]. Next-Generation Sequencing (NGS) can be used for more comprehensive characterization, such as identifying Blastocystis subtypes in environmental samples [26].
Table 3: Key Molecular Targets for Protozoan Parasite Identification in Vector Studies
| Parasite | Genetic Target | Method | Application in Vector Studies |
|---|---|---|---|
| Cryptosporidium spp. | COWP gene, SSU rRNA | Nested PCR, Sequencing | Species identification; zoonotic strain confirmation (e.g., C. parvum) [23] |
| Giardia duodenalis | Beta-giardin, TPI, GDH | PCR, Multiplex PCR | Assemblage determination (host-specific vs. zoonotic) [23] |
| Entamoeba histolytica | 18S rRNA, specific surface antigen genes | PCR, Real-time PCR | Differentiation from non-pathogenic E. dispar [18] |
| Blastocystis sp. | SSU rRNA | Conventional PCR, NGS | Subtyping (ST1-ST17 in humans/animals) for transmission tracking [26] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful investigation into the role of insects as mechanical vectors requires specific reagents and materials for field collection, laboratory processing, and molecular analysis.
Table 4: Essential Research Reagents and Materials for Mechanical Vector Studies
| Category / Item | Specific Examples | Function / Application | Key Considerations |
|---|---|---|---|
| Collection & Storage | Sweep nets, Sticky traps, Sterile plastic jars, Freezer (-20°C) | Field capture and immobilization of insects; transport to lab | Maintain cold chain; prevent cross-contamination [23] |
| Washing & Dissection | Phosphate-Buffered Saline (PBS), Saturated salt solution, 70% Alcohol, Dissecting microscope, Fine forceps | Removal of external parasites; sterile dissection for internal parasites | Vortexing essential for dislodging external stages [23] [25] |
| Microscopy & Staining | Light microscope, ZnSO4 / Sucrose flotation solution, Ziehl-Neelsen stain, Trichrome stain, IFA kits | Parasite concentration, morphological identification, and differentiation | Staining enhances detection of specific parasites (e.g., ZN for Cryptosporidium) [21] [23] |
| Molecular Biology | DNA extraction kits, PCR master mixes, Specific primers (e.g., COWP, SSU rRNA), Gel electrophoresis equipment, Sequencer | Genetic detection, species confirmation, and genotyping | Nested PCR increases sensitivity for low-abundance parasites [23] |
Integration with Zoonotic Transmission Pathways and One Health Perspective
The mechanical transmission of intestinal protozoa by filth flies and cockroaches represents a critical link in zoonotic transmission pathways, intimately connected to environmental conditions, animal reservoir hosts, and human behavior.
Epidemiological Connections: Outbreaks and cases of food-borne diarrheal diseases in urban and rural areas are closely related to seasonal increases in the abundance of filth flies [22]. Enforced fly control has been correlated with reductions in the occurrence of such diseases [22]. A 2025 study in Kazakhstan on calf protozoal infections, while not directly studying insects, highlighted that understanding factors influencing infection risks in livestock is critical for controlling zoonotic parasites like Cryptosporidium and Giardia [21]. These parasites in livestock settings can be disseminated by flies to human environments.
Environmental and Sanitary Drivers: Poor Water, Sanitation, and Hygiene (WASH) services are consistently identified as major risk factors for parasitic transmission involving insect vectors [20]. A 2025 study in Chile on waterborne transmission of Blastocystis sp. found that higher water temperature and greater rainfall were significantly associated with parasite presence, while potable water was associated with significantly lower odds of infection [26]. These environmental factors also influence insect vector populations and their contact with contaminated materials.
The following diagram illustrates the interconnected transmission cycle of intestinal protozoa, highlighting the role of mechanical vectors:
Zoonotic Transmission Cycle with Mechanical Vectors
Filth flies and cockroaches play a significant and mechanistically well-defined role as mechanical vectors in the dissemination of cysts and oocysts of zoonotic intestinal protozoa. Their synanthropic behavior, breeding habits, and specific morphological adaptations make them highly efficient in picking up pathogens from contaminated sources such as animal and human feces and depositing them on human food and surfaces. Field and laboratory evidence consistently demonstrates high carriage rates of parasites like Cryptosporidium, Giardia, and Entamoeba in these insects. Robust methodological approaches, combining traditional morphological techniques with modern molecular tools, are essential for accurately incriminating these vectors and understanding the specific zoonotic strains they carry. From a One Health perspective, controlling these mechanical vectors through improved sanitation, waste management, and targeted vector control represents a critical intervention point for breaking the transmission cycle of these pervasive parasitic diseases. Future research should continue to integrate vector studies with environmental and genetic epidemiology to develop more effective, integrated control strategies.
Intestinal protozoan parasites represent a significant global public health burden, particularly in developing regions. The resilience of their environmental stages—oocysts and cysts—is a critical factor influencing their transmission and the incidence of infection [27]. These robust forms enable parasites to survive outside a host for extended periods, facilitating their spread through water, soil, and food. This persistence is fundamental to the epidemiology of numerous zoonotic diseases, as it bridges the ecological gap between animal reservoirs and human populations [28] [29]. Understanding the factors that govern the survival of these infectious stages in various environments is therefore paramount for developing effective intervention strategies. This whitepaper synthesizes current research on the environmental persistence of key zoonotic protozoa, providing a technical guide for researchers and public health professionals focused on mitigating their impact.
Key Protozoan Parasites and Their Environmental Stages
Major Zoonotic Pathogens
Several protozoan parasites utilize robust environmental stages to ensure their transmission through indirect routes. Among the most significant are Cryptosporidium spp., Giardia duodenalis, and Toxoplasma gondii.
- Cryptosporidium spp.: The oocysts of Cryptosporidium are immediately infectious upon excretion and are remarkably resistant to common disinfectants, including chlorine, making waterborne outbreaks a frequent occurrence [30] [29].
- Giardia duodenalis: This parasite produces cysts that are shed in feces and can survive in cool, moist environments for months. Giardiasis is a common cause of diarrheal disease worldwide, with transmission often linked to contaminated water [29] [27].
- Toxoplasma gondii: The oocysts of T. gondii, shed only by felids, are a key to the parasite's global distribution. Their robust wall allows for long-term persistence and dissemination through watersheds, leading to infections in a vast range of intermediate hosts, including humans and marine animals [28].
The following table summarizes the survival characteristics of these and other protozoan parasites in the environment.
Table 1: Environmental Persistence of Key Zoonotic Protozoan Parasites
| Parasite | Infectious Stage | Key Environmental Reservoirs | Survival Duration | Key Resistance Factors |
|---|---|---|---|---|
| Toxoplasma gondii | Oocyst | Water, soil, shellfish, fresh produce | Up to 18 months in soil; years in fresh and marine water [28] [31] | Robust oocyst wall; resistant to many environmental stresses [28] |
| Cryptosporidium spp. | Oocyst | Water, soil, manure, fresh produce | Months in soil and water; at least a year in seawater [31] [32] | Extreme chlorine resistance; robust oocyst wall [30] |
| Giardia duodenalis | Cyst | Water, soil, surfaces | Months in cold water [29] | Cyst wall provides physical and chemical protection |
| Cyclospora cayetanensis | Oocyst | Water, soil, fresh produce | Persists for weeks to months on herbs and in soil; requires sporulation to become infectious [31] | Oocyst wall confers resistance to disinfection and environmental stress [31] |
| Entamoeba histolytica | Cyst | Water, soil, contaminated food | Days to weeks, depending on conditions [27] | Cyst wall provides protection against desiccation |
The Role of the Oocyst Wall in Persistence
The remarkable environmental persistence of parasites like T. gondii and Cryptosporidium is largely attributed to the structural and biochemical properties of the oocyst wall. This wall is a highly durable, double-layered structure that is impermeable to many substances and protects the sporozoites within from environmental extremes, including temperature fluctuations, UV radiation, and chemical disinfectants [28] [31]. This robustness enables oocyst dissemination through watersheds and long-term persistence in diverse ecosystems, constituting a primary source of infection for humans and animals through contaminated water, soil, or food [28].
Factors Influencing Environmental Survival and Inactivation
The persistence of protozoan (oo)cysts is not absolute; it is heavily influenced by a complex interplay of abiotic and biotic factors. Understanding these variables is crucial for risk assessment and implementing effective control measures.
Abiotic Factors
Abiotic factors such as temperature, humidity, and soil characteristics are primary determinants of (oo)cyst survival.
- Temperature: Elevated temperatures significantly accelerate the inactivation of (oo)cysts. Studies show that die-off rates for Cryptosporidium and Giardia increase with rising incubation temperatures [32]. For example, C. parvum oocysts in manure inactivate significantly faster under summer conditions (21–42°C) compared to winter conditions (1–18°C) [33].
- Desiccation: Low humidity and dry conditions are generally deleterious. C. cayetanensis oocysts show short-lived persistence in arid soil conditions, while Cryptosporidium oocysts are also susceptible to desiccation, with higher inactivation rates in arid environments [31] [32].
- Soil Properties: Soil texture and chemistry modulate survival. T. gondii oocysts can persist in soil for up to 18 months [31]. Furthermore, the composition of the protozoan community itself shifts dramatically in extreme soil environments, such as those contaminated by acid mine drainage (AMD), where groups like Apicomplexa and Euglenozoa can dominate highly acidic soils [34].
Biotic Factors
The presence of organic matter, such as feces or serum, can have a protective effect on (oo)cysts, enhancing their survivability by providing a buffer against environmental stresses and potentially shielding them from desiccation [32]. The specific surface material also impacts survival, with porous materials like fabric often leading to faster die-off compared to non-porous surfaces like stainless steel [32].
Table 2: Impact of Environmental Conditions on Protozoan (Oo)Cyst Survival
| Environmental Factor | Effect on Survival & Persistence | Experimental Evidence |
|---|---|---|
| Temperature | Inversely correlated with survival; higher temperatures increase die-off rates. | First-order decay kinetics show significantly higher C. parvum inactivation in manure in summer (k= -0.01379 day⁻¹) vs. winter (k= -0.00405 day⁻¹) [33]. |
| Humidity / Moisture | Critical for survival; arid conditions accelerate inactivation. | C. cayetanensis oocysts persist longer in soil under "wet" vs. "arid" watering conditions [31]. Cryptosporidium has greater inactivation in arid environments [32]. |
| Surface Porosity | Survival is generally lower on porous materials. | Fastest die-off for Giardia and Cryptosporidium observed on fabric, followed by ceramic, formica, skin, and stainless steel [32]. |
| Organic Matter | Protective effect; enhances survivability. | Presence of bovine serum albumin reduced the die-off rate of Giardia cysts on various surfaces [32]. |
| pH | Shapes protozoan community structure; certain groups thrive in extremes. | Apicomplexa and Euglenozoa dominated protozoan communities in extremely acidic (pH < 3) soils impacted by acid mine drainage [34]. |
Methodologies for Studying Persistence and Detection
Robust experimental protocols are essential for generating reliable data on the persistence and detection of protozoan parasites in environmental samples.
Experimental Workflow for Persistence Studies
A generalized workflow for studying the persistence of protozoa like Cyclospora cayetanensis in soil and produce involves controlled contamination and molecular detection over time [31].
Molecular Detection Protocols
The detection of low numbers of (oo)cysts in complex environmental matrices like soil, water, or fresh produce requires highly sensitive and specific molecular methods. Microscopy is often insufficient due to low pathogen load and heterogeneous distribution [31]. For C. cayetanensis, the U.S. FDA developed a modified real-time PCR method (Mit1C qPCR) targeting a specific mitochondrial gene, which can detect as few as five oocysts in a 25-50g sample of fresh produce [31]. Similarly, for soil, a method combining flotation in high-density solutions with this qPCR assay can detect as few as 10 oocysts in a 10g sample [31]. Broader profiling of protozoan communities in environmental water samples can be achieved using PCR targeting the 18S rRNA gene followed by sequencing, which allows for the identification of diverse phyla, including amoebae, Apicomplexa, ciliates, and flagellates [30].
Assessing Viability and Infectivity
Beyond mere presence, determining viability is critical for risk assessment. Methods include:
- Staining Techniques: Using vital dyes (e.g., propidium iodide/PI and fluorescein isothiocyanate/FITC) to assess oocyst wall integrity and viability [33].
- In vitro Excystation: Monitoring the release of sporozoites under conditions that mimic the gut environment [35].
- Animal Infectivity Models: Using susceptible animal models to confirm the infectivity of recovered (oo)cysts, which is considered a gold standard but is resource-intensive [35].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents and Materials for Environmental Persistence Research
| Reagent / Material | Function in Research | Example Application / Note |
|---|---|---|
| Purified (Oo)cysts | Serve as the standard inoculum for controlled persistence and inactivation studies. | C. parvum (Iowa isolate) oocysts, G. muris cysts, or C. cayetanensis oocysts from validated repositories [32] [31]. |
| Real-time PCR Reagents | Enable sensitive and specific detection and quantification of parasite DNA in environmental samples. | Used with specific primers/probes (e.g., Mit1C qPCR for C. cayetanensis) [31]. |
| 18S rRNA Primers | Allow for broad-range detection and identification of diverse protozoan species via sequencing. | Primers like P-SSU-342f and Medlin B can generate a ~1,360 bp amplicon for sequencing and species identification [30]. |
| Viability Stains | Differentiate between live and dead (oo)cysts based on membrane integrity and enzymatic activity. | Dyes like Propidium Iodide (PI) and Fluorescein Isothiocyanate (FITC) are used in combination [33]. |
| High-Density Solution | Used in flotation protocols to separate buoyant (oo)cysts from heavier debris in soil samples. | Critical for purification and concentration prior to DNA extraction or microscopy [31]. |
| Environmental Chambers | Provide precise control over temperature and relative humidity for studying inactivation kinetics. | Allow simulation of seasonal diurnal cycles (e.g., using LSTM-modeled data) [33] [32]. |
| Protective Organic Matter | Used to simulate "soiled" conditions and test its protective effect on (oo)cyst survival. | Bovine serum albumin (BSA) is commonly used in survival studies on surfaces [32]. |
The environmental persistence of protozoan oocysts and cysts is a cornerstone of their transmission, posing a substantial challenge to public health systems worldwide. The robust nature of these stages, capable of surviving for months to years in soil and water, ensures their widespread dissemination and availability for human and animal infection. Factors such as temperature, humidity, and the presence of organic matter are critical modulators of their survival. Tackling the threat posed by these persistent pathogens requires a transdisciplinary approach that integrates advanced molecular detection, precise environmental modeling, and a deeper understanding of the ecology of zoonotic transmission. Future research must continue to elucidate the specific environmental triggers that lead to inactivation and leverage this knowledge to develop targeted interventions, ultimately reducing the global burden of diseases caused by these resilient parasites.
Gastrointestinal protozoa represent a significant threat to global public health, with cattle and other domestic animals serving as critical reservoirs for zoonotic transmission. Pathogens such as Cryptosporidium spp., Giardia duodenalis, and Blastocystis sp. infect a wide range of hosts, including humans and livestock, primarily through the fecal-oral route via contaminated water, food, or direct contact [36]. The interconnectedness of human, animal, and environmental health underscores the importance of the One Health framework in understanding and mitigating these threats [37]. This whitepaper examines the role of cattle and small ruminants as reservoirs for intestinal protozoa, analyzes transmission pathways, and details advanced methodological approaches for pathogen detection and characterization, providing researchers and drug development professionals with comprehensive technical guidance for addressing these zoonotic challenges.
Major Zoonotic Protozoa in Cattle and Domestic Animals
Cryptosporidium spp.
Cryptosporidium is one of the most prevalent zoonotic parasitic protozoa, infecting more than 260 animal species and representing the second most common cause of childhood diarrhea globally after rotavirus [36]. It is particularly concerning as an opportunistic infection in immunocompromised individuals. At least 47 Cryptosporidium species have been identified, with 14 species demonstrating the capacity to infect sheep. Among these, C. ubiquitum, C. xiaoi, C. andersoni, and C. parvum are most common, with C. parvum and C. ubiquitum posing particular concerns for public health when detected in sheep and goats [36].
Giardia duodenalis
G. duodenalis is another significant enteric parasite with a broad host range, representing a species complex divided into eight assemblages (A–H) [36]. Assemblages A and B are most relevant for zoonotic transmission, while assemblage E predominates in livestock such as sheep and goats. Although assemblages C-H were previously considered host-adapted, human infections with assemblages C, D, E, and F have been reported, suggesting broader zoonotic potential than previously assumed [36]. Studies of sheep and goats have identified a predominance of G. duodenalis assemblage E, with assemblage A occurring less frequently and assemblage B rarely detected.
Blastocystis sp.
Blastocystis sp. is one of the most common enteric protists, carried by more than one billion people worldwide [36]. More than 30 subtypes (STs) have been identified based on polymorphism of the small subunit ribosomal RNA (SSU rRNA) gene. In livestock, ST10 is most frequently detected, while in humans, ST1–ST4 predominate, accounting for over 95% of Blastocystis sp. infections in humans. ST6–ST9 have also been detected in humans and birds, suggesting potential zoonotic links, though the pathogenic role of Blastocystis sp. remains debated, and its zoonotic transmission dynamics are not yet fully understood [36].
Enterocytozoon bieneusi
E. bieneusi is a fungus-like protozoan with a global distribution among animals and humans [36]. More than 500 genotypes have been described, clustering into 11 phylogenetic groups. Groups 1 and 2 harbor most zoonotic genotypes, while Groups 3–11 are largely host adaptation or environment-specific. In ruminants, genotypes from Groups 1 and 2 predominate, with genotype BEB6 frequently reported in sheep and goats in China. Recent molecular epidemiological surveys indicate that genotypes once considered ruminant-specific are increasingly found in humans, underscoring the importance of continued surveillance [36].
Table 1: Major Zoonotic Protozoa in Domestic Animals
| Pathogen | Major Animal Hosts | Predominant Genotypes in Animals | Zoonotic Potential | Clinical Significance in Humans |
|---|---|---|---|---|
| Cryptosporidium spp. | Cattle, sheep, goats | C. xiaoi, C. ubiquitum, C. bovis, C. andersoni | High | Childhood diarrhea, opportunistic infections in immunocompromised |
| Giardia duodenalis | Cattle, sheep, goats | Assemblage E | Moderate to High | Diarrhea, malabsorption syndromes |
| Blastocystis sp. | Sheep, goats | ST10, ST14, ST26, ST5 | Debated | Pathogenicity uncertain, potential association with IBS |
| Enterocytozoon bieneusi | Sheep, goats | BEB6, COS-I, CHS8, CHS7 | High | Diarrhea in immunocompromised hosts |
Prevalence and Distribution Data
Epidemiological studies across diverse geographical regions demonstrate substantial prevalence of intestinal protozoa in cattle and small ruminants, with significant implications for zoonotic transmission.
Studies in Kazakhstan
A recent large-scale study conducted in 2018-2019 and 2021-2024 in Kazakhstan examined 1,586 fecal samples from calves on 12 industrialized dairy farms across 11 districts. The research revealed distinct age-related patterns of infection [38] [21]:
- Cryptosporidium spp. infections were highly concentrated in the youngest calves, with prevalence of 49.2% detected in the 1-30-day group. The risk of infection dropped dramatically with older age (p < 0.001).
- Eimeria spp. prevalence in the 1-month group was 2.0% and significantly increased with age. Calves aged 31-90 days had 27.3 times higher odds of infection (95% CI: 17.07–45.35, p < 0.001), with elevated odds persisting in older groups (p < 0.001).
- Giardia spp. infected 5.2% of the youngest calves, with the species more evenly distributed across age groups and no statistically significant variation.
- No significant seasonal variation in infection rates was found, suggesting that age-targeted parasite control strategies may be more effective than seasonal approaches for managing parasitic infections in calves under intensive dairy farming conditions [38] [21].
Studies in China
A comprehensive survey in Heilongjiang Province, Northeast China, analyzed 1,011 fecal samples from domestic small ruminants (845 sheep and 166 goats) across 13 regions between May 2023 and July 2024 [36]. The findings revealed:
- Overall infection rates for Cryptosporidium spp. at 4.15% (42/1011), G. duodenalis at 2.67% (27/1011), E. bieneusi at 12.15% (127/1011), and Blastocystis sp. at 3.56% (36/1011).
- Mixed infections with two or more protozoa occurred in 2.97% (30/1011) of samples.
- Geographic location was identified as a significant risk factor for the prevalence of Cryptosporidium spp., E. bieneusi, and Blastocystis sp. in domestic small ruminants.
- Molecular characterization identified four Cryptosporidium genotypes (C. xiaoi, C. ubiquitum, C. bovis, C. andersoni), seven E. bieneusi genotypes (BEB6, COS-I, CHS8, CHS7, CHG1, CHG3, J), two G. duodenalis assemblages (assemblage E, assemblage A), and six Blastocystis subtypes (ST10, ST14, ST26, ST5, ST15, ST30).
Table 2: Prevalence of Zoonotic Protozoa in Domestic Ruminants Across Regions
| Region | Host Species | Sample Size | Cryptosporidium spp. | Giardia duodenalis | Enterocytozoon bieneusi | Blastocystis sp. | Reference |
|---|---|---|---|---|---|---|---|
| Kazakhstan | Calves | 1,586 | Age-dependent (1-30 days: 49.2%) | Age-dependent (1-30 days: 5.2%) | Not reported | Not reported | [38] |
| Heilongjiang, China | Sheep & Goats | 1,011 | 4.15% | 2.67% | 12.15% | 3.56% | [36] |
| Peruvian Amazon | Humans (HIV+) | 315 | 25.7% | 2.9% | Not reported | Not specified | [39] |
Experimental Protocols and Methodologies
Sample Collection and Storage
Proper sample collection and storage are critical for accurate pathogen detection. In the Kazakhstan study, fecal samples were individually collected from calves of varying ages and breeds in 12 industrialized farms [38]. Similarly, in the Heilongjiang study, 1,011 fecal samples from domestic small ruminants were collected across all 13 administrative regions [36]. The protocols included:
- Stratified Random Sampling: Population of calves categorized into age groups (1–30, 31–90, 91–120, and >120 days) to evaluate prevalence and age-associated risk [38].
- Sample Preservation: Immediate placement of fecal samples into sterile tubes labeled with information, storage in car refrigerators at low temperature during transport, followed by laboratory storage at −20°C until processing [36].
- Quality Control: Maintenance of cold chain during transport, with samples kept in coolers to preserve integrity during transit [39].
Microscopic Techniques
Traditional microscopic techniques remain fundamental for initial detection and identification of intestinal protozoa:
- Flotation Microscopic Techniques: Cryptosporidium spp., Giardia spp., and Eimeria spp. were identified using Fuelleborn, Heine and ZnSO4 flotation microscopic techniques [38].
- Staining Methods: Modified Ziehl–Neelsen (MZN) staining for Cryptosporidium spp. oocysts; Lugol's iodine solution for identification of Giardia spp., Entamoeba spp., Blastocystis spp., and other intestinal protozoa [39].
- Quality Assurance: All positive samples assessed by two technicians, with 20% of negative stools re-examined as quality control. Discordant results re-evaluated until consensus reached [39].
Molecular Detection and Characterization
Advanced molecular techniques enable precise species and genotype identification, crucial for understanding transmission dynamics:
- DNA Extraction: Genomic DNA extracted from fecal samples using commercial kits (e.g., Solarbio Stool Genomic DNA Extraction Kit), with quality and quantity determined by electrophoresis in 1.5% agarose gel and spectrophotometer measurement at A260/280 [36].
- PCR Amplification:
- Sequencing and Genotype Analysis: Positive PCR products sequenced in both directions by commercial facilities (e.g., Sangon Biotech Ltd., Shanghai), with sequences analyzed to determine species/genotypes [36].
Figure 1: Experimental Workflow for Detection and Characterization of Zoonotic Protozoa
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for Zoonotic Protozoa Studies
| Reagent/Kit | Application | Specific Function | Example from Studies |
|---|---|---|---|
| Solarbio Stool Genomic DNA Extraction Kit | DNA extraction | Isolation of high-quality genomic DNA from fecal samples | Used in Heilongjiang study for DNA extraction from sheep and goat feces [36] |
| Fuelleborn, Heine, ZnSO4 flotation solutions | Microscopy | Concentration and identification of parasite oocysts/cysts | Used in Kazakhstan calf study for initial parasite identification [38] |
| Modified Ziehl–Neelsen (MZN) stain | Microscopy | Staining of Cryptosporidium oocysts for microscopic visualization | Used in Peruvian HIV+ patient study for Cryptosporidium detection [39] |
| Lugol's iodine solution | Microscopy | Enhancement of protozoan visualization in wet mounts | Employed in HIV+ patient study for Giardia and Entamoeba detection [39] |
| PCR reagents (Taq polymerase, dNTPs, buffers) | Molecular detection | Amplification of species-specific genetic targets | Standard component in all cited molecular studies [38] [36] [39] |
| Species-specific primers (SSU rRNA, bg, ITS genes) | Molecular detection | Targeted amplification of pathogen-specific genetic markers | Critical for genotyping in Heilongjiang and other molecular studies [36] |
| Immunochromatographic tests (ICT) | Rapid detection | Antigen-based detection of specific pathogens | Used in Peruvian study for Cryptosporidium, Giardia, Entamoeba [39] |
Transmission Pathways and Public Health Implications
Zoonotic transmission of intestinal protozoa occurs through multiple pathways, with significant public health implications, particularly in vulnerable populations.
Transmission Dynamics
The primary transmission route for these protozoan pathogens is fecal-oral, through direct or indirect contact with contaminated materials or infection sources [38]. Epidemiological surveillance and case-control studies have confirmed that cattle can be a source for Cryptosporidium and Giardia species and genotypes infectious to humans, establishing these animals as zoonotic reservoirs [21] [40] [41]. The distribution of these pathogens and the extent of zoonotic transmission vary across different geographical regions worldwide.
Risk Factors and Vulnerable Populations
Several factors influence infection risks and outcomes:
- Immunocompromised Status: Intestinal protozoa are a common cause of morbidity in people living with HIV (PWH), particularly in tropical regions with poor sanitation. Cryptosporidiosis is classified as an AIDS-defining illness, predominantly affecting PWH with profound immunosuppression, where it can lead to severe, life-threatening diarrheal disease [39].
- Environmental and Socioeconomic Factors: Limited access to potable water, poor sanitation infrastructure, and high poverty levels increase transmission risk. Studies in the Peruvian Amazon found a 51.4% prevalence of any intestinal protozoa among PWH, with Cryptosporidium prevalence at 25.7% [39].
- Agricultural Practices: Intensive farming practices, particularly industrial-scale, high-density operations, create ideal conditions for pathogens to evolve, spread, and potentially jump from animals to humans [37]. These settings confine large numbers of genetically similar animals in small spaces, facilitating rapid transmission of infectious agents.
Figure 2: Zoonotic Transmission Pathways of Intestinal Protozoa from Domestic Animals to Humans
Cattle and other domestic animals serve as significant reservoirs for zoonotic transmission of intestinal protozoa, with substantial public health implications globally. The age-dependent prevalence patterns observed in calves, particularly the high concentration of Cryptosporidium infections in neonates, highlight critical windows for intervention. Molecular characterization of circulating genotypes reveals both host-adapted and zoonotic strains, emphasizing the need for ongoing surveillance. The experimental methodologies detailed herein provide robust frameworks for detection and characterization, essential for understanding transmission dynamics and developing targeted control strategies. A One Health approach, integrating human, animal, and environmental health perspectives, remains fundamental to mitigating the burden of these zoonotic pathogens, particularly in vulnerable populations and resource-limited settings where the disease burden is highest.
From Bench to Outbreak: Advanced Diagnostic and Modeling Applications
The diagnosis of intestinal protozoa is undergoing a fundamental transformation, moving from morphology-based microscopic identification toward sophisticated immunodiagnostic and molecular detection platforms. This evolution is particularly critical within zoonotic transmission pathway research, where accurately identifying pathogenic species and understanding their genetic diversity directly impacts public health outcomes. Traditional microscopic examination, while widely available, faces significant limitations including suboptimal sensitivity, inability to differentiate morphologically identical species, and reliance on skilled microscopists [42]. These challenges are especially pronounced in zoonotic intestinal protozoa research, where distinguishing human-pathogenic strains from non-pathogenic variants or animal-specific genotypes is essential for tracking transmission routes and implementing targeted control measures [43] [44].
The emergence of immunodiagnostic and molecular methods has substantially improved diagnostic precision for intestinal protozoa including Cryptosporidium spp., Giardia duodenalis, Entamoeba histolytica, and Blastocystis sp.—all of which exhibit significant zoonotic potential [43] [12] [45]. These advanced platforms enable not only species-specific identification but also genotyping and tracking of transmission pathways across human and animal hosts. As molecular characterization studies reveal, domestic animals including small ruminants, dogs, and cats often harbor zoonotic protozoan genotypes, acting as reservoirs for human infection [43] [44] [12]. This technical guide comprehensively explores the current immunodiagnostic and molecular detection platforms, their applications in zoonotic research, and detailed experimental methodologies for investigating intestinal protozoan transmission pathways.
Immunodiagnostic Platforms
Immunodiagnostic methods detect parasite-specific antigens or host antibodies through antibody-antigen interactions, offering improved specificity over conventional microscopy. These platforms are particularly valuable for differentiating pathogenic from non-pathogenic species and for high-throughput screening in epidemiological studies [42].
Core Immunoassay Technologies
Enzyme-Linked Immunosorbent Assay (ELISA) platforms utilize antibodies against protozoan antigens and are widely used for intestinal protozoa detection. For E. histolytica, monoclonal antibody-based ELISA kits targeting the Gal/GalNAc lectin antigen demonstrate sensitivities of 80-94% compared to polymerase chain reaction (PCR) [42]. These assays can be configured to detect either antigens during active intestinal infection or anti-Entamoeba antibodies during extraintestinal amoebic liver abscess (ALA) [42]. Rapid Immunochromatographic Tests (ICTs) provide point-of-care capabilities with results in 15-30 minutes, making them suitable for field studies and clinical settings with limited resources. Direct Fluorescent Antibody (DFA) assays, combining antibody staining with fluorescent microscopy, offer high sensitivity and are considered a reference method for Giardia and Cryptosporidium detection [42].
Target Antigens and Performance Characteristics
Table 1: Key Immunodiagnostic Targets for Zoonotic Intestinal Protozoa
| Parasite | Target Antigen | Platforms | Sensitivity | Specificity | Zoonotic Significance |
|---|---|---|---|---|---|
| Entamoeba histolytica | Gal/GalNAc lectin | ELISA, ICT | 80-94% | High | Differentiates pathogenic E. histolytica from non-pathogenic E. dispar |
| Giardia duodenalis | Cyst wall protein, Surface antigens | DFA, ELISA, ICT | 66-100% | High | Detects assemblages with zoonotic potential (A, B) |
| Cryptosporidium spp. | Oocyst wall antigens | DFA, ELISA, ICT | >95% (DFA) | High | Identifies zoonotic species (C. parvum, C. hominis) |
| Blastocystis sp. | Surface proteins | ELISA (research use) | Variable | Moderate | Differentiates potentially pathogenic subtypes |
Advantages and Limitations in Zoonotic Research
Immunodiagnostic methods offer several advantages for zoonotic research, including relatively low cost, technical simplicity, and rapid turnaround time. The high throughput capability of ELISA makes it suitable for large-scale prevalence studies in human and animal populations [42]. However, these methods have important limitations: cross-reactivity between related species can occur, potentially leading to false positives in multi-species infections [42]. Additionally, antigenic diversity among different strains and genotypes may affect test performance, and most commercial assays cannot provide genotyping information crucial for tracking transmission pathways [42] [44].
Molecular Detection Platforms
Molecular diagnostics detect parasite-specific nucleic acid sequences, offering superior sensitivity and specificity while enabling genotype differentiation essential for understanding zoonotic transmission dynamics [42] [44].
Core Molecular Technologies
Polymerase Chain Reaction (PCR) and its variants represent the cornerstone of molecular detection for intestinal protozoa. Conventional PCR allows targeted amplification of specific DNA sequences, while multiplex PCR enables simultaneous detection of multiple pathogens in a single reaction [46]. Real-time PCR (qPCR) provides quantitative data on parasite load and eliminates post-amplification steps, reducing contamination risk. Next-Generation Sequencing (NGS) platforms offer comprehensive analysis of entire genomic regions, enabling identification of mixed infections, novel genotypes, and genetic diversity studies across human and animal hosts [47] [46].
Molecular Targets and Genotyping Applications
Table 2: Molecular Targets for Zoonotic Intestinal Protozoa Detection and Genotyping
| Parasite | Genetic Targets | Methodologies | Genotyping Resolution | Zoonotic Applications |
|---|---|---|---|---|
| Cryptosporidium spp. | SSU rRNA, gp60, COWP | Nested PCR, qPCR, NGS | Species and subtype level | Distinguishes C. hominis (human) from C. parvum (zoonotic); tracks outbreaks |
| Giardia duodenalis | SSU rRNA, bg, gdh, tpi | Multiplex PCR, qPCR, MLST | Assemblage level | Differentiates zoonotic assemblages (A, B) from host-specific ones |
| Entamoeba histolytica | SSU rRNA, chitinase | Conventional PCR, qPCR | Species level | Differentiates from morphologically identical E. dispar, E. moshkovskii |
| Blastocystis sp. | SSU rRNA | PCR, NGS | Subtype level (ST1-ST17) | Identifies zoonotic subtypes circulating between humans and animals |
Performance Characteristics and Research Applications
Molecular methods demonstrate significantly improved sensitivity compared to conventional microscopy, particularly for low-intensity infections. A study in Colombia found PCR detection substantially improved Cryptosporidium spp. and Blastocystis spp. detection and allowed differentiation of E. histolytica from commensals in the Entamoeba complex [44]. In HIV/AIDS patients, molecular methods revealed much higher prevalence of intestinal protozoa compared to conventional methods, detecting 22% Blastocystis, 17% microsporidia, 12% Cryptosporidium spp., and 11% G. intestinalis [45]. Molecular genotyping enables precise tracking of zoonotic transmission pathways, as demonstrated in studies from China where domestic small ruminants were found to harbor zoonotic genotypes of Cryptosporidium spp. (C. ubiquitum), G. duodenalis (assemblage A), and Blastocystis sp. (ST5) [43].
Integrated Experimental Protocols
This section provides detailed methodologies for comprehensive detection, differentiation, and genotyping of zoonotic intestinal protozoa in research settings.
Sample Collection and DNA Extraction Protocol
Sample Collection: Collect fresh fecal samples (approximately 5-10g) from human and animal hosts using sterile containers. For cross-sectional studies, include epidemiological data including host demographics, clinical symptoms, environmental factors, and animal contact history. Process samples within 24 hours or preserve in appropriate storage buffers (ethanol, RNAlater) for molecular studies or specific fixatives for immunodiagnostics [44] [12].
DNA Extraction: Use commercial fecal DNA extraction kits following manufacturer's protocols with modifications to enhance protozoan DNA recovery: (1) Include bead-beating step (0.5mm glass beads) for 3-5 minutes to disrupt robust cyst/oocyst walls; (2) Incorporate proteinase K digestion (1-2 hours at 56°C) to enhance cell lysis; (3) Include inhibitor removal steps, particularly for animal-derived samples. Elute DNA in 50-100μL elution buffer and quantify using spectrophotometry. Store extracted DNA at -20°C for short-term or -80°C for long-term preservation [44] [45].
Multiplex PCR Protocol for Simultaneous Protozoa Detection
Primer Design: Select primers targeting conserved regions of specific genes: Cryptosporidium (SSU rRNA, ~132bp), G. duodenalis (bg gene, ~175bp), E. histolytica (SSU rRNA, ~100bp), and Blastocystis (SSU rRNA, ~110bp). Design primers with similar annealing temperatures (55-60°C) and non-overlapping amplicon sizes for clear differentiation by gel electrophoresis [44].
Reaction Setup: Prepare 25μL reaction mixture containing: 2.5μL 10X PCR buffer, 1.5μL MgCl₂ (25mM), 0.5μL dNTPs (10mM each), 0.5μL each forward and reverse primers (10μM), 0.2μL Taq polymerase (5U/μL), 2μL DNA template, and nuclease-free water to volume. Include positive controls (reference DNA for each target) and negative controls (no-template) in each run [44] [45].
Amplification Conditions: Initial denaturation: 95°C for 5 minutes; 35-40 cycles of: denaturation at 95°C for 30 seconds, annealing at 58°C for 45 seconds, extension at 72°C for 1 minute; final extension at 72°C for 7 minutes. Analyze products by gel electrophoresis (2% agarose, 100V for 45 minutes) with appropriate molecular weight markers [44].
Molecular Genotyping and Phylogenetic Analysis Protocol
Nested PCR for Cryptosporidium: Perform primary PCR targeting SSU rRNA gene followed by secondary reaction using internal primers for enhanced sensitivity. For subtyping, amplify gp60 gene using nested PCR with specific annealing temperatures for different Cryptosporidium species [43].
Sequence-Based Genotyping: Purify PCR products using commercial cleanup kits. Prepare sequencing reactions with BigDye Terminator mix and run on capillary sequencers. Analyze sequence chromatograms for quality assessment, then align with reference sequences using bioinformatics software (MEGA, Geneious). Construct phylogenetic trees using neighbor-joining or maximum likelihood methods with bootstrap analysis (1000 replicates) to determine genetic relationships between isolates from different hosts [43] [44].
Next-Generation Sequencing: For comprehensive analysis, prepare libraries using Illumina-compatible protocols with dual indexing. Sequence on appropriate platforms (Illumina MiSeq, NovaSeq) with sufficient coverage (minimum 50,000 reads per sample). Process raw data through quality filtering, chimera removal, and cluster into operational taxonomic units (OTUs) at 97% similarity. Assign taxonomy against curated protozoan databases and analyze for subtype distribution across host species [43].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for Zoonotic Intestinal Protozoa Detection
| Reagent Category | Specific Examples | Research Application | Technical Notes |
|---|---|---|---|
| DNA Extraction Kits | QIAamp DNA Stool Mini Kit, PowerSoil DNA Isolation Kit | Efficient DNA recovery from fecal samples | Include bead-beating step for cyst/oocyst disruption; critical for PCR efficiency |
| PCR Master Mixes | HotStart Taq Master Mix, Q5 High-Fidelity DNA Polymerase | Amplification of target genes | Select based on fidelity requirements; HotStart reduces primer-dimer formation |
| Species-Specific Primers | SSU rRNA, gp60, bg, tpi gene targets | Species identification and genotyping | Validate against reference strains; design for appropriate specificity level |
| Positive Controls | Reference DNA from ATCC, well-characterized clinical isolates | Assay validation and quality control | Essential for each run; confirms reaction efficiency and specificity |
| Electrophoresis Reagents | Agarose, SYBR Safe, DNA ladders | PCR product visualization | Use high-resolution agarose (2-3%) for discriminating similar amplicon sizes |
| Sequencing Reagents | BigDye Terminator v3.1, POP-7 Polymerase | Sanger sequencing of PCR products | Purify PCR products before sequencing to improve read quality |
| Immunoassay Kits | ELISA for Cryptosporidium, Giardia, E. histolytica | High-throughput screening, antigen detection | Useful for large studies; confirm positives with molecular methods |
| Microscopy Reagents | Trichrome stain, Modified acid-fast stain | Reference method comparison, morphology studies | Despite limitations, provides context for molecular findings |
Data Interpretation and Zoonotic Significance
Accurate interpretation of diagnostic data is crucial for understanding zoonotic transmission pathways and implementing appropriate public health interventions.
Genetic Data Analysis for Zoonotic Assessment
Phylogenetic analysis of sequence data reveals genetic relationships between isolates from different hosts and geographic locations. In studies of domestic small ruminants in China, identification of Cryptosporidium ubiquitum and G. duodenalis assemblage A (both with zoonotic potential) highlighted the role of these animals as reservoirs for human infection [43]. Similarly, research in Colombia demonstrated identical Cryptosporidium genotypes in humans and their companion animals, providing evidence of cross-species transmission [44]. Statistical analysis incorporating risk factors (water source, sanitation, animal contact) strengthens the evidence for zoonotic transmission pathways [12].
Diagnostic Test Performance in Research Settings
Understanding test characteristics is essential for appropriate method selection and data interpretation. Molecular methods consistently demonstrate superior sensitivity compared to conventional microscopy, particularly for low-intensity infections and chronic cases. In HIV/AIDS patients, molecular detection revealed much higher prevalence of intestinal protozoa than conventional methods, with 22% Blastocystis, 17% microsporidia, 12% Cryptosporidium spp., and 11% G. intestinalis [45]. The high sensitivity of PCR-based methods makes them particularly valuable for surveillance studies and detecting subclinical infections in animal reservoirs [44].
The evolution beyond microscopy to immunodiagnostic and molecular platforms has fundamentally transformed intestinal protozoa research, particularly in understanding and tracking zoonotic transmission pathways. These advanced detection methods provide the specificity, sensitivity, and genotyping resolution necessary to unravel complex transmission dynamics between human and animal hosts. As research continues to reveal the extensive genetic diversity and host adaptability of intestinal protozoa, the integration of these platforms—complemented by sophisticated bioinformatics analysis—will be essential for developing evidence-based control strategies to reduce the burden of zoonotic protozoan infections globally.
The accurate identification of pathogenic species is a cornerstone of modern infectious disease research and control. This is particularly critical for intestinal protozoa, where morphologically identical species can have vastly different pathogenic potentials. Polymerase Chain Reaction (PCR)-based assays have emerged as the definitive tool for this task, providing the species-level differentiation necessary to unravel complex zoonotic transmission pathways [48] [49]. The shift from traditional microscopy to molecular methods represents a paradigm change in diagnostic parasitology, moving beyond mere detection to precise genetic characterization [49].
This technical guide explores the design, validation, and application of PCR-based assays for the specific identification of pathogens, with a focus on intestinal protozoa within the context of zoonotic disease research. The ensuing sections provide a detailed examination of the core principles that underpin assay specificity, present standardized protocols, and discuss the integration of these tools into a One Health framework to understand and mitigate cross-species transmission.
Core Principles of Species-Specific PCR Assay Design
The power of PCR for species-specific identification hinges on strategic assay design targeting genetic regions that exhibit sufficient variation to discriminate between species.
Genetic Targets and Primer/Probe Design
The initial and most critical step involves selecting an appropriate genetic marker. The small subunit ribosomal RNA (SSU rRNA) gene is a predominant target due to its multicopy nature, which enhances sensitivity, and the presence of both highly conserved and variable regions [48] [49]. Conserved regions facilitate the design of broad-range primers, while variable sequences allow for species-specific probe design or primer targeting.
Design criteria for primers and probes must be rigorously followed to ensure optimal assay performance. Key parameters include:
- GC content of approximately 50% [48].
- A length of 20-24 bases [48].
- An estimated melting temperature (Tm) of ~58°C [48].
Furthermore, the selection of dyes and quenchers for probe-based assays (e.g., TaqMan) is determined by the detection capabilities of the real-time PCR instrumentation available [48]. A BLASTN search is an indispensable final step to confirm the uniqueness of the selected primer and probe sequences, thereby minimizing the risk of cross-reactivity with non-target organisms [48].
Overcoming Diagnostic Challenges
A significant challenge in molecular diagnostics is the extensive intraspecific diversity found in many protozoan parasites [49]. Assays designed from limited sequence data may fail to detect divergent strains of the same species, leading to false negatives. This underscores the necessity of designing primers and probes based on comprehensive genetic databases that capture the existing diversity of the target organism [49].
Another common pitfall is cross-reactivity with non-target species. As evidenced in research on Dientamoeba fragilis, assays developed for human diagnostics can produce false positives when applied to animal specimens due to non-specific amplification of DNA from related organisms, such as Simplicimonas sp. [50]. Melt curve analysis is a valuable technique to identify such cross-reactivity, as non-target amplification often produces a distinct melt curve temperature compared to the true target [50].
Table 1: Key Genetic Targets for Common Intestinal Protozoa
| Organism | Primary Genetic Target | Key Differentiable Species |
|---|---|---|
| Entamoeba histolytica/dispar | Small subunit ribosomal RNA gene [48] | Pathogenic E. histolytica from non-pathogenic E. dispar [48] |
| Giardia duodenalis | Small subunit ribosomal RNA gene [48] | Differentiates Assemblages with zoonotic potential [49] |
| Cryptosporidium spp. | Small subunit ribosomal RNA gene [48] | Species and genotypes with varying host specificities |
| Blastocystis spp. | Small subunit ribosomal RNA gene [48] | Multiple subtypes with debated pathogenicity |
| Chilomastix mesnili | 18S ribosomal RNA [48] | Non-pathogenic indicator of fecal contamination |
Experimental Protocols for Pathogen Differentiation
This section outlines a standardized workflow and detailed methodology for setting up a duplex qPCR assay, providing a template for specific pathogen detection.
Standardized Workflow for Species-Specific qPCR
The following diagram illustrates the generalized workflow for developing and executing a species-specific qPCR assay, from initial design to final analysis.
Detailed Duplex qPCR Protocol for Intestinal Protozoa
The protocol below is adapted from a recent study implementing duplex assays for the detection of Entamoeba species and Cryptosporidium spp. + Chilomastix mesnili [48].
1. Primer and Probe Preparation:
- Reconstitute lyophilized primers and probes in nuclease-free water to create 100 µM stock solutions.
- Prepare working primer mixes for each duplex assay. For example, for the Entamoeba histolytica + E. dispar assay, combine forward and reverse primers for both targets to a final concentration of 0.5 µM each in the reaction mix [48].
- Prepare working probe mixes labeled with distinct fluorophores (e.g., FAM for one target, HEX/VIC for the other).
2. qPCR Reaction Setup:
- Use a total reaction volume of 10 µL [48].
- The reaction mix should contain:
- 5 µL of 2x qPCR Master Mix (containing DNA polymerase, dNTPs, and MgCl₂).
- 1 µL of the combined primer working mix (final concentration 0.5 µM each primer).
- 0.5 µL of the combined probe working mix.
- 2.5 µL of nuclease-free water.
- 1 µL of template DNA.
3. Thermal Cycling Conditions:
- Initial denaturation: 95°C for 2-5 minutes.
- 40-45 cycles of:
- Denaturation: 95°C for 15 seconds.
- Annealing/Extension: 60°C for 1 minute (acquire fluorescence signal at this step).
4. Data Analysis:
- Determine cycle threshold (Ct) values for each sample and fluorophore channel.
- A sample is considered positive if the Ct value is below a predetermined cutoff (e.g., Ct < 40) and the amplification curve exhibits a characteristic sigmoidal shape.
Validation and Troubleshooting
Assay Validation:
- Analytical Specificity: Test the assay against a panel of DNA from closely related non-target species to confirm no cross-reactivity [50].
- Analytical Sensitivity: Determine the limit of detection (LOD) using a standard curve created from serial dilutions of a known quantity of target DNA or a synthetic plasmid [49].
- Clinical Validation: Evaluate assay performance using a set of well-characterized clinical samples, comparing results to a reference standard.
Addressing Cross-Reactivity:
- If melt curve analysis reveals unexpected peaks or shifts in Tm, suspect cross-reactivity [50].
- Confirm the identity of the amplified product by Sanger sequencing of the qPCR amplicon.
- Redesign primers or probes if cross-reactivity with a non-target organism is confirmed [49] [50].
Table 2: Research Reagent Solutions for PCR-Based Pathogen Differentiation
| Reagent/Category | Specific Example | Function in Assay |
|---|---|---|
| Primers | E. histolytica SSU rDNA Forward & Reverse [48] | Binds to specific flanking sequences to initiate targeted DNA amplification. |
| Probes | TaqMan probe with 5' FAM dye [48] | Provides species-level specificity through hybridization; emits fluorescence upon cleavage for real-time detection. |
| Enzyme Master Mix | 2x qPCR Probe Master Mix | Contains thermostable DNA polymerase, dNTPs, and optimized buffer for efficient amplification. |
| Internal Control | Exogenous DNA from qPCR Extraction Control Kit [50] | Monitors DNA extraction efficiency and detects PCR inhibitors in the sample. |
| Positive Control | Plasmid with cloned target sequence [49] | Verifies successful assay performance and provides a basis for quantification. |
Quantitative Analysis and Data Interpretation
Moving from qualitative detection to quantitative analysis provides deeper insights into pathogen burden, which is crucial for understanding disease severity and transmission dynamics.
Absolute Quantification with Internal Controls
For absolute quantification of pathogen load in complex samples like stool, the use of spike-in internal controls (ICs) is essential. These controls correct for variations in DNA extraction efficiency and the presence of PCR inhibitors. A 2023 study demonstrated that Thermus thermophilus (at 10³ CFU/mL) and T1 phage (at 10⁴ PFU/mL) are highly effective as mNGS ICs for pathogen quantitation, outperforming artificial DNA sequences in accuracy, linearity, and resistance to interference from human cell concentrations [51]. The same principles apply to qPCR, where the ratio of pathogen reads to IC reads can be used to calculate the absolute concentration of the pathogen in the original sample.
Applications in Zoonotic Transmission Research
The application of these molecular tools in field studies is elucidating the scope of zoonotic parasite transmission.
Table 3: Prevalence of Zoonotic Intestinal Protozoa in Select Populations
| Location | Study Population | Pathogen | Prevalence | Detection Method |
|---|---|---|---|---|
| Pemba Island, Tanzania [48] | 70 patients | Entamoeba histolytica/dispar | 31.4% | Duplex qPCR |
| Entamoeba histolytica (pathogenic) | ~10.5% (1/3 of positives) | Duplex qPCR | ||
| Ecuadorian Coast [12] | 160 humans | Entamoeba histolytica | 10.0% | Coproparasitic techniques |
| Hebei Province, China [15] | Wildlife (Leopard Cat) | Giardia intestinalis (Assemblage F) | Detected | PCR & Sequencing |
| Marginalized Ecuadorian Communities [12] | 500 domestic dogs | Zoonotic parasites (e.g., Ancylostoma) | 78.0% overall | Coproparasitic techniques |
Quantitative data from such studies reveal transmission hotspots. For instance, the high prevalence of Entamoeba histolytica in humans in Ecuador [12], coupled with the detection of Giardia in wildlife in China [15], highlights ecosystems where human-animal-parasite interactions are frequent. The high overall parasite burden in domestic dogs in Ecuador [12] further identifies them as potential reservoirs for zoonotic transmission.
PCR-based assays provide an powerful and precise methodology for the species-specific identification of intestinal protozoa, directly contributing to the understanding of their zoonotic transmission pathways. The strategic design of primers and probes, coupled with robust validation and the use of internal controls for quantification, allows researchers to accurately map the distribution and burden of pathogenic species across human and animal populations.
Future advancements will likely involve the broader implementation of multiplex PCR panels capable of simultaneously detecting dozens of pathogens and antimicrobial resistance genes [52]. Furthermore, the integration of metagenomic next-generation sequencing (mNGS) and digital PCR (dPCR) will enhance the capacity for discovery-based detection of novel pathogens and absolute quantification of low-abundance targets, respectively [51] [52]. As these technologies mature and become more accessible, they will be indispensable tools for global One Health initiatives aimed at tracking, controlling, and ultimately preventing the cross-species transmission of intestinal pathogens.
Mathematical modeling serves as a fundamental tool in epidemiology, enabling researchers and public health officials to understand disease transmission dynamics, predict outbreak trajectories, and evaluate intervention strategies. Compartmental models form the cornerstone of theoretical epidemiology by dividing a population into distinct segments based on disease status. The SIR model, introduced by Kermack and McKendrick, represents one of the simplest and most widely adopted frameworks, categorizing individuals as Susceptible (S), Infected (I), or Removed (R) [53]. This model has provided significant theoretical and methodological support for the development of infectious disease modeling and continues to be widely utilized today [53].
For diseases exhibiting different transmission characteristics, several model variants have been developed. The SIRS model extends the classic SIR framework by introducing the concept of "immunity decay," assuming that recovered individuals gradually lose immunity and become susceptible again [53]. The SEIR model incorporates an additional "Exposed" compartment to account for the incubation period of a pathogen, during which individuals are infected but not yet infectious [53] [54]. This model has demonstrated superior performance in simulating diseases with significant latency periods, achieving maximum relative errors of 4.81% and 5.60% in regional COVID-19 simulations, outperforming both SIS and SIR models [54].
In the context of zoonotic intestinal protozoa, these modeling frameworks require careful adaptation to account for complex transmission pathways between human and animal populations, environmental contamination, and unique parasite life cycle characteristics [55] [56]. Intestinal protozoan parasites, including Cryptosporidium spp., Giardia duodenalis, Entamoeba histolytica, and Blastocystis sp., pose significant global health challenges, particularly in developing regions with limited access to sanitation and healthcare resources [57] [19]. The One Health perspective emphasizes the interconnectedness of human, animal, and environmental health in understanding and controlling these zoonotic diseases [56].
Fundamental Model Structures and Formulations
The Basic SIR Model
The SIR model operates on a fundamental system of ordinary differential equations that describe the flow of individuals between compartments over time [54]:
$$ \begin{aligned} \frac{dS}{dt} &= -\beta \cdot S \cdot \frac{I}{N} \ \frac{dI}{dt} &= \beta \cdot S \cdot \frac{I}{N} - \gamma \cdot I \ \frac{dR}{dt} &= \gamma \cdot I \end{aligned} $$
Where:
- ( S ) represents the susceptible population
- ( I ) represents the infected population
- ( R ) represents the removed population (recovered with immunity or deceased)
- ( N ) represents the total population (( N = S + I + R ))
- ( \beta ) represents the transmission rate
- ( \gamma ) represents the recovery rate
The basic reproduction number (( R0 )), a critical epidemiological parameter, is derived as ( R0 = \frac{\beta}{\gamma} ) for the SIR model. This value represents the average number of secondary infections generated by a single infected individual in a completely susceptible population [55]. If ( R0 < 1 ), the disease will eventually die out, while if ( R0 > 1 ), the disease can spread through the population [55].
The SEIR Model with Incubation Period
For diseases with significant incubation periods, including many parasitic infections, the SEIR model provides a more realistic representation by incorporating an exposed (E) compartment [53] [54]:
$$ \begin{aligned} \frac{dS}{dt} &= -\beta \cdot S \cdot \frac{I}{N} \ \frac{dE}{dt} &= \beta \cdot S \cdot \frac{I}{N} - \delta \cdot E \ \frac{dI}{dt} &= \delta \cdot E - \gamma \cdot I \ \frac{dR}{dt} &= \gamma \cdot I \end{aligned} $$
Where ( \delta ) represents the rate at which exposed individuals become infectious (the inverse of the incubation period). This structure is particularly relevant for intestinal protozoan infections, which often have variable incubation periods depending on the specific parasite and host factors [57].
Extended Compartmental Models for Complex Transmission Dynamics
For zoonotic intestinal protozoa with complex life cycles and multiple transmission routes, basic SIR and SEIR models require significant expansion. These parasites typically involve multiple host species, environmental stages, and sometimes vectors or mechanical transporters [55] [58]. Cryptosporidium spp. and Giardia duodenalis, for instance, can be transmitted through direct contact with infected humans or animals, consumption of contaminated food or water, or contact with contaminated environments [57] [19].
The diagram below illustrates the complex transmission dynamics of zoonotic intestinal protozoa between multiple host species and the environment:
Table 1: Key Parameters in Basic SIR and SEIR Models
| Parameter | Biological Meaning | Typical Range for Protozoa | Units |
|---|---|---|---|
| ( \beta ) | Transmission rate | Highly variable (0.1-5.0) | time⁻¹ |
| ( \gamma ) | Recovery rate | 0.05-0.3 (depending on treatment) | time⁻¹ |
| ( \delta ) | Progression rate from exposed to infectious | 0.1-0.5 (depending on parasite) | time⁻¹ |
| ( R_0 ) | Basic reproduction number | 0.3-4.0 (context-dependent) | dimensionless |
| ( 1/\gamma ) | Infectious period | 3-20 days | time |
| ( 1/\delta ) | Incubation period | 2-10 days | time |
Model Adaptations for Zoonotic Intestinal Protozoa
Multi-Host Transmission Models
Zoonotic intestinal protozoa, including Giardia duodenalis, Cryptosporidium parvum, C. hominis, and Cyclospora cayetanensis, circulate between human and animal populations, necessitating modeling frameworks that explicitly account for multiple host species [57] [56]. The SIREH model (Susceptible-Infected-Recovered-Environment-Humans) represents one such adaptation, incorporating environmental contamination as a key component of transmission dynamics.
For intestinal protozoa with wildlife reservoirs, such as Giardia intestinalis detected in leopard cats and Colpodella spp. in red foxes [15], the model structure must account for the interaction between sylvatic cycles and human populations. A study on laboratory macaques, animal facility workers, and nearby villagers revealed a 37.2% overall prevalence of pathogenic intestinal protozoa, with significantly higher infection rates (56.0%) in facility workers who had direct contact with macaques compared to villagers (28.0%) [56]. This highlights the importance of occupational exposure in zoonotic transmission.
The following system of equations represents a simplified two-host SIR model for zoonotic protozoa:
$$ \begin{aligned} \frac{dSh}{dt} &= -\beta{hh} \cdot Sh \cdot \frac{Ih}{Nh} - \beta{ha} \cdot Sh \cdot \frac{Ia}{Na} \ \frac{dIh}{dt} &= \beta{hh} \cdot Sh \cdot \frac{Ih}{Nh} + \beta{ha} \cdot Sh \cdot \frac{Ia}{Na} - \gammah \cdot Ih \ \frac{dSa}{dt} &= -\beta{aa} \cdot Sa \cdot \frac{Ia}{Na} - \beta{ah} \cdot Sa \cdot \frac{Ih}{Nh} \ \frac{dIa}{dt} &= \beta{aa} \cdot Sa \cdot \frac{Ia}{Na} + \beta{ah} \cdot Sa \cdot \frac{Ih}{Nh} - \gammaa \cdot Ia \end{aligned} $$
Where subscripts ( h ) and ( a ) denote human and animal populations, respectively, and cross-species transmission rates ( \beta{ha} ) and ( \beta{ah} ) capture the zoonotic potential.
Environmental Transmission Components
Many intestinal protozoa produce environmentally resistant stages (cysts, oocysts) that can persist in water, soil, and on surfaces for extended periods [57]. Giardia duodenalis cysts, for example, can survive in cold water for up to 3 months [57], while Cryptosporidium oocysts are highly resistant to chlorine disinfection [57]. This environmental persistence necessitates the addition of an environmental compartment (W) to track pathogen load:
$$ \begin{aligned} \frac{dW}{dt} &= \xi \cdot (Ih + Ia) - \mu \cdot W - \alpha \cdot N \cdot W \ \frac{dS}{dt} &= -\beta \cdot S \cdot \frac{I}{N} - \alpha \cdot \omega \cdot S \cdot W \end{aligned} $$
Where:
- ( W ) represents the environmental pathogen load
- ( \xi ) represents the shedding rate of pathogens by infected hosts
- ( \mu ) represents the pathogen decay rate in the environment
- ( \alpha ) represents the exposure rate to contaminated environments
- ( \omega ) represents the probability of infection given exposure
Table 2: Environmental Parameters for Zoonotic Intestinal Protozoa Modeling
| Parameter | Biological Meaning | Giardia duodenalis | Cryptosporidium spp. | Entamoeba histolytica |
|---|---|---|---|---|
| Shedding rate (( \xi )) | Pathogens released per infected host per day | 10⁶-10¹⁰ cysts/day | 10⁷-10⁹ oocysts/day | 10⁵-10⁸ cysts/day |
| Infectious dose (ID₅₀) | Dose required to infect 50% of hosts | 10-100 cysts | 10-1000 oocysts | 100-1000 cysts |
| Environmental survival (( 1/\mu )) | Pathogen persistence in environment | Weeks to months | Weeks to months | Days to weeks |
| Chlorine resistance | Resistance to water disinfection | Moderate | High | Low |
Advanced Modeling Frameworks and Methodological Approaches
Improved SEIR Model with Asymptomatic Carriage and Intervention Measures
For accurate modeling of intestinal protozoa transmission, the classical SEIR framework requires expansion to account for asymptomatic infections, intervention measures, and seasonal fluctuations. An improved SEIR model for COVID-19 [59] demonstrates methodological approaches applicable to parasitic diseases, incorporating quarantined susceptible (Sq) and quarantined exposed (Eq) compartments to simulate public health interventions.
The model structure incorporates the following enhancements:
$$ \begin{aligned} \frac{dS}{dt} &= -\beta c(1-q)S\frac{I}{N} - \beta cqS\frac{I}{N} + \lambda Sq \ \frac{dSq}{dt} &= (1-\beta)cqS\frac{I}{N} - \lambda Sq \ \frac{dE}{dt} &= \beta c(1-q)S\frac{I}{N} - \delta E \ \frac{dEq}{dt} &= \beta cqS\frac{I}{N} - \deltaq Eq \ \frac{dI}{dt} &= \delta E - \gamma I \ \frac{dR}{dt} &= \gamma I \end{aligned} $$
Where:
- ( q ) represents the tracking and quarantine ratio
- ( c ) represents the contact rate
- ( \lambda ) represents the release rate from quarantine
- ( \delta_q ) represents the progression rate of quarantined exposed individuals
This framework can be adapted for intestinal protozoa by incorporating treatment interventions, water sanitation improvements, and hygiene education programs as control measures.
Quantum Mechanics-Based Framework for Infectious Disease Modeling
Recent innovations in epidemiological modeling include the integration of quantum principles to address limitations of traditional compartmental models [53]. The Quantum Healthy-Infected Model (QHIM) replaces deterministic state transitions with quantum superposition states, allowing individuals to exist simultaneously in multiple health states until "observation" or measurement occurs [53].
In QHIM, an individual's health state is represented as:
$$ |\psi\rangle = a|0\rangle + b|1\rangle $$
Where:
- ( |0\rangle ) represents the healthy ground state
- ( |1\rangle ) represents the infected ground state
- ( a ) and ( b ) are complex coefficients satisfying ( |a|^2 + |b|^2 = 1 )
- ( |a|^2 ) represents the probability of being healthy
- ( |b|^2 ) represents the probability of being infected
Infection processes are simulated using quantum gate operations, particularly the controlled rotation gate ( CRY(\theta) ), which modifies the superposition state based on interaction between susceptible and infected individuals [53]:
$$ CRY(\theta) = |0\rangle\langle 0| \otimes E + |1\rangle\langle 1| \otimes RY(\theta) $$
Where ( RY(\theta) ) is the rotation gate defined as:
$$ RY(\theta) = \begin{bmatrix} \cos(\frac{\theta}{2}) & -\sin(\frac{\theta}{2}) \ \sin(\frac{\theta}{2}) & \cos(\frac{\theta}{2}) \end{bmatrix} $$
This approach allows for continuous representation of infection probabilities and more nuanced modeling of individual heterogeneity in susceptibility and infectiousness.
Spatial and Regional Modeling Approaches
The incorporation of spatial heterogeneity significantly improves model accuracy by accounting for geographic variability in population density, environmental conditions, and intervention coverage [54]. Regional adaptation of compartmental models using a system of differential equations solved with the fourth-order Runge-Kutta method has demonstrated superior performance in capturing localized transmission dynamics [54].
For intestinal protozoa, spatial modeling must consider:
- Watershed contamination patterns influencing geographic distribution
- Animal migration routes affecting wildlife reservoir distributions
- Socioeconomic factors impacting sanitation infrastructure
- Agricultural practices influencing livestock-to-human transmission
The workflow below illustrates the process for developing and validating spatial models of zoonotic pathogen transmission:
Experimental Protocols and Research Methodologies
Molecular Detection and Genotyping Protocols
Accurate parameterization of transmission models for zoonotic intestinal protozoa requires robust laboratory methods for detection and characterization. Nested PCR protocols targeting specific genetic markers provide the sensitivity and specificity needed for prevalence studies and molecular epidemiology [56].
Standardized protocol for detecting intestinal protozoa in human and animal specimens:
Sample Collection: Collect 200mg of fecal sample into sterile containers with 2.5% (w/v) potassium dichromate for preservation [56].
DNA Extraction: Use commercial stool DNA extraction kits (e.g., E.Z.N.A. Stool DNA Kit) following manufacturer's instructions with modifications for optimal protozoan DNA recovery [56].
Nested PCR Amplification:
- Primary PCR: 25μL reaction volume with 1X PCR buffer, 2.5mM MgCl₂, 200μM dNTPs, 0.2μM each primer, 1.25U DNA polymerase, and 2μL template DNA
- Cycling conditions: Initial denaturation at 94°C for 5min; 35 cycles of 94°C for 45s, annealing at primer-specific temperature for 60s, extension at 72°C for 90s; final extension at 72°C for 10min
- Secondary PCR: Use 1μL of primary PCR product as template with internal primers under similar conditions
Sequencing and Genotype Analysis:
- Purify secondary PCR products
- Sequence using secondary PCR primers
- Edit sequences using DNASTAR software
- Align sequences using ClustalX
- Identify genotypes by comparison with reference sequences in GenBank
Phylogenetic Analysis:
- Construct neighbor-joining or maximum likelihood trees
- Perform bootstrap analysis (1000 replicates)
- Identify zoonotic subtypes and transmission clusters
Table 3: Research Reagent Solutions for Intestinal Protozoa Studies
| Reagent/Category | Specific Product Examples | Application in Protozoa Research | Key Features |
|---|---|---|---|
| DNA Extraction Kits | E.Z.N.A. Stool DNA Kit | Genomic DNA isolation from fecal samples | Efficient inhibition removal, high yield for tough samples |
| PCR Master Mixes | HotStarTaq Master Mix, Q5 High-Fidelity Master Mix | Amplification of parasite DNA | High sensitivity, specificity for nested PCR |
| Molecular Grade Water | Nuclease-Free Water | Dilution of samples and preparation of reagents | DNase/RNase-free, no enzymatic interference |
| Positive Controls | Genomic DNA from reference strains | Quality assurance for molecular assays | Ensures assay sensitivity and specificity |
| Agarose Gels | Standard and high-resolution agarose | Electrophoretic separation of PCR products | Matrix for product visualization and quantification |
| Sequencing Reagents | BigDye Terminator v3.1 | Sanger sequencing of amplified products | Accurate genotype identification |
| Phylogenetic Software | MEGA, ClustalX | Genetic analysis and genotype classification | Identifies transmission patterns and zoonotic subtypes |
Extracellular Vesicle Studies in Host-Parasite Communication
Extracellular vesicles (EVs) have emerged as crucial mediators in host-parasite interactions, carrying diverse molecular cargo that modulates host immune responses and facilitates parasite survival [19]. Experimental investigation of EV-mediated communication involves:
EV Isolation Protocol from Intestinal Protozoa:
Parasite Culture: Axenic culture of protozoa (e.g., Giardia duodenalis, Entamoeba histolytica) in appropriate media under optimal conditions
EV Harvesting:
- Centrifuge culture supernatant at 2,000 × g for 30 minutes to remove cells and debris
- Filter through 0.22μm filters
- Ultracentrifuge at 100,000 × g for 70 minutes at 4°C
- Resuspend EV pellet in sterile PBS
EV Characterization:
- Nanoparticle tracking analysis for size distribution and concentration
- Transmission electron microscopy for morphological assessment
- Western blotting for EV markers (e.g., tetraspanins, heat shock proteins)
Functional Assays:
- Treatment of host cells with parasite-derived EVs
- Measurement of cytokine production (IL-6, TNF-α, IL-10, IL-4) by ELISA
- Assessment of tight junction integrity in intestinal epithelial cells
- Analysis of immune cell recruitment and activation
These methodologies provide critical insights into the molecular mechanisms of pathogenesis and immune modulation, informing more biologically realistic mathematical models of within-host dynamics and population-level transmission.
Applications to Public Health and Disease Control
Intervention Strategy Evaluation
Mathematical models of SIR and SEIR frameworks provide quantitative tools for evaluating the potential impact of various intervention strategies on zoonotic intestinal protozoa transmission. Based on model analyses, the following interventions demonstrate significant potential:
Targeted Chemotherapy: Regular anthelminthic treatment of high-risk groups, using drugs such as metronidazole (500-750mg/day for 5-10 days) or tinidazole (300mg/day for 7 days) for giardiasis [57]. Models can optimize treatment frequency and coverage to achieve community-level protection.
Water Sanitation Improvements: Water filtration systems capable of removing protozoan cysts and oocysts, which are highly resistant to conventional chlorination [57]. Models incorporating environmental transmission components can identify critical contamination points in water systems.
Animal Reservoir Management: Biosecurity measures in laboratory animal facilities and farms, with models showing significantly higher infection rates (56.0%) in workers with direct animal contact compared to nearby villagers (28.0%) [56].
Health Education Programs: Hygiene promotion targeting high-risk behaviors, with models simulating reduced transmission probabilities through improved handwashing and food safety practices.
Outbreak Investigation and Response
Compartmental models facilitate rapid analysis during outbreak situations, enabling estimation of transmission parameters and evaluation of control measures. The improved SEIR model with time-varying parameters allows for real-time adjustment of intervention strategies based on evolving outbreak dynamics [59].
Outbreak Response Protocol:
- Initial Parameter Estimation: Use early outbreak data to estimate ( R_0 ) and generation time
- Model Calibration: Adjust location-specific parameters (contact rates, mixing patterns)
- Intervention Simulation: Project outbreak trajectories under different control scenarios
- Resource Allocation Optimization: Identify critical intervention points for maximum impact
- Real-time Updating: Continuously refine model parameters as new data becomes available
This approach proved particularly valuable during the COVID-19 pandemic [54] [59] and can be similarly applied to outbreaks of zoonotic intestinal protozoa, such as waterborne cryptosporidiosis or foodborne cyclosporiasis.
Integration with Molecular Epidemiology
The combination of mathematical modeling with genotype-specific surveillance data enables more precise tracking of transmission pathways and identification of zoonotic spillover events [56]. Molecular tools permit discrimination between human-specific and zoonotic genotypes, revealing that:
- Only assemblages A and B of Giardia duodenalis are zoonotic [56]
- Specific subtypes of Cryptosporidium parvum (e.g., IIaA15G2R1) demonstrate hyper-transmissibility [56]
- Wildlife, including foxes and leopard cats, harbor zoonotic protozoa with close genetic relationship to human-infecting strains [15]
This integration enables refinement of model structure to account for strain-specific transmission dynamics and differential intervention effectiveness across genetic variants.
SIR and SEIR frameworks provide powerful, adaptable foundations for modeling the transmission dynamics of zoonotic intestinal protozoa. While these basic structures capture essential elements of disease spread, their extension through multi-host compartments, environmental transmission parameters, and spatial heterogeneity significantly enhances their realism and utility for public health decision-making. The integration of molecular epidemiological data, quantum computing approaches, and advanced computational methods continues to expand the applications and accuracy of these modeling frameworks.
For researchers and public health professionals addressing zoonotic intestinal protozoa, the strategic application of appropriately adapted compartmental models offers valuable insights for outbreak management, intervention planning, and policy development. As modeling methodologies continue to evolve alongside laboratory techniques and computational power, these approaches will play an increasingly central role in global efforts to understand and control complex zoonotic disease systems.
Intervention analysis using mathematical models has become a cornerstone in evaluating the efficacy of control measures for zoonotic intestinal protozoa. These pathogens, including Cryptosporidium spp., Giardia duodenalis, Enterocytozoon bieneusi, and Blastocystis sp., represent a significant global health burden, with transmission occurring between humans, livestock, and wildlife reservoirs [36]. Modeling provides a powerful framework to simulate disease dynamics under various intervention scenarios, enabling researchers and public health officials to optimize resource allocation before implementing costly field interventions. The complex life cycles and multiple transmission routes of these protozoa necessitate sophisticated modeling approaches that can capture the nuances of zoonotic transmission pathways.
The paradigm of One Health, which recognizes the interconnectedness of human, animal, and environmental health, is particularly relevant to controlling zoonotic intestinal protozoa [60]. Mathematical models serve as the quantitative backbone of the One Health approach, allowing for the integration of multi-host transmission dynamics and the evaluation of cross-species control measures. As these protozoa continue to cause significant morbidity in both human and animal populations, with some species like Cryptosporidium being a leading cause of childhood diarrhea globally, the importance of evidence-based intervention strategies cannot be overstated [36]. This technical guide explores the core modeling methodologies, experimental protocols, and applications of intervention analysis within the context of zoonotic intestinal protozoa research.
Fundamental Modeling Approaches
Multi-Host Transmission Dynamics Models
Multi-host transmission dynamics models form the foundation for evaluating interventions against zoonotic intestinal protozoa. These models account for the complex interactions between different host species, which is crucial for accurately assessing control measures where pathogens circulate between humans and animal reservoirs. The core structure of these models typically involves a system of differential equations that track the flow of infection through compartmentalized host populations.
A novel multi-host transmission model for Ancylostoma ceylanicum hookworm demonstrates this approach, though the methodology is equally applicable to zoonotic intestinal protozoa [60]. The rate of change in the mean number of parasites in host i at time t, Wi(t), can be described by the equation:
dWi(t)/dt = ∑j(μW + μj)Rei,jWj(t) - (μW + μi)Wi(t)
Where μW is the per capita mortality rate of adult parasites, μi and μj are the mortality rates of hosts i and j respectively, and Rei,j are components of the effective reproduction number describing transmission in host i from host j [60]. The model components Rei,j are given by:
Rei,j = R0i,jΩ(Wj(t),kj(t),b)Φ(Wj(t),kj(t))
Where Ω(·) and Φ(·) denote density-dependent fecundity and mating probability functions respectively, which are essential for capturing the population biology of protozoan parasites [60]. This framework allows researchers to simulate how interventions such as mass drug administration (MDA) might disrupt transmission cycles across multiple host species.
Wastewater-Based Epidemiology (WBE) Modeling
Wastewater-based epidemiology has emerged as a promising complementary approach for infectious disease surveillance, including for intestinal protozoa [61]. WBE modeling utilizes wastewater surveillance data to estimate and predict various epidemiological metrics, providing real-time insights into disease dynamics across spatial scales. Diverse modeling approaches have been employed with WBE data, including:
- Compartmental models that track the flow of individuals through susceptible, exposed, infectious, and recovered compartments
- Regression-based models that establish statistical relationships between pathogen concentrations in wastewater and clinical case numbers
- Machine learning (ML) and deep learning (DL) techniques that offer flexible pattern recognition and prediction capabilities
- Statistical approaches that incorporate complex biological processes such as viral shedding dynamics and decay patterns
These models face several critical challenges for robust implementation, including model-related limitations, inherent issues with wastewater data quality, temporal alignment between wastewater signals and clinical cases, data preprocessing strategies, and evaluation of model performance and interpretability [61]. Enhancing the transferability of these models across diverse epidemiological and geographical contexts remains a key concern for widespread application.
Spatial and Statistical Transmission Models
Spatial modeling techniques have evolved from simple statistical descriptions to complex spatiotemporal predictions for infectious diseases [62]. Modern spatial transmission models integrate multi-source data through Geographic Information Systems (GIS) to simulate pathogen transmission dynamics. These models include:
- Point pattern analysis identifying clustering of cases
- Areal data analysis for aggregated regional data
- Space-time scan statistics detecting spatiotemporal clusters
- Spatial regression models assessing environmental and socioeconomic risk factors
These approaches show unique advantages in evaluating the effects of different prevention and control measures, providing scientific basis for public health decision-making [62]. The main challenge lies in translating results from fine spatiotemporal scale analyses into actionable recommendations for practical control actions.
Table 1: Comparison of Fundamental Modeling Approaches for Zoonotic Intestinal Protozoa
| Modeling Approach | Key Features | Data Requirements | Primary Applications |
|---|---|---|---|
| Multi-Host Transmission Dynamics | Captures cross-species transmission; Incorporates host demographics; Models density-dependent parasite processes | Host population sizes; Contact rates between species; Parasite biological parameters | Evaluating targeted interventions; Assessing reservoir control; Predicting elimination thresholds |
| Wastewater-Based Epidemiology (WBE) | Population-level surveillance; Early outbreak detection; Real-time monitoring | Wastewater samples; Pathogen concentration data; Shedding rates | Community-level monitoring; Assessing intervention effectiveness in populations; Early warning systems |
| Spatial Transmission Models | Identifies geographic hotspots; Incorporates environmental factors; Visualizes disease spread | Geocoded case data; Environmental data; Land use information | Targeted spatial interventions; Resource allocation optimization; Understanding environmental determinants |
Quantitative Data Analysis for Intervention Assessment
Prevalence and Molecular Characterization Data
Robust intervention analysis requires comprehensive baseline data on pathogen prevalence and genetic diversity. A 2024 study in Heilongjiang Province, China, provides exemplary data on four common zoonotic intestinal protozoa in domestic small ruminants, which are key reservoirs for human infection [36]. The overall infection rates from 1011 fecal samples demonstrate the coexistence of multiple protozoa in the same reservoirs, with significant implications for intervention design:
- Cryptosporidium spp.: 4.15% (42/1011)
- Giardia duodenalis: 2.67% (27/1011)
- Enterocytozoon bieneusi: 12.15% (127/1011)
- Blastocystis sp.: 3.56% (36/1011)
- Mixed infections with two or more protozoa: 2.97% (30/1011)
Geographic location was identified as a significant risk factor for the prevalence of Cryptosporidium spp., E. bieneusi, and Blastocystis sp., highlighting the importance of spatially-targeted interventions [36]. The molecular characterization revealed substantial genetic diversity, with four Cryptosporidium genotypes (C. xiaoi, C. ubiquitum, C. bovis, C. andersoni), seven E. bieneusi genotypes (BEB6, COS-I, CHS8, CHS7, CHG1, CHG3, J), two G. duodenalis assemblages (assemblage E, assemblage A), and six Blastocystis subtypes (ST10, ST14, ST26, ST5, ST15, ST30) identified [36]. This genetic diversity has profound implications for intervention efficacy, as different genotypes may vary in zoonotic potential, drug susceptibility, and environmental persistence.
Intervention Efficacy Metrics and Statistical Analysis
Quantitative assessment of intervention efficacy relies on specific statistical measures and analytical techniques. Cross-tabulation analysis enables researchers to examine relationships between categorical variables, such as intervention type and infection outcomes [63]. For example, data can be arranged in tabular format to display infection frequency across different intervention groups and demographic variables, allowing for the identification of intervention effectiveness across population subgroups.
MaxDiff analysis, while originally developed for market research, can be adapted to prioritize interventions based on expert preference or cost-effectiveness criteria [63]. This technique involves presenting respondents with a series of choices between intervention options and analyzing the patterns of selection to create ranked preferences. Gap analysis provides a framework for comparing actual intervention performance against established goals or benchmarks [63]. This assessment reveals performance gaps that help identify areas for improvement in intervention strategies.
Table 2: Key Quantitative Metrics for Intervention Analysis of Zoonotic Intestinal Protozoa
| Metric Category | Specific Metrics | Calculation Method | Interpretation in Intervention Context |
|---|---|---|---|
| Infection Status Measures | Prevalence, Incidence, Force of Infection | Number infected/Total population; New infections/Person-time; Mathematical derivation from model | Reductions indicate intervention success; Changes reflect intervention effect on transmission dynamics |
| Molecular Epidemiology Metrics | Genotype Diversity, Zoonotic Index, Assemblage Distribution | Proportion of different genotypes; Ratio of zoonotic to host-adapted strains; Distribution of genetic assemblages | Measures intervention impact on zoonotic transmission; Identifies targets for specific interventions |
| Intervention Coverage & Adherence | Coverage Rate, Adherence Level, Compliance Rate | Number treated/Eligible population; Proportion completing full regimen; Self-reported or measured compliance | Determines real-world intervention implementation; Affects overall intervention effectiveness |
| Statistical Analysis Measures | Risk Ratio, Odds Ratio, Hazard Ratio | [Risk exposed/Risk unexposed]; [Odds exposed/Odds unexposed]; [Hazard exposed/Hazard unexposed] | Quantifies intervention effect size; Measures association between intervention and outcome |
Experimental Protocols and Methodologies
Field Surveillance and Sample Collection Protocols
Robust field surveillance forms the foundation for parameterizing and validating intervention models. The protocol implemented in Heilongjiang Province, China, provides a comprehensive methodology for studying zoonotic intestinal protozoa in animal reservoirs [36]. The sample collection strategy should ensure comprehensive geographic coverage of the study region. For the Heilongjiang study, fecal samples were collected from 845 sheep and 166 goats across all 13 administrative regions of the province between May 2023 and July 2024 [36].
Samples should be stratified by sex and age to reflect the proportionate distribution of livestock populations. The Heilongjiang study included 686 females and 325 males, with age groups including 266 samples from animals ≤1 year and 745 samples from animals >1 year [36]. All fecal samples should be obtained directly by rectal collection, immediately placed into sterile labeled tubes, kept refrigerated at low temperature during transport, and then stored at -20°C until processing to preserve pathogen DNA integrity.
Molecular Detection and Genotyping Protocols
Molecular characterization of pathogens is essential for understanding transmission dynamics and assessing targeted interventions. The nested PCR protocols provide the necessary sensitivity for detecting low levels of protozoan infections in field samples [36]. For Cryptosporidium spp. detection, nested PCR amplification of the SSU rRNA gene should be performed using established primers and conditions [36]. For G. duodenalis, assemblage determination requires nested PCR of the bg gene following published methods [36].
E. bieneusi genotyping employs nested PCR targeting the ITS gene locus to identify the diverse genotypes with varying zoonotic potential [36]. Blastocystis sp. subtyping uses PCR amplification of the SSU rRNA gene followed by sequencing to determine subtypes [36]. All PCR reactions should contain template DNA, buffer, dNTPs, primers, and Taq polymerase, with cycling conditions including initial denaturation at 92°C for 5 minutes, followed by 35 cycles of denaturation at 92°C for 45 seconds, annealing at specific temperatures for each protocol for 45 seconds, extension at 72°C for 1 minute, and final extension at 72°C for 10 minutes [36]. Secondary PCR products should be sequenced bidirectionally to ensure accuracy.
Model Parameterization and Validation Protocols
Accurate parameter estimation is crucial for creating reliable models to evaluate interventions. The multi-host model for A. ceylanicum provides a methodological framework that can be adapted for intestinal protozoa [60]. Parameters should be sampled using Latin hypercube approaches from appropriate distributions to account for uncertainty and variability [60]. For the A. ceylanicum model, 10,000 parameter sets were sampled from independent uniform distributions using this method [60].
Model validation should assess the ability of the model to reproduce observed epidemiological patterns. The infection prevalence should be derived from assuming a negative binomial distribution of parasites among hosts, calculated as Pi(t) = 1 - (1 + Wi(t)/ki(t))^{-ki(t)} [60]. Model performance can be evaluated by comparing predicted prevalence and intensity of infection with longitudinal field data, with sensitivity analyses conducted to identify parameters with the greatest influence on model outcomes.
Applications in Zoonotic Protozoa Control
One Health Intervention Strategies
The One Health approach recognizes that effective control of zoonotic intestinal protozoa requires integrated interventions targeting human, animal, and environmental compartments. Modeling studies demonstrate that One Health interventions—targeting both animal reservoirs and human populations—could suppress prevalence in humans to ≤1% by the end of 2030, even with only modest coverage (25-50%) of the animal reservoir [60]. With increasing coverage, One Health interventions may even interrupt transmission of zoonotic pathogens.
For the zoonotic hookworm A. ceylanicum, modeling shows that expanding mass drug administration to domestic dog populations in settings where this parasite is endemic could significantly enhance elimination prospects compared to human-only strategies [60]. Similar approaches are relevant for zoonotic intestinal protozoa like Giardia and Cryptosporidium that have animal reservoirs. The World Health Organization's 2030 road map acknowledges the importance of One Health for neglected tropical diseases, highlighting the growing recognition of this approach in global policy [60].
Drug-Based Interventions and Repurposing Strategies
Pharmaceutical interventions remain a cornerstone of protozoan disease control, but face challenges including drug resistance, limited efficacy, and poor safety profiles [64]. Drug repurposing offers a promising strategy to accelerate the development of new therapeutic options for intestinal protozoa. This approach identifies new applications for existing drugs already approved for other conditions, potentially reducing development costs and timelines [65].
Recent advances in drug repurposing for various human protozoan diseases explore the cross-applicability of repurposed drugs across diverse diseases and harness common chemical motifs to provide new insights into drug design [65]. Antimalarial drugs represent particularly exciting candidates that show activities against various other protozoan ailments [65]. Natural products and combinatorial therapies also offer alternative solutions to address the limited arsenal of effective drugs against intestinal protozoa.
Spatial Targeting of Interventions
Spatial epidemiology enables precision targeting of interventions to geographic areas with the highest disease burden or transmission potential. Spatial scan statistics can identify significant clustering of cases, allowing for efficient allocation of control resources [62]. Research has demonstrated spatial co-distribution of multiple pathogens in some areas, often significantly associated with healthcare accessibility, demographic, and climatic factors [62].
This approach was exemplified in Zimbabwe, where spatial scan statistics revealed no spatial overlap in the primary clusters of HIV/AIDS and malaria, but spatial overlap of secondary clusters was found in five districts, enabling integrated screening and intervention measures for both diseases in the overlapping areas [62]. Similar spatial targeting could be applied for intestinal protozoa, particularly in areas where multiple zoonotic pathogens coexist and share common risk factors or transmission pathways.
Table 3: Research Reagent Solutions for Zoonotic Protozoa Intervention Studies
| Reagent Category | Specific Products | Application in Intervention Research | Technical Considerations |
|---|---|---|---|
| DNA Extraction Kits | Solarbio Stool Genomic DNA Extraction Kit | Efficient DNA extraction from fecal samples for molecular characterization | Maintain cold chain during transport; Assess DNA quality by spectrophotometry and gel electrophoresis |
| PCR Reagents | Taq polymerase, dNTP Mixture, Specific Primers | Amplification of pathogen-specific gene targets for detection and genotyping | Optimize annealing temperatures for different protozoa; Include appropriate controls |
| Spatial Analysis Tools | Geographic Information Systems (GIS), Remote Sensing Data, Spatial Statistics Software | Identifying disease hotspots; Modeling environmental risk factors; Optimizing intervention targeting | Address modifiable areal unit problem; Consider spatial autocorrelation in analyses |
| Model Parameterization Data | Host population sizes, Contact rates, Pathogen biological parameters | Estimating key parameters for transmission models from empirical data | Use Latin hypercube sampling for uncertainty analysis; Validate with independent data sets |
Emerging Technologies and Future Directions
Artificial Intelligence and Advanced Analytics
Artificial intelligence (AI) stands poised to mark a significant paradigm shift in intervention methodology for zoonotic protozoa [65]. AI-enabled approaches can enhance disease surveillance, pattern recognition, and prediction of outbreak risks, ultimately supporting more effective and timely interventions [66]. Machine learning algorithms can integrate complex, multi-source data to identify subtle patterns associated with transmission hotspots or intervention success.
Big data analytics combined with AI offers powerful capabilities for processing the vast datasets generated by modern surveillance systems [66]. These technologies enable real-time monitoring and prediction of outbreak risks, allowing for proactive rather than reactive interventions. The integration of AI with spatial technologies creates Geographic Artificial Intelligence (GeoAI), which can refine exposure assessment and dynamic transmission modeling for zoonotic pathogens [62].
Integrated Surveillance and Early Warning Systems
Modern technologies significantly enhance capabilities for zoonotic disease surveillance and outbreak response [66]. Advanced tools including big data analytics, the Internet of Things, geographic information systems, remote sensing, molecular diagnostics, point-of-care testing, and digital contact tracing enable comprehensive surveillance systems [66]. When integrated through collaborative partnerships, these strategies can dramatically improve the speed and effectiveness of zoonotic disease control.
Early warning systems integrate GIS, spatial statistics, and multi-source real-time data to achieve dynamic monitoring of spatiotemporal disease transmission patterns, risk prediction, and targeted intervention [62]. These systems transform abstract case data into intuitive spatial risk maps, providing powerful tools for public health decision-makers. During outbreaks, such systems can utilize methods like spatial scan statistics for rapid anomaly detection and source tracing, as demonstrated during a Salmonella outbreak in New York City where the system successfully located the source restaurant before the official announcement [62].
Challenges and Implementation Barriers
Despite promising technological advances, several challenges persist in implementing effective interventions for zoonotic intestinal protozoa, particularly in resource-limited settings [66]. Infrastructure limitations, costs, data integration complexities, training requirements, and ethical implementation concerns present significant barriers to adoption [66]. Furthermore, enhancing the transferability of models across diverse epidemiological and geographical contexts remains a key concern for robust and practical implementation [61].
The translation of spatial epidemiology into policy confronts several methodological challenges, including geocoding errors that can lead to exposure misclassification, requiring standardized protocols to improve data reliability [62]. There is also a need to strengthen mechanisms for translating innovative technologies into practical prevention and control capabilities to bridge the gap between theoretical research and implementation [62]. With strategic planning and coordinated efforts, modern technologies and solutions offer immense potential to bolster surveillance and outbreak responses against emerging zoonotic disease threats worldwide [66].
The Spatiotemporal Epidemiologic Modeler (STEM) is an open-source software project supported by the Eclipse Foundation and used by a global community of researchers and public health officials working to track and control outbreaks of infectious disease in human and animal populations [67]. Unlike a specific disease model, STEM is a flexible, modular framework supporting exchange and integration of community models, reusable plug-in components, and denominator data, available to researchers worldwide at www.eclipse.org/stem [67] [68]. This framework runs on Eclipse Equinox, an industry standard framework where every model component is an independent building block, or "plug-in," available for reuse in the graphic user interface through drag and drop [67]. The capabilities that STEM and the Eclipse framework provide are illustrated by the range of work done by members of the global STEM community, including studies of seasonal influenza variations in Israel, evaluation of social distancing measures for H1N1 in Mexico City, measles outbreak analysis in London, and insights into H7N9 avian influenza transmission in China [67].
For researchers investigating the zoonotic transmission pathways of intestinal protozoa such as Cryptosporidium spp., Giardia duodenalis, Enterocytozoon bieneusi, and Blastocystis sp., tools like STEM offer powerful capabilities for modeling complex transmission dynamics between animal reservoirs and human populations [36]. These common zoonotic intestinal protozoa cause frequent foodborne and waterborne outbreaks worldwide and represent significant challenges for both veterinary and public health [36]. Molecular epidemiological studies have revealed complex transmission patterns; for instance, a 2025 study in Heilongjiang Province, China, identified multiple genotypes and subtypes in small ruminants with varying zoonotic potential [36]. Open-source modeling frameworks provide the necessary flexibility to incorporate such molecular data and model the spatiotemporal dynamics of these pathogens across species boundaries.
Core Architecture and Modeling Capabilities of STEM
Fundamental Compartmental Models
STEM implements many standard epidemiological compartment models as differential equations, providing researchers with foundational building blocks for constructing disease transmission simulations [69]. These include:
- SI and SIS Models: Simple two-state models useful for describing microparasitic infections where individuals never acquire long-lasting immunity, such as certain rhinoviruses and coronaviruses [69].
- SIR and SIRS Models: Three-state models that include a Recovered (R) compartment for infections conferring immunity, with SIRS accounting for waning immunity over time as seen with Orthomyxoviridae viruses causing seasonal flu [69].
- SEIR and SEIRS Models: Models incorporating an Exposed (E) compartment to represent incubation periods between pathogen exposure and clinical symptoms development, important for diseases like smallpox with 7-14 day incubation periods [69].
The disease models in STEM are implementations of these compartment models expressed as differential equations with parameters similar to constants in chemical rate equations [69]. Users can modify these basic parameters with a text editor to tune models for specific diseases, while more advanced mathematical models can be added by those with Java programming knowledge [69].
Advanced Modeling Features
Beyond basic compartmental models, STEM supports several advanced features critical for modeling complex disease transmission dynamics:
- Multi-population Disease Models: STEM provides the ability to model transmission of the same disease between multiple demographic groups with varying transmission characteristics [69].
- Nonlinear Models: Implementation of nonlinear versions of basic compartment models where the bilinear transmission kinetics term (βSI) is replaced with models of nonlinear transmission to account for dose response, saturation, or "swamping" of the immune system [69].
- Seasonal Transmission Models: STEM includes advanced models with seasonally modulated transmission coefficients using periodic forcing functions, Gaussian excitations, or interpolation of sampling points to represent climatic or behavioral variations [69].
- Vector-Borne Disease Models: Specialized models for diseases like malaria and dengue that explicitly incorporate mosquito vector populations and transmission dynamics [69].
These technical capabilities make STEM particularly suited for modeling the complex transmission pathways of intestinal protozoa, which may involve multiple host species, environmental persistence, seasonal variations, and both direct and indirect transmission routes.
Application to Zoonotic Intestinal Protozoa Research
Epidemiological Context and Data Requirements
Research on zoonotic intestinal protozoa has revealed complex transmission dynamics that benefit greatly from sophisticated modeling approaches. Recent studies demonstrate the widespread distribution of these pathogens in animal reservoirs, with significant implications for human health. A 2025 study in Heilongjiang Province, China, investigated 1,011 fecal samples from domestic small ruminants across 13 regions and found substantial infection rates [36]:
Table 1: Prevalence of Zoonotic Intestinal Protozoa in Domestic Small Ruminants (Heilongjiang Province, China)
| Pathogen | Overall Infection Rate | Dominant Genotypes/Subtypes Identified | Zoonotic Potential |
|---|---|---|---|
| Cryptosporidium spp. | 4.15% (42/1011) | C. xiaoi, C. ubiquitum, C. bovis, C. andersoni | High (C. ubiquitum) |
| G. duodenalis | 2.67% (27/1011) | Assemblage E, Assemblage A | Moderate (Assemblage A) |
| E. bieneusi | 12.15% (127/1011) | BEB6, COS-I, CHS8, CHS7, CHG1, CHG3, J | Varies by genotype |
| Blastocystis sp. | 3.56% (36/1011) | ST10, ST14, ST26, ST5, ST15, ST30 | Varies by subtype |
The study identified geographic location as a significant risk factor for the prevalence of Cryptosporidium spp., E. bieneusi, and Blastocystis sp. in domestic small ruminants, highlighting the importance of spatial modeling capabilities like those in STEM [36]. Similarly, research from Kazakhstan demonstrated age-related dynamics of cryptosporidiosis in calves, with prevalence of 49.2% in the 1-30 day age group, dropping dramatically in older animals [38]. These age-structured infection patterns and geographic variations are ideally suited for modeling with STEM's spatiotemporal capabilities.
Modeling Transmission Pathways
Zoonotic intestinal protozoa exhibit complex transmission pathways that can be effectively modeled using STEM's flexible framework:
- Direct Fecal-Oral Transmission: Both animal-to-animal and animal-to-human transmission through direct contact with infected hosts or their feces [36].
- Environmental Transmission: Indirect transmission through contamination of water sources, soil, or food with infectious cysts or oocysts that can persist in the environment [38].
- Vector-Borne Considerations: While not insect vectors, mechanical transmission via flies, rodents, or other animals that transport infectious stages between locations.
- Foodborne Transmission: Transmission through consumption of contaminated meat, produce, or dairy products, as highlighted by the economic impact in livestock operations [38].
The modular design of STEM allows researchers to incorporate all these pathways simultaneously, with parameters informed by field studies such as the Heilongjiang research which found mixed infections with two or more protozoa in 2.97% of samples [36].
Figure 1: Zoonotic Transmission Pathways of Intestinal Protozoa
Experimental Protocols and Methodologies
Field Sampling and Laboratory Diagnostics
Robust parameterization of STEM models for intestinal protozoa requires high-quality surveillance data collected through standardized protocols:
Sample Collection Protocol (adapted from Heilongjiang Province study [36]):
- Collect fecal samples directly by rectal collection from target animal species
- Stratify sampling by sex and age to reflect proportionate distribution of livestock populations
- Immediately place samples into sterile tubes with proper labeling
- Maintain cold chain transportation at low temperature using car refrigerators
- Store samples at -20°C until DNA extraction can be performed
Molecular Detection Methods [36]:
- Extract genomic DNA using commercial stool DNA extraction kits
- Verify DNA quality and quantity by electrophoresis in 1.5% (w/v) agarose gel and spectrophotometer measurement
- Use nested PCR amplification for specific genetic targets:
- Sequence positive PCR products in both directions to confirm genotypes
Model Parameterization and Validation
Effective simulation of zoonotic transmission requires careful parameterization based on empirical data:
Table 2: Key Parameters for Modeling Zoonotic Intestinal Protozoa Transmission
| Parameter Category | Specific Parameters | Data Sources | Example Values |
|---|---|---|---|
| Host Factors | Age-specific susceptibility, Species-specific susceptibility, Immunity duration | Prevalence studies, Longitudinal cohorts | Calf susceptibility: 49.2% (1-30 days) [38] |
| Pathogen Factors | Genotype/subtype variability, Environmental persistence, Infectious dose | Molecular characterization, Environmental studies | Multiple genotypes with varying zoonotic potential [36] |
| Transmission Factors | Contact rates between species, Environmental contamination levels, Seasonal variations | Behavioral studies, Environmental sampling, Time-series analysis | Geographic variation in prevalence [36] |
| Control Measures | Hygiene intervention efficacy, Treatment effectiveness, Vaccination impact | Intervention trials, Clinical studies, Field evaluations | Reduced transmission through farm management [38] |
Figure 2: Integrated Research Workflow for Protozoa Modeling
The Scientist's Toolkit: Research Reagent Solutions
Successful implementation of STEM models for intestinal protozoa research requires both computational tools and laboratory reagents. The following table details essential materials and their functions:
Table 3: Essential Research Reagents and Computational Tools for Zoonotic Protozoa Studies
| Tool/Reagent | Specific Function | Application in Protozoa Research |
|---|---|---|
| Solarbio Stool DNA Kit | Extraction of high-quality genomic DNA from fecal samples | Standardized DNA extraction for PCR-based detection of all four protozoa [36] |
| Takara Ex Taq Polymerase | High-fidelity PCR amplification | Detection and genotyping of protozoa through nested PCR protocols [36] |
| Species-Specific Primers | Amplification of target genetic loci | Identification of species, assemblages, and subtypes through sequence analysis [36] |
| STEM Modeling Framework | Spatiotemporal epidemic simulation | Integration of field data to model transmission dynamics and test interventions [67] |
| Eclipse Equinox Platform | Modular software component management | Flexible integration of custom disease models and data sources [67] |
| Geospatial Data Tools | Management of spatial and demographic data | Incorporation of geographic risk factors and population movements [67] |
Implementation Guide for Researchers
Getting Started with STEM
Implementing STEM for zoonotic intestinal protozoa research involves several key steps:
Framework Installation: Download STEM from the official Eclipse Foundation website (www.eclipse.org/stem) and install the base framework with core components [67].
Data Integration: Incorporate relevant denominator data including:
- Animal population distributions from agricultural census data
- Human demographic data from available sources
- Environmental factors influencing protozoa survival and transmission
- Transportation networks relevant to animal and human movement
Model Selection: Choose appropriate compartmental models based on pathogen characteristics:
- SIR-style models for infections conferring immunity
- SEIR models for pathogens with significant incubation periods
- Multi-population models for transmission across species boundaries
- Vector-borne models for environmental transmission pathways
Parameter Estimation: Derive model parameters from empirical studies such as the Heilongjiang prevalence data [36] and Kazakhstan calf infection patterns [38].
Advanced Applications and Customization
For advanced research needs, STEM supports extensive customization:
Development of Custom Plugins: Researchers with Java programming knowledge can develop disease-specific plugins incorporating unique transmission characteristics of intestinal protozoa [69].
Integration with Molecular Data: Incorporate genotype-specific transmission parameters based on molecular epidemiological findings, such as the varying zoonotic potential of different Cryptosporidium species and G. duodenalis assemblages [36].
Scenario Analysis: Test intervention strategies such as:
- Targeted treatment of high-prevalence age groups in animal reservoirs
- Environmental management to reduce contamination
- Hygiene interventions at human-animal interfaces
- Vaccination strategies if available
The STEM framework's open-source nature facilitates collaboration and model sharing, allowing researchers to build upon existing components developed for other diseases, such as the salmonella model that tracked transmission "from farm to fork" in Germany [67]. This capability is particularly valuable for modeling the complex supply chain pathways relevant to foodborne transmission of intestinal protozoa.
The Spatiotemporal Epidemiologic Modeler (STEM) provides a powerful, flexible open-source framework for modeling the transmission dynamics of zoonotic intestinal protozoa. By integrating molecular epidemiological data, environmental factors, and host population dynamics, researchers can develop sophisticated models to understand and control these significant public health threats. The modular architecture of STEM supports customization for the unique characteristics of pathogens like Cryptosporidium spp., G. duodenalis, E. bieneusi, and Blastocystis sp., while the open-source approach promotes collaboration and building on previous work. As molecular epidemiological studies continue to reveal the complex transmission patterns of these pathogens, tools like STEM will be increasingly essential for translating field observations into effective public health interventions.
Navigating Challenges: Drug Resistance, Diagnostic Limitations, and Protocol Optimization
The control of infections caused by intestinal protozoa represents a significant and ongoing global health challenge, particularly within the context of zoonotic transmission pathways. For decades, the 5-nitroimidazole class of drugs, particularly metronidazole (MTZ), has served as the cornerstone of treatment for giardiasis, amebiasis, and trichomoniasis [1]. However, the increasing incidence of treatment-refractory cases threatens to undermine the clinical management of these parasitic diseases. This emerging problem is especially acute for Giardia duodenalis, a protozoan with established zoonotic transmission cycles that can involve humans, wildlife, and domestic animals [70]. The growing documentation of clinical failures necessitates a comprehensive examination of both the epidemiological patterns and underlying biological mechanisms of resistance. This technical review aims to synthesize current understanding of metronidazole-refractory protozoal infections, with particular emphasis on giardiasis within zoonotic research frameworks, and to provide detailed methodologies for investigating resistance mechanisms in laboratory settings.
Epidemiological Patterns of Treatment Failure
Geographic Distribution of Refractory Infections
Surveillance data from returning travelers has revealed striking geographic disparities in the prevalence of nitroimidazole-refractory giardiasis. Studies conducted in European clinical settings consistently identify South Asia, and particularly India, as a high-risk region for refractory infections [71] [9]. A comprehensive population-based study in Stockholm, Sweden, analyzing 4,285 giardiasis cases between 2008-2020, found that 12% (64/545) of infections acquired in India were nitroimidazole-refractory, compared to just 1.0% (38/3,740) from the rest of the world [9]. Even more alarming, the proportion of refractory cases acquired in India nearly doubled during the study period, rising from 8.5% in 2008-2014 to 17.2% in 2014-2020 [9]. This suggests an accelerating resistance problem in this region, potentially driven by high drug pressure and widespread use of 5-nitroimidazoles.
Table 1: Geographic Distribution of Nitroimidazole-Refractory Giardiasis Based on Swedish Surveillance Data (2008-2020)
| Region of Acquisition | Total Cases | Refractory Cases | Refractory Percentage |
|---|---|---|---|
| India | 545 | 64 | 12.0% |
| Nepal | 7 | 2 | 29.0% |
| Rest of Asia (excluding India) | 792 | 9 | 1.1% |
| Africa | 1,115 | 17 | 1.5% |
| Europe | 1,247 | 11 | 0.9% |
| Americas | 349 | 1 | 0.3% |
| Domestic (Sweden) | 881 | 5 | 0.6% |
Clinical Management and Risk Factors
Clinical studies have demonstrated that management of refractory giardiasis remains challenging, with no standardized treatment approach. A retrospective analysis of 339 returned travelers with giardiasis in Germany found that 93 of 308 patients (30%) initially treated with metronidazole experienced treatment failure [71]. Multivariable analysis identified travel to South Asia as the strongest predictor of metronidazole failure (Odds Ratio 8.73). For second-line therapy, combination regimens appeared more effective than monotherapy, though results were inconsistent across studies. Quinacrine has shown particular promise, with one study reporting clinical improvement in all 56 refractory patients treated with this drug [9]. These findings highlight the need for clinicians to consider geographic exposure history when selecting initial treatment regimens and to be prepared to implement alternative therapeutic strategies for returning travelers from high-risk regions.
Molecular Mechanisms of Metronidazole Resistance
Proposed Biochemical Pathways
Metronidazole functions as a prodrug that requires activation within the anaerobic metabolic pathways of susceptible parasites. In Giardia duodenalis, the primary mechanism of action involves nitroreductase-mediated activation of the drug's nitro group, leading to the formation of toxic nitro radicals that cause DNA damage and cell death [70]. Resistance development is associated with complex modifications to this activation pathway, including:
- Downregulation of pyruvate:ferredoxin oxidoreductase (PFOR), the key enzyme involved in transferring electrons to ferredoxin, which subsequently reduces metronidazole [70]
- Altered expression of nitroreductase genes, particularly those encoding oxygen-insensitive nitroreductases that facilitate metronidazole activation [70]
- Enhanced DNA repair mechanisms that counteract metronidazole-induced DNA damage [1]
- Potential efflux pump activity that may reduce intracellular drug accumulation, though this mechanism is less well-characterized in Giardia compared to bacterial systems [70]
The following diagram illustrates the key metabolic pathways involved in metronidazole activation and potential resistance mechanisms in Giardia:
Role of Extracellular Vesicles in Resistance Dissemination
Recent research has revealed that extracellular vesicles (EVs) may play a significant role in inter-parasite communication and potentially in the dissemination of drug resistance factors [72]. These membrane-bound nanoparticles, ranging from 30-150 nm in diameter, are secreted by protozoan parasites and contain diverse biomolecules including proteins, lipids, and RNA. Studies demonstrate that Giardia-derived EVs can disrupt tight junctions in intestinal epithelia and modulate host immune responses [72]. More importantly, EVs have been shown to transport drug-resistance proteins and potentially nucleic acids that may confer survival advantages to recipient parasite populations, though the exact mechanisms remain an active area of investigation [72]. The biogenesis of these vesicles in Giardia appears to occur through unique pathways involving peripheral vacuoles and ESCRT-associated proteins, as the parasite lacks a conventional Golgi complex and certain ESCRT components [72].
Experimental Approaches for Investigating Resistance
In Vitro Induction and Assessment of Resistance
Laboratory investigation of metronidazole resistance typically begins with the stepwise exposure of parasite cultures to increasing sublethal concentrations of the drug. The following protocol outlines a standardized approach for generating and characterizing resistant Giardia lines:
Table 2: Research Reagent Solutions for Metronidazole Resistance Studies
| Reagent/Resource | Specifications | Primary Function |
|---|---|---|
| Giardia duodenalis cultures | Reference strains (e.g., WB clone C6, Portland-1) and clinical isolates | In vitro model for resistance induction and mechanistic studies |
| Metronidazole stock solution | 10-100 mM in DMSO or culture medium, stored at -20°C | Selective pressure for resistance development |
| Anaerobic culture system | Anaerobic chambers or sealed bags with gas generation systems | Maintains microaerophilic conditions essential for Giardia growth and drug activation |
| TYI-S-33 medium | Trypticase-yeast extract-iron-serum medium, pH 7.0-7.2 | Standard axenic culture medium for Giardia trophozoites |
| Cell viability assays | MTT, alamarBlue, or propidium iodide staining | Quantification of drug efficacy and resistance levels |
| RNA/DNA extraction kits | High-quality nucleic acid isolation systems | Molecular analysis of resistance mechanisms |
Protocol: Stepwise Induction of Metronidazole Resistance in Giardia duodenalis
Culture Conditions: Maintain reference strains or clinical isolates of Giardia trophozoites in TYI-S-33 medium with adult bovine bile and antibiotics at 37°C under microaerophilic conditions [70].
Baseline Susceptibility Testing: Determine the IC50 (50% inhibitory concentration) of metronidazole using serial dilutions (typically 0.5-100 μM) and 48-72 hour exposure periods. Calculate viability using MTT assay or direct cell counting.
Resistance Induction: Initiate selection with metronidazole concentrations at 10-25% of the established IC50. Culture parasites under continuous drug pressure with weekly passages.
Gradual Dose Escalation: Increase metronidazole concentration by 10-25% with each passage once stable growth is observed at the current selective pressure.
Characterization of Resistant Lines: Assess stable resistant lines for:
- Cross-resistance to other nitroimidazoles (tinidazole, secnidazole) and alternative drug classes (albendazole, quinacrine)
- Growth kinetics compared to parent strains
- PFOR enzyme activity spectrophotometrically
- Gene expression changes via transcriptomic analysis or RT-qPCR of candidate genes (nitroreductases, ferredoxins, oxidoreductases)
The following workflow diagram outlines the key steps in generating and characterizing metronidazole-resistant Giardia lines:
Molecular Characterization Techniques
Comprehensive characterization of resistant lines employs multiple complementary approaches:
Transcriptomic Analysis: RNA sequencing of isogenic sensitive and resistant pairs identifies differentially expressed genes. Focus on pathways involving nitroreductase activity, oxidative stress response, and energy metabolism [70].
Protein Expression Profiling: Western blotting or quantitative proteomics to validate changes in candidate resistance markers, particularly PFOR, ferredoxin, and nitroreductase protein levels [70].
Functional Validation: Genetic manipulation using CRISPR/Cas9 or antisense approaches to modulate expression of candidate resistance genes, followed by reassessment of drug susceptibility [1].
Comparative Genomics: Whole-genome sequencing of sensitive and resistant isolates to identify potential single nucleotide polymorphisms or gene amplifications/deletions associated with resistance.
Future Directions and Therapeutic Alternatives
The growing challenge of metronidazole-refractory protozoal infections necessitates both improved diagnostic capabilities and novel therapeutic approaches. Current research priorities include:
Development of Rapid Resistance Diagnostics: Implementation of molecular assays to detect resistance markers in clinical isolates would enable personalized treatment approaches. Potential targets include polymorphisms in nitroreductase genes and expression signatures associated with resistant phenotypes [9].
Drug Repurposing and Combination Therapies: Quinacrine, an antimalarial agent, has demonstrated excellent efficacy (≈98% clinical cure rate) in refractory giardiasis [9]. Similarly, nitazoxanide and albendazole offer alternative mechanisms of action that can circumvent nitroimidazole resistance [1] [70]. Combination therapies utilizing drugs with different molecular targets may prevent the emergence of resistance while improving treatment efficacy.
Vaccine Development: While no effective vaccine currently exists for giardiasis, research continues to identify potential antigenic targets that could prevent infection and reduce reliance on chemotherapeutic interventions [1].
Zoonotic Transmission Considerations: Future control strategies must account for the complex zoonotic transmission cycles of parasites like Giardia, which involve multiple assemblages with varying host specificities and potentially different drug susceptibility profiles [70]. Integrated One Health approaches that address transmission at the human-animal-environment interface will be essential for sustainable management of these parasitic infections.
The escalating challenge of metronidazole-refractory protozoal infections represents a critical junction in parasitic disease management. Addressing this threat will require coordinated efforts across multiple disciplines, from basic mechanistic studies to clinical management guidelines and public health interventions tailored to high-risk regions.
Intestinal protozoan infections represent a significant global health burden, with particular importance in zoonotic disease research. Pathogens such as Cryptosporidium spp., Giardia duodenalis, Enterocytozoon bieneusi, and Blastocystis sp. are common zoonotic intestinal protozoa that cause frequent foodborne and waterborne outbreaks worldwide [36]. Despite their public health importance, accurate epidemiological data remain scarce in many regions, partly due to diagnostic limitations [36]. The detection and characterization of these etiological agents are fundamental starting points for understanding disease pathophysiology, optimizing patient care, and preventing future infections [73].
The diagnostic landscape for intestinal protozoa is dominated by traditional methods, primarily microscopy and increasingly, rapid antigen tests. However, these approaches present significant sensitivity challenges that can compromise research outcomes and public health interventions. Microscopy, while widely available and inexpensive, suffers from operator dependency and requires substantial expertise for accurate identification. Antigen tests, though convenient and rapid, demonstrate variable performance characteristics. For instance, systematic reviews of antigen-based lateral flow tests for other pathogens have revealed an overall sensitivity of approximately 75.0% (95% CI: 71.0–78.0) compared to molecular reference methods [74]. This sensitivity gap becomes particularly problematic in zoonosis research, where accurate detection in both human and animal reservoirs is essential for understanding transmission dynamics.
This technical guide examines the critical pitfalls associated with these diagnostic methods and presents advanced approaches to overcome their limitations within the context of zoonotic intestinal protozoa research. By addressing these challenges, researchers can generate more reliable data to inform control strategies and zoonotic risk assessments [36].
Quantifying the Sensitivity Gap: Comparative Performance Data
The diagnostic sensitivity of microscopy and antigen tests varies considerably based on the target pathogen, specimen quality, and methodological execution. Understanding the magnitude of this sensitivity gap is essential for interpreting research findings and epidemiological data.
Table 1: Comparative Sensitivity of Diagnostic Methods for Intestinal Protozoa
| Diagnostic Method | Target Pathogens | Reported Sensitivity | Key Limitations |
|---|---|---|---|
| Microscopy (direct wet mount) | Multiple intestinal protozoa | Highly variable (often <60% for low burden infections) | Operator dependency, requires high parasite load [75] |
| Microscopy (with concentration techniques) | Multiple intestinal protozoa | Improved vs. direct mounts, but still suboptimal | Limited sensitivity for intermittent shedding [75] |
| Antigen Tests (lateral flow) | Giardia, Cryptosporidium, E. histolytica | ~75% overall for similar platforms [74] | Variable by manufacturer, dependent on viral load [74] |
| Molecular Methods (PCR) | All major intestinal protozoa | Gold standard (near 100% with proper protocols) | Requires specialized equipment and expertise [36] |
The performance of antigen tests is particularly influenced by pathogen load. Research on similar diagnostic platforms has demonstrated that sensitivity correlates strongly with target abundance, with significantly higher detection rates in samples with high organism burdens [74]. This relationship is particularly relevant for zoonotic protozoa research, where infection intensity may vary substantially between animal reservoirs and human cases.
Furthermore, sensitivity estimates for antigen tests show important population differences. One systematic review found that sensitivity was higher for symptomatic versus asymptomatic individuals (75.8% vs. 51.0%, P = 0.02) and higher within 7 days from symptom onset compared to populations with extended days of symptoms [74]. These factors must be considered when designing surveillance studies for zoonotic protozoa in both human and animal populations.
Fundamental Limitations of Conventional Diagnostic Methods
Microscopy Pitfalls
Traditional microscopy remains the most accessible diagnostic method for intestinal protozoa in many resource-limited settings where zoonotic parasites are endemic. However, this approach suffers from several inherent limitations that affect diagnostic accuracy and research reliability.
The sensitivity of microscopy is highly dependent on parasite burden and shedding patterns. Many intestinal protozoa exhibit intermittent shedding, making detection in single samples challenging. For example, in studies of calves, Cryptosporidium spp. infections were highly concentrated in the youngest animals, with prevalence of 49.2% detected in the 1-30-day group, but this dropped dramatically with older age [38]. Without repeated sampling, prevalence estimates from single time points may significantly underestimate true infection rates.
Operator expertise represents another critical variable. Morphological differentiation between pathogenic and non-pathogenic species requires substantial training and experience. For instance, distinguishing Entamoeba histolytica from non-pathogenic Entamoeba coli presents challenges that can lead to misclassification in field studies [12]. Even with concentration techniques such as formalin-ethyl acetate sedimentation and flotation methods, sensitivity remains suboptimal for low-intensity infections [76].
Antigen Test Limitations
Rapid antigen tests offer the advantage of convenience and rapid turnaround time but present their own set of limitations for research applications. The design and analytical sensitivity of these tests vary substantially between manufacturers, leading to inconsistent performance across studies [74].
The diagnostic sensitivity of antigen tests is heavily dependent on the timing of sample collection relative to infection course. This is particularly relevant for zoonotic protozoa with specific age-dependent prevalence patterns. Research in dairy calves has demonstrated that Cryptosporidium spp. infections peak in young animals (1-30 days), while Eimeria spp. prevalence increases with age [38]. Applying the same diagnostic approach across different age groups may therefore yield variable sensitivity.
Another significant limitation is the narrow target spectrum of most commercial antigen tests. While tests exist for major pathogens like Giardia and Cryptosporidium, they may not detect emerging or less common zoonotic protozoa. Molecular studies have revealed a surprising diversity of zoonotic genotypes in animal reservoirs, including seven E. bieneusi genotypes (BEB6, COS-I, CHS8, CHS7, CHG1, CHG3, J) and six Blastocystis subtypes (ST10, ST14, ST26, ST5, ST15, ST30) in small ruminants alone [36]. Most antigen tests cannot capture this diversity, potentially missing important zoonotic transmission events.
Enhanced Detection Protocols: Methodological Advancements
Optimized Microscopy Workflows
To overcome the limitations of basic microscopy, researchers can implement enhanced protocols that significantly improve detection rates. The following workflow illustrates a comprehensive approach to microscopic diagnosis:
This comprehensive approach maximizes sensitivity through multiple technical enhancements. The combination of direct examination with concentration techniques improves overall detection rates. Specific staining methods such as modified acid-fast staining for Cryptosporidium and fluorescent stains like auramine-O enhance the visibility of organisms that might be missed in standard preparations [75].
The critical step of multiple sample collection addresses the challenge of intermittent shedding. Research protocols should specify collection of at least three samples over 7-10 days to accurately determine infection status, as single samples may miss up to 50% of true infections [75].
Molecular Diagnostic Approaches
Molecular methods represent the gold standard for sensitive detection and characterization of zoonotic intestinal protozoa. The following protocol details a nested PCR approach for multi-pathogen detection:
Table 2: PCR-Based Detection Protocols for Zoonotic Intestinal Protozoa
| Target Pathogen | Genetic Target | PCR Type | Primary Application |
|---|---|---|---|
| Cryptosporidium spp. | SSU rRNA | Nested PCR | Species identification and genotyping [36] |
| Giardia duodenalis | bg gene | Nested PCR | Assemblage determination (zoonotic vs host-adapted) [36] |
| Enterocytozoon bieneusi | ITS region | Nested PCR | Genotype classification [36] |
| Blastocystis sp. | SSU rRNA | PCR | Subtype identification [36] |
The molecular workflow enables not only sensitive detection but also crucial genotyping information essential for zoonotic risk assessment. For example, research in Heilongjiang Province identified four Cryptosporidium genotypes (C. xiaoi, C. ubiquitum, C. bovis, C. andersoni), with C. ubiquitum being of particular zoonotic concern [36]. Similarly, G. duodenalis assemblage determination distinguishes between host-adapted (assemblage E) and potentially zoonotic (assemblage A) strains, providing critical data for understanding transmission dynamics [36].
Integrated Multi-Method Approach for Zoonosis Research
To overcome the limitations of individual diagnostic methods, a multi-platform approach provides the most comprehensive solution for zoonotic intestinal protozoa research. The integrated strategy combines the strengths of each method while mitigating their individual weaknesses.
Table 3: Strategic Integration of Diagnostic Methods in Zoonosis Research
| Method Combination | Application | Research Advantage |
|---|---|---|
| Microscopy + Antigen Test | Initial screening | Increased detection sensitivity over single method |
| Antigen Test + PCR | Confirmation of positives | Validation of results and genetic characterization |
| Microscopy + PCR | Comprehensive surveys | Maximum sensitivity with morphological correlation |
| Multi-locus PCR | Zoonotic tracking | High-resolution strain typing for transmission studies |
This integrated approach is particularly valuable in understanding the complex epidemiology of zoonotic protozoa. For instance, studies applying these methods have revealed that domestic small ruminants act as reservoirs for multiple zoonotic protozoans, with geographic location identified as a significant risk factor for prevalence of Cryptosporidium spp., E. bieneusi, and Blastocystis sp. [36]. Such findings would not be possible with single-method diagnostic approaches.
The strategic integration of methods also enables researchers to address specific epidemiological questions. For example, combining rapid antigen tests for initial screening with PCR confirmation allows for large-scale surveillance while maintaining diagnostic accuracy. This approach is particularly valuable in field studies with logistical constraints, where sample preservation and transport may challenge more sophisticated methodologies.
Essential Research Reagent Solutions
Successful detection and characterization of zoonotic intestinal protozoa requires specific research reagents and materials. The following table details essential components for establishing a comprehensive diagnostic and research pipeline:
Table 4: Essential Research Reagents for Intestinal Protozoa Studies
| Reagent Category | Specific Examples | Research Application | Technical Notes |
|---|---|---|---|
| DNA Extraction Kits | Solarbio Stool Genomic DNA Extraction Kit | Molecular studies | Effective inhibition removal for complex stool matrices [36] |
| PCR Reagents | Ex Taq polymerase, dNTP Mixture, 10× Ex Taq Buffer | Amplification of target genes | Provides high fidelity amplification [36] |
| Primer Sets | SSU rRNA (Cryptosporidium), bg (Giardia), ITS (E. bieneusi) | Species-specific detection | Validated primers available from established protocols [36] |
| Staining Reagents | Iodine, Modified Acid-Fast stains, Auramine-O | Microscopic identification | Enhances contrast for morphological assessment [76] [75] |
| Concentration Solutions | Formalin-ethyl acetate, Zinc sulfate | Sample processing | Increases diagnostic yield by concentrating parasites [76] |
| Antigen Tests | Commercial lateral flow assays (Giardia/Cryptosporidium) | Rapid screening | Useful for initial field screening; confirm positives with PCR [74] |
The selection of appropriate reagents should be guided by research objectives. For prevalence studies aiming to understand the full spectrum of zoonotic protozoa, a comprehensive approach incorporating multiple primer sets is essential. In one study of small ruminants, this approach identified not only the expected dominant genotypes (C. xiaoi, assemblage E, BEB6 and ST10) but also less common variants with potential zoonotic significance [36].
Proper storage and handling of reagents is critical for maintaining diagnostic accuracy. Molecular biology reagents, in particular, require strict temperature control and quality verification to prevent degradation that could compromise sensitivity. Implementation of positive and negative controls in each batch run is essential for verifying reagent performance and interpreting results accurately.
The diagnostic pitfalls associated with microscopy and antigen tests for intestinal protozoa present significant challenges for zoonosis research. However, through implementation of enhanced protocols, strategic method integration, and application of molecular tools, researchers can overcome these limitations to generate more accurate and meaningful data.
The comprehensive approach outlined in this technical guide enables not only sensitive detection but also crucial genotyping information essential for understanding zoonotic transmission pathways. As research continues to reveal the complex epidemiology of these pathogens, with factors such as geographic location significantly influencing prevalence [36], refined diagnostic approaches become increasingly important.
Future directions in the field will likely include the development of multiplexed platforms capable of simultaneous detection and genotyping of multiple zoonotic protozoa, as well as point-of-care molecular methods suitable for field studies in resource-limited settings where these infections have their greatest impact. By addressing current diagnostic limitations, the research community can generate the robust data needed to develop targeted control strategies and accurately assess zoonotic risks in diverse ecological settings.
The effective control of pathogens, particularly those with zoonotic potential, requires a sophisticated understanding of how infection risk varies across different host populations. Age-targeted control represents a strategic framework that moves beyond one-size-fits-all interventions by focusing resources on host demographics demonstrating the highest susceptibility, transmission potential, or clinical severity. This approach is particularly critical for managing intestinal protozoa, a group of parasites including Cryptosporidium spp., Giardia spp., and Eimeria spp., which impose significant burdens on both animal health and public health systems through zoonotic transmission pathways [21] [35].
The scientific premise for age-targeted control is rooted in the consistent observation that host age correlates profoundly with immunological status, physiological development, and behavioral patterns, all of which modulate infection outcomes. Within the context of intestinal protozoal infections, understanding these age-dynamic risks is not merely an academic exercise but a practical necessity for developing efficient, cost-effective intervention strategies that can reduce pathogen circulation in animal reservoirs and consequently minimize spillover events to human populations [21] [76] [35]. This technical guide synthesizes recent research findings to provide a framework for implementing age-targeted controls, with a specific focus on intestinal protozoa.
Epidemiological Foundations: Age as a Determinant of Infection Risk
Quantitative Evidence from Veterinary and Human Studies
Cross-sectional surveys and longitudinal studies in both animal and human populations provide compelling data on the age-structured prevalence of intestinal protozoa. A large-scale study conducted on 12 industrialized dairy farms in Kazakhstan, which examined 1,586 calves, offers a quintessential model of age-dependent infection patterns. The research revealed strikingly divergent age-prevalence profiles for three key protozoan pathogens, underscoring the necessity for pathogen-specific control strategies [21].
Table 1: Age-Specific Prevalence of Intestinal Protozoa in Calves (Kazakhstan Study)
| Parasite Species | Prevalence in 1-30 Days | Prevalence in 31-90 Days | Key Age-Associated Risk |
|---|---|---|---|
| Cryptosporidium spp. | 49.2% | Dramatic decrease (p<0.001) | Concentrated in the youngest calves |
| Eimeria spp. | 2.0% | Significant increase (OR: 27.3) | Risk increases markedly with age |
| Giardia spp. | 5.2% | Evenly distributed | No statistically significant variation |
The data demonstrates that Cryptosporidium spp. infections are highly concentrated in the youngest calves (1-30 days), with a prevalence of 49.2% in this group. The risk of infection drops dramatically with older age (p < 0.001). In stark contrast, Eimeria spp. prevalence was a mere 2.0% in the 1-month group but significantly increased with age. Calves aged 31-90 days had 27.3 times higher odds of infection (95% CI: 17.07–45.35, p < 0.001) compared to the youngest group [21]. Giardia spp. presented a different profile altogether, being more evenly distributed across age groups with no statistically significant variation [21].
Parallel evidence from human populations reinforces the validity of this approach. A cross-sectional survey among 380 under-five children in the Borena district of central Ethiopia found a 31.1% (95% CI: 26.6–36.1) prevalence of intestinal protozoan infections, with Giardia lamblia (18.7%) and Entamoeba histolytica/dispar (14.2%) being the dominant species [76]. This high prevalence in a specific pediatric age group highlights children as a key reservoir and target for intervention in communities.
The Zoonotic Imperative
The transmission of protozoan pathogens like Cryptosporidium and Giardia from animals to humans represents a persistent public health threat. Cattle are a recognized reservoir for species and genotypes infectious to humans, with the distribution and zoonotic potential of these pathogens varying across geographical regions [21] [35]. The high prevalence of these parasites in calf populations, as documented in Kazakhstan and other regions, underscores the role of intensive farming in maintaining transmission cycles that can spill over to human communities, particularly among immunocompromised individuals [21] [35]. Therefore, age-targeted control in animal reservoirs is not only a veterinary priority but a critical component of a One Health approach to disease prevention.
Mechanistic Insights: Biological Drivers of Age-Dependent Risk
The observed epidemiological patterns are driven by underlying biological and immunological mechanisms that vary with host development.
Immunological Naivety and Maturation: Neonatal calves and young children possess immature immune systems, making them highly susceptible to primary infection. The concentration of Cryptosporidium in calves under one month of age is a direct consequence of this naivety, as the adaptive immune response has not yet been primed to combat the parasite effectively [21].
Physiological and Behavioral Factors: In young animals, factors such as the development of rumen function and the cessation of colostrum-based passive immunity create windows of heightened vulnerability. Furthermore, age-specific behaviors, such as exploratory oral contact in young animals and children, increase exposure to fecally contaminated environments [76].
Pathogen-Specific Life History Strategies: Different protozoa have evolved to exploit distinct host niches. Cryptosporidium's strategy of targeting immunologically naive neonates ensures rapid propagation within a population. In contrast, Eimeria spp., which can cause more chronic infections and require time to build up sufficient environmental oocyst loads, manifest more strongly in older calves [21].
Experimental and Methodological Framework
Core Diagnostic and Field Methodologies
Implementing age-targeted control relies on robust surveillance data, which is generated through standardized diagnostic protocols. The following workflow outlines the key steps for establishing age-specific prevalence, as utilized in the cited studies.
Title: Workflow for Age-Targeted Parasite Surveillance
Detailed Experimental Protocols:
Stratified Random Sampling: The population is categorized into predefined age strata (e.g., 1–30 days, 31–90 days, 91–120 days, >120 days for calves). A systematic random sampling approach is then used to enroll a representative number of subjects from each stratum, ensuring statistical power to detect age-specific differences [21].
Field Sample Collection: Fresh fecal samples are individually collected from each study subject. Samples should be placed in sterile containers, labeled with unique identifiers, and transported under cool conditions to the laboratory to preserve parasite viability and morphology [21].
Laboratory Processing: Upon receipt, samples are processed using concentration techniques to increase detection sensitivity. The primary methods used in the referenced studies include:
- Flotation Techniques: Using solutions like ZnSO4 to separate parasite oocysts/cysts from fecal debris based on density [21].
- Richie's Modified Formol-Ethyl Acetate Concentration Technique: A standardized method for concentrating parasites for microscopic examination, particularly useful for human stool samples [76].
- Immunomagnetic Separation (IMS): Often used prior to immunofluorescence assay (IFA) to isolate specific parasites like Cryptosporidium oocysts and Giardia cysts from complex fecal samples, thereby improving diagnostic accuracy [77].
Microscopic Analysis: Concentrated samples are examined under a microscope. Saline and iodine wet mounts are used for initial examination, while specific stains (e.g., IFA with fluorescently-labeled antibodies) are employed for definitive identification and quantification of oocysts and cysts [21] [77] [76].
Data Analysis & Risk Calculation: Laboratory results are linked to host demographic data. Statistical analysis, including logistic regression, is performed to calculate age-specific prevalence and odds ratios (ORs) for infection in different age groups, using the youngest group as a reference [21].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Research Reagents for Protozoal Infection Studies
| Reagent / Material | Function in Experimental Protocol |
|---|---|
| ZnSO4 Flotation Solution | Parasite concentration via density gradient separation for microscopic analysis [21]. |
| Formol-Ethyl Acetate | Preserves stool samples and facilitates parasite concentration via sedimentation [76]. |
| Immunomagnetic Beads (IMS) | Antibody-coated magnetic beads for specific isolation of target oocysts/cysts from fecal samples [77]. |
| Fluorescently-Labelled Antibodies (IFA) | Specific staining of Cryptosporidium oocysts and Giardia cysts for detection and enumeration by microscopy [77]. |
| PCR & qPCR Reagents | Molecular detection and quantification of parasite DNA; allows for species and genotype identification [77]. |
| Cell Culture Lines (e.g., BGM) | Propagation and quantification of viable viruses in transmission studies or method validation [77]. |
Implementation Strategy: Designing Age-Targeted Interventions
The transition from epidemiological insight to effective intervention requires a structured approach. The following diagram outlines the decision-making pathway for developing an age-targeted control program, derived from the evidence presented.
Title: Age-Targeted Intervention Design Pathway
Intervention Components Based on Evidence:
For Pathogens Targeting Neonates (e.g., Cryptosporidium):
- Target: Calves aged 1-30 days.
- Actions: Enhance hygiene in calving pens; implement prophylactic treatment for dams and newborns; ensure adequate colostrum intake to bolster passive immunity [21].
For Pathogens with Increasing Age Risk (e.g., Eimeria):
- Target: Calves aged >31 days, particularly during weaning or movement stress.
- Actions: Apply strategic anticoccidial treatments in feed or water during high-risk periods; manage stocking density and pasture rotation to reduce environmental oocyst load [21].
For Generalized Pathogens (e.g., Giardia):
The Kazakhstan study concluded that under conditions of intensive dairy farming, "age-targeted parasite control strategies may be more effective than seasonal approaches for managing parasitic infections' control in calves" [21]. This principle can be extended to other host species and production systems.
The paradigm of age-targeted control offers a powerful and efficient framework for combating intestinal protozoa within their zoonotic transmission context. The compelling quantitative evidence demonstrates that a nuanced understanding of host-age risk dynamics allows for the strategic allocation of limited resources to the subpopulations where they will have the greatest impact on animal health, production, and the reduction of zoonotic risk.
Future research should focus on refining our understanding of the immunological basis of age-related susceptibility, developing rapid, field-deployable diagnostic tools for specific age groups, and evaluating the economic and health impacts of implemented age-targeted programs. By moving beyond blanket interventions and embracing a dynamic, evidence-based model of disease control, researchers, veterinarians, and public health professionals can significantly advance the goal of reducing the burden of parasitic diseases across the human-animal interface.
Intestinal protozoa, including Cryptosporidium spp., Giardia duodenalis, and Entamoeba histolytica, represent a significant global health burden with complex transmission dynamics at the human-animal-environment interface. The zoonotic potential of these pathogens necessitates a One Health approach to understanding their epidemiology, particularly as environmental factors undergo rapid change [78] [35]. Climate change exerts both direct and indirect influences on parasitic disease transmission through alterations in temperature, precipitation patterns, and humidity [79]. These environmental shifts interact critically with sanitation infrastructure, creating evolving challenges for disease control. This technical review examines the mechanisms through which climate and sanitation factors influence the transmission of intestinal protozoa and assesses intervention methodologies critical for researchers and drug development professionals working on zoonotic disease pathways.
The significance of this research area is underscored by the substantial disease burden imposed by intestinal protozoa. Cryptosporidiosis alone accounts for approximately 13 million disability-adjusted life years (DALYs) globally each year, while giardiasis reaches prevalence rates of 33% in developing countries [38]. Beyond their impact on human health, these parasites cause considerable economic losses in the livestock industry through reduced productivity and treatment costs [78] [36]. Understanding how environmental factors modulate transmission is thus essential for developing effective countermeasures against these pathogens.
Quantitative Epidemiology: Prevalence and Environmental Correlates
Table 1: Age-specific prevalence of intestinal protozoa in calves from intensive dairy farms in Kazakhstan (n=1,586)
| Parasite | 1-30 days (%) | 31-90 days (%) | 91-120 days (%) | >120 days (%) | Odds Ratio (31-90 days vs 1-30 days) |
|---|---|---|---|---|---|
| Cryptosporidium spp. | 49.2 | 22.1 | 15.3 | 8.7 | 0.29 (p<0.001) |
| Eimeria spp. | 2.0 | 38.5 | 42.1 | 40.3 | 27.3 (95% CI: 17.07-45.35, p<0.001) |
| Giardia spp. | 5.2 | 6.1 | 5.8 | 5.5 | 1.18 (NS) |
Table 2: Prevalence of zoonotic intestinal protozoa in small ruminants from Heilongjiang Province, China (n=1,011)
| Pathogen | Overall Prevalence (%) | Dominant Genotypes | Zoonotic Potential |
|---|---|---|---|
| Cryptosporidium spp. | 4.15 | C. xiaoi, C. ubiquitum | High (C. ubiquitum) |
| Giardia duodenalis | 2.67 | Assemblage E, Assemblage A | Moderate (Assemblage A) |
| Enterocytozoon bieneusi | 12.15 | BEB6, COS-I | High (Group 1 genotypes) |
| Blastocystis sp. | 3.56 | ST10, ST14 | Moderate (ST1-ST4 in humans) |
The age-dependent prevalence patterns observed in Table 1 demonstrate distinct transmission dynamics among protozoan species. Cryptosporidium infections are highly concentrated in neonatal calves, with risk decreasing dramatically with age, suggesting early exposure and development of protective immunity [38]. In contrast, Eimeria spp. prevalence increases significantly with age, indicating cumulative exposure or development of parasite communities over time. The relatively stable prevalence of Giardia across age groups suggests persistent transmission throughout calf development [38].
Molecular characterization of protozoal genotypes, as shown in Table 2, provides critical insights into zoonotic potential. The identification of Cryptosporidium ubiquitum and Giardia Assemblage A in small ruminants is particularly significant, as these genotypes are established human pathogens [36]. The high prevalence (12.15%) of Enterocytozoon bieneusi in small ruminants, with genotypes belonging to zoonotic Group 1, highlights an emerging public health concern that warrants increased surveillance [36].
Climate Change: Altering Protozoal Transmission Dynamics
Temperature and Humidity Effects on Parasite Survival
Climate change directly affects parasitic transmission through alterations in temperature and humidity regimes that influence parasite development, survival, and environmental persistence. Experimental and modeling studies demonstrate that temperature and humidity variables critically determine the viability and developmental rates of parasitic stages in the environment [80]. For soil-transmitted helminths with similar environmental requirements to many protozoa, intestinal species show peak infection risk at approximately 10°C (50°F), while gastric species respond more strongly to humidity, reaching peak risk at ≥80% relative humidity [80].
The lifecycle of protozoan parasites includes free-living stages as environmental-resistant oocysts, cysts, or eggs, followed by parasitic stages within host organisms. Climate variables exert selective pressure on both phases, with warming temperatures potentially accelerating embryonic development within eggs, thereby reducing incubation time [81]. However, this developmental advantage may be counterbalanced by reduced egg viability at temperature extremes, creating complex nonlinear responses to climate warming [81]. Increased rainfall patterns associated with climate change may facilitate protozoan transmission by expanding habitat availability for parasite-carrying organisms and preventing desiccation of environmental stages [79] [81].
Geographic Range Shifts and Seasonal Transmission Patterns
Climate change is projected to significantly alter the geographic distribution and seasonal transmission patterns of intestinal protozoa. Mechanistic models predict that infection hotspots will shift northward in Europe, facilitated by increasingly milder conditions in central and northern regions [80]. Scandinavian countries may experience increases in infection risk up to 100% for intestinal parasite species by the end of the century under high-emission scenarios [80].
Seasonal transmission windows are also expected to change, with intestinal helminths (sharing similar environmental transmission pathways with protozoa) potentially developing two infection peaks (spring and fall) rather than the historical single summer peak [80]. These seasonal shifts have important implications for the timing of public health interventions and prophylactic treatment regimens. Furthermore, the intensification of infection risk at mid-to-high latitudes may increase the probability of co-infections with multiple parasite species, potentially complicating clinical management and drug development strategies [80].
Figure 1: Climate change impacts on protozoan parasite transmission dynamics. Climate variables directly influence parasite development, survival, and geographic distribution, ultimately altering transmission risk patterns.
Historical evidence from paleoparasitology supports the responsiveness of parasitic diseases to climatic variations. Analysis of intestinal parasites in Central European lake dwellings from the 4th millennium BCE revealed marked shifts in parasitic spectra corresponding to climatic fluctuations [81]. During cool, wet periods with high lake levels, populations showed increased parasites associated with fishing practices, while warmer, drier periods with low lake levels favored parasites indicative of agropastoral activities [81]. These historical patterns demonstrate the long-standing influence of climate on human-parasite interactions and suggest potential pathways for future climate-driven epidemiological shifts.
Sanitation and Water Treatment: Intervention Evidence
Water Treatment Efficacy Against Protozoan Pathogens
Access to safe drinking water and adequate sanitation represents a cornerstone of intestinal protozoa control. Meta-analytical evidence demonstrates that water treatment interventions are associated with significantly lower odds of infection with key protozoan pathogens [82]. The protective effect varies by parasite species, with water treatment most effective against Blastocystis hominis (OR 0.52, 95% CI 0.34-0.78), followed by Entamoeba histolytica/dispar (OR 0.61, 95% CI 0.38-0.99), Giardia intestinalis (OR 0.63, 95% CI 0.50-0.80), and Cryptosporidium spp. (OR 0.83, 95% CI 0.70-0.98) [82].
The differential efficacy of water treatment against various protozoa reflects variations in environmental resistance and infectious dose. Cryptosporidium oocysts are particularly challenging to eliminate from water supplies due to their small size (4-6μm) and exceptional chlorine resistance, requiring advanced filtration or UV treatment for effective removal [78]. This technical challenge explains the more modest protective effect of water treatment against cryptosporidiosis compared to other intestinal protozoa.
Sanitation Infrastructure and Exposure Reduction
Sanitation facilities provide a critical barrier against fecal-oral transmission of intestinal protozoa. The availability and use of sanitation facilities is associated with significantly lower odds of infection with Entamoeba histolytica/dispar (OR 0.56, 95% CI 0.42-0.74) and Giardia intestinalis (OR 0.64, 95% CI 0.51-0.81), though not for Blastocystis hominis (OR 1.03, 95% CI 0.87-1.23) [82]. The lack of sanitation facilities significantly increases the odds of Entamoeba spp. infection (OR 1.18, 95% CI 1.05-1.32), with meta-regression analysis indicating that this association has strengthened over time (coefficient: 2.36, p<0.05) [83].
The protective effect of sanitation infrastructure varies by age group, with adults generally benefiting more than children from improved sanitation [83]. This differential effect may reflect variations in hygiene behavior, environmental exposure, or immune competence across age groups. Interestingly, the lack of safe drinking water was found to have a more pronounced effect on Entamoeba infection risk in adults (OR: 2.76) compared to children (OR: 0.57) and all-age populations (OR: 1.50) [83], suggesting age-specific vulnerabilities in waterborne transmission pathways.
Experimental Methodologies for Environmental Detection
Viability Assessment in Complex Matrices
Accurate detection of viable protozoan parasites in environmental samples is essential for risk assessment and intervention evaluation. Molecular methods that differentiate viable from non-vi pathogens are particularly valuable for public health decision-making. Systematic evaluation of viability methods for detecting Cryptosporidium parvum, Giardia enterica, and Toxoplasma gondii on spinach demonstrated that reverse transcription quantitative PCR (RT-qPCR) outperformed propidium monoazide PCR (PMA-PCR) and propidium iodide staining [84].
The RT-qPCR approach accurately detected 2-9 (oo)cysts per gram of spinach (in 10g processed) when only viable parasites were present [84]. When viable and inactivated parasites were mixed, mRNA concentrations correlated with increasing proportions of viable (oo)cysts, though low levels of false-positive mRNA signals were detectable in the presence of high amounts of inactivated protozoa [84]. This method provides a robust approach for screening leafy greens and other fresh produce for viable protozoan contamination, addressing a critical need in food safety monitoring.
Figure 2: Experimental workflow for detection and viability assessment of protozoan parasites in environmental samples. The multi-step process enables discrimination of viable pathogens and determination of zoonotic potential for risk assessment.
Molecular Characterization of Zoonotic Potential
Molecular tools are indispensable for tracking transmission pathways and assessing the zoonotic potential of environmental protozoal isolates. PCR-based methods targeting specific genetic loci enable species identification and genotyping, providing critical data for source attribution and risk assessment [36]. For Cryptosporidium, nested PCR amplification of the SSU rRNA gene followed by sequencing distinguishes between human-infectious species like C. parvum and C. ubiquitum and host-adapted species with limited zoonotic potential [36].
Similarly, Giardia duodenalis assemblage determination relies on nested PCR of the beta-giardin (bg) gene or other loci, differentiating assemblages with high zoonotic potential (Assemblage A and B) from those primarily adapted to animals (Assemblage E in livestock) [36]. These molecular techniques enable researchers to map transmission networks at the human-animal-environment interface and prioritize interventions based on the relative contribution of different transmission pathways.
Table 3: Research reagent solutions for protozoan detection and characterization
| Reagent/Kit | Application | Specification | Research Function |
|---|---|---|---|
| Solarbio Stool Genomic DNA Extraction Kit | DNA extraction from fecal samples | Commercial silica-column based kit | Efficient DNA purification from complex matrices |
| SSU rRNA gene primers (Cryptosporidium) | Species identification | Nested PCR targeting 18S rRNA | Differentiation of Cryptosporidium species and genotypes |
| bg gene primers (Giardia) | Assemblage determination | Nested PCR targeting beta-giardin gene | Distinguishes zoonotic assemblages A and B from host-adapted assemblages |
| ITS gene primers (E. bieneusi) | Genotyping | Nested PCR targeting internal transcribed spacer | Phylogenetic grouping to assess zoonotic potential |
| Propidium monoazide (PMA) | Viability assessment | DNA-intercalating dye excluding intact membranes | Differentiation viable vs. non-viable organisms in molecular assays |
| Reverse transcriptase qPCR | Viability detection | mRNA detection targeting heat shock protein genes | Gold standard for viability assessment in produce |
Discussion: Integrated Approaches for Future Control
The complex interactions between climate variables, sanitation infrastructure, and protozoan transmission necessitate integrated intervention strategies that address multiple transmission pathways simultaneously. The evidence presented in this review demonstrates that environmental factors significantly modulate the effectiveness of conventional control measures, suggesting that climate resilience must be incorporated into future intervention designs.
For researchers and drug development professionals, these environmental considerations have important implications. First, the geographic focus of intervention trials may need to shift as climate change alters parasite distribution patterns. Second, the seasonality of prophylactic treatment regimens may require adjustment as transmission windows change. Third, drug development programs should consider how climate-induced physiological changes in parasites might affect drug susceptibility and treatment efficacy.
Future research should prioritize longitudinal studies that capture climate variability, molecular epidemiological investigations to track transmission dynamics in changing environments, and intervention trials that evaluate the climate resilience of different control strategies. Additionally, improved integration between meteorological data and disease surveillance systems would enhance early warning capabilities for climate-sensitive protozoal diseases.
Climate and sanitation factors represent critical determinants in the transmission of intestinal protozoa with zoonotic potential. As climate change accelerates, understanding these environmental drivers becomes increasingly essential for effective disease control. The technical methodologies reviewed here—from viability testing in complex matrices to molecular characterization of zoonotic potential—provide powerful tools for researchers addressing these challenges. By integrating environmental perspectives with advanced laboratory techniques, the scientific community can develop more resilient interventions against these persistent pathogens, ultimately reducing their burden on human and animal health within the One Health framework.
Parasitic protozoan diseases represent a significant and persistent global health challenge, particularly in tropical and subtropical regions, affecting over a billion people worldwide and contributing to high rates of morbidity and mortality [85]. These infections, including malaria, trypanosomiasis, leishmaniasis, and various intestinal protozoal diseases, have been classified as neglected tropical diseases, receiving limited attention from pharmaceutical developers despite their devastating impact [85]. The intricate life cycles, genetic diversity, and immune evasion capabilities of protozoal pathogens have substantially complicated the discovery of effective therapeutic targets [85]. Currently available treatments are hampered by devastating side effects, emerging drug resistance, and limited efficacy across different disease stages, creating an urgent need for innovative antiprotozoal drugs [85] [86].
Drug repurposing (also known as drug repositioning, reprofiling, or re-tasking) has emerged as a promising strategy to accelerate the development of new therapies for protozoan diseases [85]. This approach leverages existing approved or investigational drugs outside their original therapeutic scope, capitalizing on their well-established pharmacokinetic and safety profiles to overcome traditional drug development bottlenecks [85]. The strategy offers conspicuous advantages over de novo drug discovery, including reduced development costs, shorter timelines, and potentially safer therapeutic options [85] [87]. Within the specific context of zoonotic intestinal protozoa, drug repurposing holds particular promise given the complex transmission dynamics between animals and humans, which complicate control efforts through conventional means [35] [88].
This review explores recent advances in drug repurposing for protozoal diseases, with particular emphasis on zoonotic intestinal pathogens, examining computational and experimental approaches, detailing specific repurposing candidates, and providing technical guidance for researchers engaged in this rapidly evolving field.
Epidemiology and Clinical Impact of Zoonotic Intestinal Protozoa
Zoonotic intestinal protozoal infections represent a significant disease burden worldwide, with transmission occurring through contaminated water, food, or direct contact with infected animals [35] [89]. The epidemiology of these diseases is closely tied to socioeconomic factors, with higher prevalence observed in regions with poor sanitation infrastructure and limited access to clean water [88]. Companion animals, including the approximately 77 million dogs and 93 million cats in households across the United States, serve as important reservoirs for several zoonotic protozoal parasites, creating significant public health challenges [35].
Table 1: Major Zoonotic Intestinal Protozoa: Epidemiology and Clinical Features
| Parasite | Global Incidence/Prevalence | Primary Reservoirs | Transmission Routes | Key Clinical Manifestations |
|---|---|---|---|---|
| Giardia lamblia | ~250 million cases annually [88] | Humans, livestock, wildlife [88] | Fecal-oral, contaminated water [89] | Persistent diarrhea, malabsorption, cramping, fatigue [88] |
| Entamoeba histolytica | ~100 million cases annually [88] | Humans [88] | Fecal-oral, contaminated food/water [89] | Diarrhea, abdominal pain, colitis, liver abscesses [89] [88] |
| Cryptosporidium parvum | 8.37 million DALYs globally [90] | Cattle, other livestock [88] | Fecal-oral, waterborne [89] | Mild-to-acute diarrhea, nausea, abdominal pain; severe in immunocompromised [88] |
| Toxoplasma gondii | ~10.3 million foodborne cases annually [90] | Cats (definitive host) [35] | Foodborne, fecal-oral, transplacental [35] | Generally mild in immunocompetent; severe neurologic disease in immunocompromised and congenital transmission [35] |
The clinical manifestations of intestinal protozoal infections range from self-limiting gastroenteritis to life-threatening conditions, with severity often dependent on the immune status of the host [85] [88]. Immunocompromised individuals, including those with HIV/AIDS, malnourished children, and patients undergoing immunosuppressive therapy, are particularly vulnerable to severe and chronic infections [35] [91]. The significant disease burden and limitations of current treatments underscore the critical need for improved therapeutic options through approaches such as drug repurposing.
Current Treatment Landscape and Limitations
The current pharmacopeia for protozoal diseases remains limited, with many available drugs dating back several decades and plagued by significant limitations [85] [90]. Treatment regimens for many parasitic diseases rely on a narrow arsenal of chemotherapeutic agents, often associated with severe side effects, emerging resistance, and limited efficacy across different disease stages [86] [90].
Table 2: Current Treatments for Major Protozoal Diseases and Their Limitations
| Disease | First-line Treatments | Major Limitations |
|---|---|---|
| Malaria | Artemisinin-based combination therapies (ACTs) [90] | Emerging resistance, particularly in Southeast Asia [85] [90] |
| Chagas Disease | Benznidazole, Nifurtimox [85] [86] | Limited efficacy in chronic phase, severe side effects, poor treatment adherence [86] |
| Leishmaniasis | Amphotericin B, miltefosine, antimonial compounds [85] [90] | Toxicity, resistance, requirement for parenteral administration [90] |
| Giardiasis | Metronidazole, tinidazole [85] [88] | Resistance emerging, side effects including metallic taste and gastrointestinal upset [88] |
| Cryptosporidiosis | Nitazoxanide [85] [90] | Limited efficacy in immunocompromised patients [90] |
| Toxoplasmosis | Pyrimethamine + sulfadiazine [35] [90] | Significant side effects including bone marrow suppression [35] |
The BENEFIT (BENznidazole Evaluation For Interrupting Trypanosomiasis) trial demonstrated that benznidazole, the frontline treatment for Chagas disease, does not halt disease progression in chronic patients, highlighting the critical therapeutic gap [86]. Similarly, the emergence of artemisinin resistance in Plasmodium falciparum poses a significant threat to global malaria control efforts [85]. These limitations underscore the urgent need for new treatment strategies, with drug repurposing offering a potentially accelerated path to address these challenges.
Methodological Approaches to Drug Repurposing
Computational and AI-Driven Strategies
Computational methods have revolutionized drug repurposing efforts by enabling systematic screening of large compound libraries against protozoal targets. These approaches include molecular docking, molecular dynamics simulations, and machine learning algorithms that predict novel drug-disease associations [85] [92] [87].
Figure 1: Computational workflow for drug repurposing against protozoal targets, integrating molecular docking, dynamics simulations, and AI-based prediction models.
Recent advances in artificial intelligence have significantly enhanced predictive capabilities for drug repurposing. The UKEDR (Unified Knowledge-Enhanced deep learning framework for Drug Repositioning) integrates knowledge graph embedding, pre-training strategies, and recommendation systems to address challenges such as cold start problems and limited attribute representations [93]. This framework demonstrates exceptional generalization capabilities, particularly in predicting clinical trial outcomes from approved drug data, improving the area under the receiver operating characteristic curve (AUC) by 39.3% over the next-best model [93].
Web-based platforms such as RepurposeDrugs provide interactive tools for exploring drug-indication associations, containing data on 4,314 compounds and 161 drug combinations linked to 1,756 indications [87]. These resources employ machine learning predictors to evaluate the approval potential of new drug-disease indications, offering valuable tools for researchers without specialized programming skills [87].
Experimental Approaches and Screening Methods
Biological experimental approaches complement computational methods by providing empirical validation of repurposing candidates. These include in vitro screening against protozoal cultures, transport assays to evaluate drug uptake, and membrane integrity assessments to understand mechanisms of action [86].
Protocol 1: Evaluation of Trypanocidal Activity [86]
- Parasite culture: Maintain epimastigotes of T. cruzi (CL Brener strain) at 28°C in brain-heart infusion-tryptose (BHT) medium supplemented with 10% fetal calf serum (FCS), antibiotics, and hemin.
- Drug exposure: Culture 1 × 10^7 epimastigotes/mL in exponential growth phase with increasing concentrations of candidate drugs in 96-well plates.
- Incubation: Maintain cultures at 28°C for 24 hours.
- Assessment: Determine parasite growth by counting in a Neubauer chamber.
- Data analysis: Calculate IC50 values using appropriate statistical methods.
Protocol 2: Metabolite Transport Assay [86]
- Parasite preparation: Centrifuge 1 × 10^7 epimastigotes at 8,000 × g for 30 seconds, wash once with PBS.
- Resuspension: Resuspend cells in 0.1 mL PBS.
- Reaction initiation: Add 0.1 mL of transport mix containing radiolabeled substrates (e.g., L-[3H] arginine, [3H] thymidine, or [3H] amino acid mix).
- Incubation: Incubate for 15 minutes at 28°C.
- Reaction termination: Add 1 mL of ice-cold PBS to stop reaction.
- Washing and analysis: Centrifuge, wash cells twice, resuspend pellet in water, and count radioactivity in liquid scintillation cocktail.
Promising Drug Repurposing Candidates
Antifungal Agents
The antifungal drug nystatin has demonstrated significant potential for repurposing against Trypanosoma cruzi, the causative agent of Chagas disease [86]. Nystatin inhibits arginine uptake in T. cruzi by targeting membrane transport processes, exhibiting potent trypanocidal effects against both epimastigotes (IC50 0.17 μM) and trypomastigotes (IC50 4.90 μM) [86]. Importantly, treatment of infected cells with nystatin decreased the release of trypomastigotes with better efficacy than benznidazole (IC50s 4.83 μM and 8.60 μM, respectively), suggesting it affects the progression of the intracellular life cycle [86]. Furthermore, researchers observed a synergistic effect when nystatin was combined with benznidazole, potentially enabling enhanced efficacy while improving treatment safety and adherence [86].
Amphotericin B, another polyene antifungal, is already used in the treatment of leishmaniasis, demonstrating the precedent for repurposing this drug class against protozoal infections [86]. The mechanism of action for nystatin in T. cruzi involves plasma membrane disruption, with membrane transport processes affected prior to evident membrane damage, likely due to initial interaction with the membrane before pore formation [86].
Cardiovascular Drugs and Natural Products
Computational screening approaches have identified additional promising repurposing candidates. A systematic computational drug repurposing approach screening 3,500 FDA-approved compounds against Plasmepsin II (PM2), a critical hemoglobin-degrading enzyme in malaria parasites, identified gamma-oryzanol and azilsartan as promising inhibitors [92]. These compounds demonstrated superior binding stability during molecular dynamics simulations, achieving MM-GBSA binding energies of -54.26 ± 2.92 kcal/mol and -45.32 ± 3.87 kcal/mol, respectively [92]. Both compounds maintained stable binding within the catalytic pocket throughout the simulation period, forming consistent interactions with critical residues including the catalytic dyad (Asp34, Asp214), flap region (Lys72-Phe85), and proline-rich loop (Ile290-Pro297) [92].
Natural products represent another rich source for antiprotozoal agents, either as direct therapeutic candidates or as inspiration for synthetic analogs [90]. The immense chemical diversity inherent in nature provides a promising avenue for discovering novel antiparasitic chemotypes, with recent advances in analytical techniques facilitating the identification of bioactive compounds from plants, microorganisms, and marine organisms [90].
Table 3: Promising Repurposing Candidates for Protozoal Diseases
| Drug Candidate | Original Indication | Target Protozoan | Mechanism of Action | Experimental Evidence |
|---|---|---|---|---|
| Nystatin | Antifungal [86] | Trypanosoma cruzi [86] | Inhibits arginine uptake, membrane disruption [86] | IC50 0.17 μM (epimastigotes), 4.90 μM (trypomastigotes) [86] |
| Gamma-Oryzanol | Dietary supplement [92] | Plasmodium falciparum [92] | Plasmepsin II inhibition [92] | MM-GBSA binding energy: -54.26 ± 2.92 kcal/mol [92] |
| Azilsartan | Antihypertensive [92] | Plasmodium falciparum [92] | Plasmepsin II inhibition [92] | MM-GBSA binding energy: -45.32 ± 3.87 kcal/mol [92] |
| Anticancer drugs (e.g., Idelalisib) | Cancer therapy [85] | Plasmodium spp. [85] | Not fully elucidated [85] | Activity against chloroquine-resistant and artemisinin-sensitive strains [85] |
The Scientist's Toolkit: Essential Research Reagents and Platforms
Table 4: Key Research Reagent Solutions for Drug Repurposing Studies
| Reagent/Platform | Function/Application | Specific Examples/Features |
|---|---|---|
| RepurposeDrugs | Interactive web-portal for drug-indication exploration [87] | Database of 4,314 compounds, 161 drug combinations; ML-based approval prediction [87] |
| Molecular Dynamics Software | Simulation of drug-target interactions [92] | 100 ns MD simulations for binding stability assessment [92] |
| Radiolabeled Substrates | Transport assays to evaluate drug effects on nutrient uptake [86] | L-[3H] arginine, [3H] thymidine for metabolite transport studies [86] |
| Parasite Culture Media | Maintenance of protozoan parasites in vitro [86] | Brain-heart infusion-tryptose (BHT) for T. cruzi; supplemented with FCS, antibiotics [86] |
| UKEDR Framework | Deep learning approach for drug repositioning predictions [93] | Integrates knowledge graph embedding, pre-training strategies, recommendation systems [93] |
Drug repurposing represents a promising strategy for addressing the significant unmet therapeutic needs in protozoal diseases, particularly those with zoonotic transmission pathways. The approach leverages existing pharmacological knowledge and compounds to accelerate the development of new treatment options, potentially reducing both time and costs compared to traditional drug discovery pipelines. Computational methods, including AI and machine learning, have dramatically enhanced our ability to identify promising repurposing candidates, while experimental approaches provide essential validation of efficacy and mechanistic insights.
The fight against protozoal diseases requires continued innovation and collaboration across disciplines. As drug resistance spreads and climate change expands the geographical range of many parasitic diseases, the need for effective therapeutics becomes increasingly urgent. Drug repurposing offers a pragmatic pathway to address these challenges, potentially bringing new hope to millions affected by these neglected diseases.
Evidence and Evaluation: Validating Diagnostics, Models, and Control Strategies
Intestinal protozoan infections represent a significant global health burden, causing millions of cases of diarrheal disease annually and disproportionately affecting populations in areas with poor sanitation [18]. Accurate diagnosis is fundamental to clinical management, public health surveillance, and drug development efforts, particularly given the zoonotic transmission pathways of many protozoan parasites [94]. For decades, microscopic examination of stool specimens has been the cornerstone of diagnosis. However, technological advances have introduced immunodiagnostic methods like the Enzyme-Linked Immunosorbent Assay (ELISA) and molecular techniques such as Polymerase Chain Reaction (PCR), each with distinct advantages and limitations.
This technical guide provides an in-depth comparison of the sensitivity of microscopy, ELISA, and PCR for detecting pathogenic intestinal protozoa. Framed within the context of zoonotic transmission research, it summarizes critical performance data, details essential experimental protocols, and offers a practical toolkit for researchers and drug development professionals navigating the complex landscape of diagnostic validation.
Comparative Analysis of Diagnostic Sensitivity
The diagnostic sensitivity of a test—its ability to correctly identify positive samples—is a primary metric for evaluation. The table below synthesizes findings from recent studies directly comparing microscopy, ELISA, and PCR for key protozoan parasites.
Table 1: Comparative Sensitivity of Diagnostic Methods for Intestinal Protozoa
| Parasite | Microscopy Sensitivity | ELISA/RDT Sensitivity | PCR/qPCR Sensitivity | Key Comparative Findings |
|---|---|---|---|---|
| Giardia lamblia | 20-90% [95] | Variable: 58%-100% for RDTs [96] | High; complete agreement with best RDTs [97] [96] | PCR and top-tier RDTs show superior and more consistent sensitivity than microscopy [96]. |
| Cryptosporidium spp. | Low; requires special staining [96] | High: 91.7%-100% for RDTs [96] | High [97] | Antigen-detection tests and PCR are significantly more sensitive than unstained microscopy [96]. |
| Entamoeba histolytica | Cannot distinguish from non-pathogenic E. dispar [96] | High sensitivity, but may cross-react with E. dispar [96] | High; allows species-level differentiation [96] | PCR is critical for accurate differentiation from E. dispar, which microscopy cannot achieve [97] [96]. |
| Dientamoeba fragilis | ~0.63% (in a large prospective study) [98] | Not widely available | ~8.86% (in a large prospective study) [98] | PCR is drastically more sensitive than microscopy for this easily missed pathogen [98]. |
| Blastocystis spp. | ~6.55% (in a large prospective study) [98] | Not widely available | ~19.25% (in a large prospective study) [98] | PCR detects a significantly higher number of infections compared to microscopy [98]. |
| General Protozoa | Detects 58-72% of positives with a single sample [95] | Varies by target and test format | Detected protozoa in 74.4% of samples vs. lower microscopy yield [99] | Multiplex qPCR demonstrates markedly higher overall detection rates for a panel of intestinal protozoa [98] [99]. |
Detailed Experimental Protocols
To ensure reliable and reproducible results, adherence to standardized protocols is essential. The following sections outline core methodologies for the three diagnostic approaches.
Microscopic Examination (Ova and Parasite Examination)
Principle: Visual identification of cysts, trophozoites, oocysts, or helminth eggs in stool samples, often aided by concentration techniques and special stains.
Workflow:
- Specimen Collection: Collect fresh stool samples in clean, sealed containers. Multiple samples (e.g., 3 collected every other day) are required to improve sensitivity [95].
- Preservation: Preserve samples immediately in appropriate fixatives (e.g., 10% formalin, sodium acetate-acetic acid-formalin (SAF)) to preserve parasite morphology [95].
- Concentration:
- Staining:
- Direct Smear: Examine a saline and/or iodine wet mount for motile trophozoites and cysts.
- Permanent Stain: Use stains like trichrome for detailed observation of internal structures, aiding in species identification.
- Acid-Fast Staining: Essential for visualizing Cryptosporidium oocysts [96].
- Examination: Systematically scan the prepared slides under a microscope (10x and 40x objectives) by a trained technologist.
Antigen Detection by ELISA
Principle: Detection of parasite-specific antigens in stool samples using antibodies immobilized on a microplate.
Protocol (Indirect ELISA for Serology, adapted from [100]):
- Coating: Coat wells of a microplate with a capture antigen (e.g., Excretory/Secretory Antigens for Fasciola). Incubate overnight at 37°C.
- Washing: Wash plates with phosphate-buffered saline containing Tween 20 (PBST) to remove unbound antigen.
- Blocking: Add a blocking buffer (e.g., 3% skimmed milk in PBST) to cover unbound sites. Incubate for 2 hours at 37°C.
- Sample Incubation: Add diluted patient serum or stool supernatant to the wells. Incubate at 37°C for 30 minutes. Wash.
- Conjugate Incubation: Add a species-specific antibody conjugated to an enzyme (e.g., anti-human IgG horseradish peroxidase). Incubate at 37°C for 30 minutes. Wash.
- Substrate Addition: Add an enzyme substrate (e.g., OPD - o-phenylenediamine dihydrochloride). Incubate in the dark for 30 minutes.
- Stop and Read: Stop the reaction with sulfuric acid. Measure the optical density (OD) immediately with a spectrophotometer.
Molecular Detection by Real-Time PCR (qPCR)
Principle: Amplification and detection of parasite-specific DNA sequences, allowing for sensitive and species-specific identification.
Protocol (Duplex qPCR, adapted from [99]):
- DNA Extraction: Extract genomic DNA from stool samples preserved in appropriate buffers using a commercial genomic DNA kit. This is a critical step for assay performance [97].
- Primer/Probe Design: Design primers and TaqMan probes targeting conserved, species-specific genetic regions (e.g., SSU rRNA, ITS1). Ensure no cross-reactivity by checking against databases [99].
- Reaction Setup: Prepare a 10 µL reaction mixture containing:
- Master Mix (e.g., containing DNA polymerase, dNTPs, MgCl₂)
- Forward and Reverse Primers (optimized concentration)
- TaqMan Probes for different targets (e.g., FAM-labeled for one parasite, HEX-labeled for another, Texas Red for an internal control)
- Template DNA
- Amplification: Run the plate on a real-time PCR cycler with the following typical conditions:
- Initial Denaturation: 95°C for 2-5 minutes
- 40-50 cycles of:
- Denaturation: 95°C for 15 seconds
- Annealing/Extension: 60°C for 30-60 seconds (data acquisition)
- Analysis: Analyze the cycle threshold (Ct) values. A sample is considered positive if amplification exceeds the background signal within a defined cycle threshold.
Diagram 1: Diagnostic method workflows for parasite detection.
The Scientist's Toolkit: Research Reagent Solutions
Successful diagnostic validation relies on a suite of specific reagents and tools. The following table catalogues key solutions for researching intestinal protozoa.
Table 2: Essential Research Reagents for Protozoan Diagnostic Validation
| Reagent / Material | Function / Application | Example Use-Case |
|---|---|---|
| Excretory/Secretory Antigens (ES Ags) | Coating antigen for ELISA to detect host antibodies against parasites. | Serodiagnosis of human fascioliasis [100] and other helminth infections. |
| Parasite-Specific Primers & TaqMan Probes | Amplification and detection of unique DNA sequences in qPCR assays. | Duplex qPCR for simultaneous detection of Entamoeba histolytica and E. dispar [99]. |
| Commercial DNA Extraction Kits | Standardized purification of high-quality genomic DNA from complex stool samples. | Critical first step for all PCR-based detection; impacts sensitivity [97] [100]. |
| Multiplex PCR Panels | Simultaneous detection of multiple pathogens (bacteria, viruses, parasites) in a single reaction. | Syndromic testing for gastroenteritis (e.g., Luminex xTAG GPP) [95]. |
| Monoclonal Antibodies for Antigen Detection | Key component of Rapid Diagnostic Tests (RDTs) and ELISAs for specific parasite antigens. | Immunochromatographic lateral-flow tests for Giardia and Cryptosporidium [96]. |
| Plasmids with Target Inserts | Positive controls and for generating standard curves in qPCR assay development and validation. | Determining the limit of detection and quantification for a novel qPCR assay [99]. |
Diagnostic Strategy in Zoonosis Research
Selecting the appropriate diagnostic method depends on the research question, resources, and required performance. The following decision logic can guide researchers in structuring their diagnostic approach.
Diagram 2: Diagnostic method selection logic for research.
The diagnostic landscape for intestinal protozoa is evolving from reliance on traditional microscopy toward the integration of highly sensitive and specific molecular tools. While microscopy remains crucial for detecting a broad range of parasites and is indispensable in specific scenarios, the data consistently demonstrate the superior sensitivity of qPCR [98] [99]. ELISA and RDTs offer a valuable middle ground for high-throughput or rapid screening for specific pathogens.
For researchers investigating the zoonotic transmission pathways of intestinal protozoa, the choice of diagnostic method directly impacts the accuracy of prevalence studies, the understanding of transmission dynamics, and the evaluation of new interventions. A layered strategy, often starting with a sensitive multiplex qPCR followed by microscopic examination for specific populations or parasites, represents the most robust approach for generating reliable data to inform public health and drug development efforts.
Within the complex domain of infectious disease dynamics, mathematical and computational models serve as indispensable tools for understanding transmission patterns, evaluating intervention strategies, and forecasting outbreak trajectories. Their utility extends profoundly to the study of zoonotic pathogens, such as intestinal protozoa, where transmission bridges animal reservoirs and human populations, creating intricate ecosystems that are challenging to decipher. The reliability of these models, however, is entirely contingent upon rigorous validation processes. Model validation provides the critical foundation for scientific credibility, ensuring that simulations accurately reflect real-world phenomena and that predictions can confidently inform public health policy and drug development efforts.
This technical guide examines the core principles and methodologies of model validation through case studies of influenza, measles, and Ebola outbreaks. By exploring these viral examples, we establish a robust framework that can be directly adapted and applied to the study of zoonotic transmission pathways of intestinal protozoa. The validation techniques detailed herein—from comparing predicted curves with observed epidemiological data to assessing the accuracy of machine learning forecasts—provide a essential toolkit for researchers aiming to build trustworthy models of parasitic diseases like cryptosporidiosis and giardiasis, for which animal-to-human transmission is a key concern.
Core Principles of Epidemiological Model Validation
Model validation is a multi-faceted process that assesses whether a computational model is an accurate and reliable representation of the real-world system it is intended to simulate. In epidemiological contexts, this process ensures that a model's outputs align with observed disease dynamics and that its predictions are sufficiently robust to guide decision-making.
- Conceptual Validity: This initial stage verifies that the model's structure, assumptions, and underlying mechanisms are logically sound and consistent with established biological and epidemiological principles. For a zoonotic protozoa model, this would involve correctly representing transmission routes (e.g., fecal-oral), host-parasite interactions, and environmental persistence of cysts or oocysts.
- Operational Validity: This step assesses the model's performance by comparing its output against historical or experimental data. A key quantitative metric is the goodness-of-fit, which statistically evaluates how closely the model's simulation matches empirical observations.
- Predictive Validity: The most rigorous test of a model is its ability to forecast future events or system behaviors not used in its calibration. Predictive validation involves "testing the model against new data, i.e. data that have not been used for the model calibration" to ensure it can generalize beyond a single fitted scenario [101].
Case Study 1: Influenza
Challenge Models and Viral Shedding Dynamics
Human challenge studies represent a unique form of validation, where a controlled experiment provides high-resolution data against which transmission models can be calibrated. In one such study, a wild-type influenza A/H1N1pdm09 virus was administered intranasally to healthy volunteers [102].
Table 1: Key Findings from Influenza A/H1N1 Challenge Model [102]
| Parameter | Finding | Implication for Transmission Modeling |
|---|---|---|
| Infectious Dose | 10^7 TCID₅₀ induced disease in 69% of volunteers | Provides a precise value for initial conditions in models. |
| Viral Shedding | Mean shedding for 4-5 days, detected via qRT-PCR | Informs the duration of the infectious period in model compartments. |
| Shedding vs. Symptoms | Shedding peaked 12-24 hours before symptom onset | Validates models that incorporate pre-symptomatic transmission. |
| Serological Response | Significant rise in convalescent HAI and NAI titers | Allows model validation against immunologic data, not just infection. |
The study's findings underscore the importance of pre-symptomatic transmission, a dynamic that must be accurately parameterized in models to reflect true spread. For protozoal research, similar controlled studies, though ethically complex, could define critical parameters such as the infectious dose of Cryptosporidium oocysts and the timing of cyst shedding in relation to clinical symptoms.
Predictive Validation of Complex Simulation Models
Beyond initial fitting, models must be validated against entirely independent outbreaks. In one investigation, an individual-based model (IBM) was fitted to a single influenza epidemic (1998-99) and then tasked with forecasting the intensity and timing of subsequent epidemics (1999-2006) without further calibration [101]. This process of testing a model's predictive power against future, unseen epidemics is a cornerstone of robust validation. The study demonstrated that while complex models could forecast absolute intensity and the peak week of an epidemic, the reliability of these predictions was highly dependent on the forecasting method (static or dynamic) [101].
Another advanced approach uses ensemble modeling to improve prediction robustness. One framework combined bootstrap aggregating (bagging) with rank aggregation to optimize penalized regression models (lasso, ridge, elastic net) that used internet search query data to monitor influenza activity. This ensemble method improved the predictability of conventional models by reducing overfitting and more effectively identifying informative predictors from a large pool of candidate search terms [103].
Diagram 1: Predictive validation workflow for influenza models.
Case Study 2: Measles
Real-Time Modeling for Outbreak Response
The 2015 measles outbreak in Lola Prefecture, Guinea, provided a salient case study in real-time model validation during an active public health crisis. Modelers faced the dual challenge of estimating the size of the susceptible population and forecasting future cases to guide the deployment of a vaccination campaign [104]. Three distinct methodological approaches were employed to estimate susceptibility, each providing a unique lens for validation:
- Method 1 (Orenstein et al.): Utilized the proportion of cases occurring in vaccinated individuals and vaccine efficacy (VE) to back-calculate the proportion of the population vaccinated and susceptible.
- Method 2 (Demographic Survey Integration): Combined pre-existing Demographic and Health Surveys (DHS) data with models accounting for a 25% reduction in vaccination rates due to the concurrent Ebola epidemic, using spatially explicit data on birth rates and population distributions [104].
- Method 3 (TSIR Model): Fitted a time-series susceptible–infected–recovered (TSIR) model to the initial epidemic curve using Markov chain Monte Carlo (MCMC) methods, assuming values for the basic reproductive number (R₀) consistent with measles literature [104].
Table 2: Measles Susceptibility Estimation Methods in Lola Prefecture [104]
| Method | Key Inputs | Estimated Susceptibility (Ages 1-5) | Advantages & Limitations |
|---|---|---|---|
| Orenstein (Case Data) | Case vaccination status, Vaccine Efficacy (86-97%) | Not explicitly stated | Directly uses outbreak data; sensitive to case ascertainment. |
| Demographic Model | DHS data, assumed 25% vaccination drop, WorldPop data | 24% (95% CI: 2-65%) | Incorporates external health system shocks; relies on assumptions of vaccination reduction. |
| TSIR Model | Initial epidemic curve, Assumed R₀ (8, 12, 18) | 43% (implied) | Dynamically incorporates transmission; sensitive to initial conditions and R₀ assumption. |
The convergence of these independent methods on a high baseline susceptibility (24%-43%) provided health authorities with validated, actionable intelligence, demonstrating how triangulation of evidence from multiple models strengthens validation confidence during an outbreak.
Machine Learning for Case Prediction
More recently, a machine learning model was developed to predict measles cases at the US county level [105]. The model was trained on 2014 and 2018 US county-level measles case data using 17 predictor variables and was subsequently tested on data from 2019. The model demonstrated high predictive validity, accurately identifying 72% (sensitivity) of the counties that experienced one or more measles cases in 2019, which accounted for 94% of all cases that year. Its specificity was 95%, accurately predicting the vast majority of counties without cases [105]. This showcases the power of machine learning for high-resolution risk assessment when validated against a hold-out dataset.
Case Study 3: Ebola
Compartmental Model Stability Analysis
Mathematical models for Ebola virus disease (EVD) have been developed to understand spread and evaluate control strategies. One study created a deterministic compartmental model (SLIRD) dividing the population into Susceptible (S), Latent (L, infected but not infectious), Infectious and Isolated (I), Recovered (R), and Dead (D) compartments [106]. A critical validation step involved stability analysis of the model's equilibrium points.
The analysis revealed that the disease-free state (where the entire population is susceptible) was unstable if traditional burial practices (a major transmission route) were allowed in the model. However, when these practices were prohibited in the model (reflecting actual public health guidance), the endemic-free equilibrium became stable if the basic reproductive number (R₀) was less than 1 [106]. This finding was consistent with empirical knowledge that safe burials are essential for outbreak control, thereby providing conceptual and operational validity to the model structure.
Predictive Spillover Risk Modeling
Leveraging ecological drivers, a predictive model was developed to estimate the annual relative odds of Ebolavirus spillover from animal reservoirs to humans across equatorial Africa [107]. The model incorporated high-resolution, annual data on forest loss, forest fragmentation, and human population distribution from 2001-2021.
The model was validated using a leave-one-year-out cross-validation approach, which tests the model's ability to predict the spillover events of any given year when it was not trained on data from that year. The multispecies analysis achieved a cross-validation AUC (Area Under the Curve) of 0.88, indicating high predictive accuracy [107]. When applied to predict spillover locations in 2022, the model successfully identified that spillover events occurred in locations that were in the top 0.1% of its overall risk estimates or where risk estimates had increased from the previous year [107]. This powerful application demonstrates how validation against future events confirms a model's utility in preemptively targeting surveillance.
Diagram 2: Framework for predictive Ebola spillover risk modeling.
Synthesis: A Generalized Validation Framework for Zoonotic Protozoa Research
The case studies reveal a common, iterative pathway for robust model validation. This pathway begins with conceptual model design based on biological principles, proceeds to operational calibration and fitting, and culminates in predictive testing against independent data. This framework is directly applicable to modeling the zoonotic transmission of intestinal protozoa like Cryptosporidium spp., Giardia intestinalis, and Enterocytozoon bieneusi.
For instance, a model of Giardia transmission in a community with domestic dogs would first be calibrated using prevalence data from cross-sectional studies, such as one finding an overall protozoa prevalence of 31.87% in humans and 78% in domestic dogs [12]. Predictive validity could then be tested by assessing the model's ability to forecast incidence in a neighboring community or following an intervention like a dog deworming campaign. Furthermore, just as Ebola spillover models integrated forest cover data, protozoa models must integrate environmental covariates such as sanitation infrastructure, water source, and temperature/humidity, which are known to significantly influence protozoan transmission and were identified as key risk factors in recent studies [12] [56].
The Scientist's Toolkit: Research Reagent Solutions
The following table details key reagents and materials essential for conducting the foundational research that informs and validates epidemiological models of intestinal protozoa.
Table 3: Essential Research Reagents for Zoonotic Intestinal Protozoa Studies
| Reagent/Material | Function in Experimental Protocols | Specific Application Example |
|---|---|---|
| E.Z.N.A. Stool DNA Kit | Extraction of high-quality genomic DNA from complex fecal samples. | Standardized DNA extraction for subsequent PCR-based detection and genotyping of Cryptosporidium, Giardia, and E. bieneusi [56]. |
| PCR Primers (SSU rRNA, ITS) | Target-specific amplification of parasite DNA for detection and genotyping. | Nested PCR for detecting C. cayetanensis (SSU rRNA) and genotyping E. bieneusi (ITS region) to track zoonotic transmission [56]. |
| Bootstrap Aggregating (Bagging) | A resampling technique to improve stability and predictive performance of statistical models. | Creating an ensemble penalized regression model to reduce overfitting and enhance the prediction of influenza activity from search query data [103]. |
| Markov Chain Monte Carlo (MCMC) | A computational algorithm for fitting complex models to data and estimating parameter distributions. | Fitting a TSIR model to an initial measles outbreak curve to estimate susceptibility and project future cases in real-time [104]. |
| Boosted Regression Trees (BRT) | A machine learning method that combines multiple simple models for improved predictive accuracy. | Developing a spatial model to estimate the relative odds of Ebolavirus spillover based on ecological and human population predictors [107]. |
Intestinal protozoal infections (IPIs) represent a significant global health burden, with transmission dynamics and prevalence rates exhibiting substantial geographic variation. Understanding this variation is crucial for the development of targeted control strategies within the broader context of zoonotic transmission pathways. This technical review synthesizes current epidemiological data from diverse endemic regions, highlighting distinct prevalence patterns of key intestinal protozoa including Giardia duodenalis, Cryptosporidium spp., and Entamoeba histolytica. The analysis encompasses multiple geographic scales—from international comparisons to regional and community-level variations—and examines the complex interplay of environmental, socioeconomic, and host factors that drive these differences. The findings presented herein provide a scientific foundation for risk assessment, resource allocation, and the design of evidence-based interventions for researchers, public health professionals, and drug development specialists working in the field of parasitic zoonoses.
Global and Regional Prevalence Patterns
Epidemiological surveillance data reveal striking geographic disparities in the prevalence of intestinal protozoa, influenced by a complex interplay of climatic, socioeconomic, and ecological factors.
Table 1: Comparative Prevalence of Intestinal Protozoa in Human Populations Across Geographic Regions
| Region/Country | Study Population | Overall IPI Prevalence | Giardia spp. Prevalence | Cryptosporidium spp. Prevalence | Entamoeba histolytica Prevalence | Primary Risk Factors |
|---|---|---|---|---|---|---|
| Malaysia (Nationwide) | General Population | 24% (95% CI: 17.0-29.0) [108] [109] | 11% (95% CI: 0.08-0.14) [108] [109] | 9% (95% CI: 0.03-0.14) [108] [109] | 18% (95% CI: 0.12-0.24) [108] [109] | Indigenous status, rural residence, low income, poor sanitation, untreated water [108] [109] |
| Slovakia (Rožňava region) | General Population | 5.95% [110] | G. duodenalis: Most prevalent species [110] | Not specified | Not specified | Roma ethnicity, rural residence, poor sanitation, low education [110] |
| Ecuador (Coastal Communities) | Marginalized Communities | 31.87% [12] | Not specified | Not specified | E. histolytica: 10% [12] E. coli: 18.13% [12] | Poor sanitation, inadequate water infrastructure, barefoot walking [12] |
| Türkiye (Van Province) | Disabled Patients | 41% (Patient group) [111] | G. intestinalis: 9% [111] | Cryptosporidium spp.: 15% [111] | E. coli: 4% [111] | Disability (especially spina bifida), poor hygiene [111] |
At the global scale, intestinal protozoa demonstrate endemicity patterns particularly concentrated in tropical and subtropical regions. A comprehensive meta-analysis from Malaysia revealed an overall pooled IPI prevalence of 24%, with Entamoeba spp. (18%) being most prevalent, followed by Giardia lamblia (11%) and Cryptosporidium spp. (9%) [108] [109]. This contrasts with data from Slovakia, where a regional study reported a much lower overall prevalence of 5.95%, with Giardia duodenalis and Ascaris lumbricoides as the dominant pathogens [110].
Significant within-country variation highlights the importance of subnational epidemiological assessment. In Malaysia, state-level analysis found Kelantan and Perak had the highest prevalence (39% and 29% respectively), while Selangor and Kuala Lumpur reported the lowest (13.6%) [108] [109]. Similarly, in Slovakia, infection rates were significantly higher among Roma communities and residents of rural areas, illustrating how socioeconomic and ethnic disparities drive disease distribution [110].
Vulnerable populations consistently demonstrate elevated prevalence rates regardless of geographic setting. In Türkiye, disabled individuals attending a rehabilitation center showed a 41% prevalence of intestinal protozoa, markedly higher than the 9% found in non-disabled controls [111]. Particularly striking was the infection rate among spina bifida patients (83.3%), highlighting the intersection of disability, hygiene challenges, and biological susceptibility [111]. Indigenous communities in Malaysia also demonstrated high burden (27% prevalence), followed by local rural communities (23%) [108] [109].
Zoonotic Transmission Pathways and Animal Reservoirs
Understanding the zoonotic transmission dynamics of intestinal protozoa requires examination of animal reservoirs and human-animal-environment interfaces across different ecological settings.
Table 2: Zoonotic Parasites in Domestic Dogs Across Geographic Regions
| Location | Study Population | Giardia duodenalis Prevalence | Cryptosporidium spp. Prevalence | Ancylostoma caninum Prevalence | Toxocara canis Prevalence | Zoonotic Genotypes Identified |
|---|---|---|---|---|---|---|
| South Korea | Shelter Dogs (n=846) | 5.4% [112] | 0.5% [112] | Not specified | Not specified | G. duodenalis assemblages C, D, F; E. bieneusi zoonotic group 1; Blastocystis ST4 [112] |
| Ecuador | Domestic Dogs (n=500) | Not specified | Not specified | 53.6% [12] | 12.4% [12] | Cutaneous larva migrans, hydatidosis, strongyloidiasis [12] |
Animal reservoirs play a crucial role in maintaining transmission cycles of intestinal protozoa. Research from South Korea identified a 7.4% positivity rate for zoonotic pathogens in shelter dogs, with Giardia duodenalis being most prevalent (5.4%) [112]. Molecular characterization revealed zoonotic genotypes including G. duodenalis assemblages C, D, and F, and the Blastocystis sp. ST4 subtype [112]. In Ecuador, studies of human-dog parasitic relationships in marginalized coastal communities found an overall prevalence of 78% in domestic dogs, with Ancylostoma caninum (53.6%) and Taenia spp. (15.2%) as the most prevalent parasites [12].
The human-animal interface represents a critical point of cross-species transmission. In Ecuadorian coastal communities, significant associations were found between parasitic infections and factors such as water source, sanitation infrastructure, barefoot walking, and peridomiciliary habitat of dogs [12]. Similarly, in Slovakia, the presence of stray dogs in Roma settlements was identified as a contributing factor to environmental contamination and parasite transmission [110].
Environmental transmission pathways further complicate the ecoepidemiology of these parasites. The Slovakian study identified population density, access to water and sanitation, and education levels as key factors influencing infection rates [110]. Overcrowding (with approximately eight people per dwelling in Roma settlements compared to the national average of 2.5) and lack of running water (affecting nearly 36% of Roma residents) created ideal conditions for sustained parasite transmission [110].
Age-Specific Infection Dynamics in Animal Models
Cattle populations provide valuable insights into age-structured infection dynamics that inform our understanding of transmission patterns and susceptibility across host development.
Table 3: Age-Stratified Prevalence of Intestinal Protozoa in Calves from Kazakhstan Dairy Farms
| Age Group (Days) | Cryptosporidium spp. Prevalence | Eimeria spp. Prevalence | Giardia spp. Prevalence |
|---|---|---|---|
| 1-30 days (n=not specified) | 49.2% [38] [21] | 2.0% [38] [21] | 5.2% [38] [21] |
| 31-90 days (n=not specified) | Significant decrease (p<0.001) [38] [21] | 27.3 times higher odds than reference (95% CI: 17.07-45.35, p<0.001) [38] [21] | No statistically significant variation [38] [21] |
| 91-120 days (n=not specified) | Continued low prevalence [38] [21] | Elevated odds persist (p<0.001) [38] [21] | No statistically significant variation [38] [21] |
| >120 days (n=not specified) | Continued low prevalence [38] [21] | Elevated odds persist (p<0.001) [38] [21] | No statistically significant variation [38] [21] |
Research from intensive dairy farms in central and northern Kazakhstan revealed pronounced age-dependent infection patterns in calves [38] [21]. Cryptosporidium spp. infections were highly concentrated in the youngest calves (1-30 days), with prevalence reaching 49.2%, followed by a dramatic decline in older age groups (p<0.001) [38] [21]. Conversely, Eimeria spp. prevalence showed an opposite trend, starting low (2.0%) in the 1-30 day group but increasing significantly with age [38] [21]. Calves aged 31-90 days had 27.3 times higher odds of infection compared to the youngest group, with elevated odds persisting in older cohorts [38] [21]. Giardia spp. demonstrated a more uniform distribution across age groups, with no statistically significant variation observed [38] [21].
These distinct age patterns suggest different transmission dynamics and host-parasite relationships for each protozoan species. The concentration of cryptosporidiosis in neonatal calves indicates either high environmental contamination in calving areas, immature immune function, or age-specific physiological susceptibility [38] [21]. The progressive increase in eimeriosis suggests cumulative exposure with age or developmentally regulated susceptibility [38] [21].
Notably, the Kazakhstan study found no significant seasonal variation in infection rates for any of the protozoa studied, suggesting that in intensive farming systems with standardized management practices, age is a more important determinant of infection risk than season [38] [21]. This has important implications for control strategies, supporting age-targeted rather than seasonal intervention approaches in such operations [38] [21].
Methodologies for Epidemiological Surveillance
Standardized diagnostic approaches are essential for generating comparable data on intestinal protozoa prevalence across different geographic regions and study populations.
Field Sampling Protocols
Stratified random sampling approaches ensure representative data collection across target populations. The Kazakhstan calf study categorized the population into four age groups (1-30, 31-90, 91-120, and >120 days) and collected 1,586 fecal samples from 12 industrialized dairy farms across 11 districts, achieving >10% representation within each age group where possible [38] [21].
Community-based sampling requires careful consideration of ethical and logistical factors. The Ecuador study used convenience sampling of 160 families across urban-marginal and rural sectors, including participants based on ownership of multiple dogs and poor sanitary infrastructure to maximize yield within resource constraints [12]. The Slovakia study collected 2,503 human stool samples from 59 locations, with detailed demographic data including ethnicity, age, gender, and address to enable analysis of risk factors [110].
Laboratory Diagnostic Techniques
Microscopic methods remain widely used for initial detection of intestinal protozoa. The Kazakhstan study employed Fuelleborn, Heine and ZnSO4 flotation microscopic techniques for parasite identification [38] [21]. The Slovakia study used both commercial sedimentation kits (Paraprep L) and SAF (sodium acetate-acetic acid-formalin) modified concentration methods, with microscopic examination at 100×, 400×, and 1000× magnification with immersion oil for protozoan cysts [110]. The Türkiye study combined native-Lugol examination with modified acid-fast staining for detection of Cryptosporidium spp. and Cyclospora cayetanensis [111].
Molecular techniques provide enhanced sensitivity and specificity, plus genotyping capability. The South Korean dog study utilized nested PCR (nPCR) approaches targeting specific genes (bg, tpi, and gdh for G. duodenalis; SSU rRNA for Cryptosporidium spp. and Blastocystis sp.; ITS for E. bieneusi) followed by phylogenetic analysis [112]. DNA extraction was performed using the QIAamp Fast DNA Stool Mini Kit [112].
Statistical Analysis Approaches
Prevalence estimation and risk factor analysis typically employ logistic regression models to estimate odds ratios (ORs) and 95% confidence intervals (CIs). The Kazakhstan study used ANOVA and a generalized additive logistic model (GAM) to examine effects of age and season on infection likelihood [38] [21]. The Malaysia meta-analysis employed random effects models to calculate pooled prevalence estimates with 95% CIs, accounting for high heterogeneity (I² > 98%) across studies [108] [109].
Spatial analysis techniques can identify high-risk areas for targeted interventions. The Slovakia study used geographic information systems and statistical modeling to create risk maps highlighting villages with higher transmission potential, correlating infection data with environmental and socioeconomic variables [110].
Research Reagent Solutions Toolkit
Table 4: Essential Research Reagents and Kits for Intestinal Protozoa Studies
| Reagent/Kit | Application | Specific Use Case | Reference |
|---|---|---|---|
| QIAamp Fast DNA Stool Mini Kit | Genomic DNA extraction from fecal samples | DNA extraction for molecular characterization of parasites in dog fecal samples | [112] |
| Paraprep L Sedimentation Kit | Concentration and microscopic detection of helminth eggs and protozoan cysts | Commercial sedimentation kit for human stool sample analysis | [110] |
| SAF (Sodium Acetate-Acetic Acid-Formalin) Solution | Fecal sample preservation and concentration | Modified concentration technique for detection of helminth eggs and protozoa cysts | [110] |
| ZnSO4 Flotation Solution | Parasite egg and cyst flotation | Microscopic identification of Giardia spp., Cryptosporidium spp., and Eimeria spp. in calf feces | [38] [21] |
| Modified Acid-Fast Staining Reagents | Differential staining of coccidian parasites | Detection of Cryptosporidium spp. and Cyclospora cayetanensis in human stool samples | [111] |
| Native-Lugol Solution | Direct microscopic examination of stool samples | Visualization of intestinal protozoan cysts and trophozoites | [111] |
Integrated Experimental Workflow
The following diagram illustrates a standardized workflow for comparative studies of intestinal protozoa prevalence across geographic regions, integrating both field and laboratory components:
Comparative Prevalence Study Workflow
This integrated workflow encompasses three major phases: (1) Field Sampling employing stratified random sampling approaches with simultaneous collection of demographic and risk factor data; (2) Laboratory Analysis utilizing both traditional microscopic techniques and modern molecular methods for comprehensive parasite detection and characterization; and (3) Data Analysis & Integration applying statistical modeling, risk factor analysis, and spatial mapping to enable comparative assessment across geographic regions. This standardized approach facilitates generation of comparable data on intestinal protozoa prevalence across different ecological and socioeconomic contexts, supporting evidence-based control strategy development.
The geographic variation in intestinal protozoa prevalence reflects complex interactions between pathogen, host, and environmental factors. Key determinants include socioeconomic status, sanitation infrastructure, climate, zoonotic reservoirs, and specific population vulnerabilities. This comparative analysis demonstrates that while certain patterns hold across regions (e.g., higher burden in marginalized communities), the dominant parasite species and specific risk factors show significant geographic variation. Future research should prioritize molecular characterization of parasite genotypes to elucidate transmission dynamics and zoonotic potential, while control programs require tailored approaches that address the specific epidemiological context of each region. The integration of traditional parasitological methods with modern molecular tools and spatial analysis provides the most comprehensive approach for understanding and addressing the geographic variation of intestinal protozoa in the context of zoonotic transmission pathways.
The One Health (OH) approach is an integrated, unifying framework that aims to sustainably balance and optimize the health of people, animals, and ecosystems [113]. This conceptual approach stems from the recognition that approximately 75% of emerging and re-emerging infectious diseases are of zoonotic origin, with parasitic zoonoses constituting a significant portion of this burden [114] [113]. The approach advocates for increased collaboration among diverse scientific disciplines to mitigate complex health problems that intersect human, animal, and environmental domains [114] [115]. The OH framework is particularly relevant for studying intestinal protozoa due to their complex transmission dynamics that frequently involve multiple hosts and environmental reservoirs [114] [8].
The fundamental principle of OH implementation involves simultaneous assessment of all three domains (human, animal, and environment) to fully understand transmission pathways and develop effective control strategies [114] [116]. This holistic perspective is especially crucial for protozoan parasites with complex life cycles and multiple transmission routes. Despite its intuitive appeal, the practical application of the OH approach faces significant challenges, including disciplinary silos, insufficient integrated surveillance, and the need for evidence of its quantitative benefits [115] [113]. This technical assessment synthesizes current evidence and methodologies for evaluating the efficacy of OH strategies in controlling intestinal protozoan infections, with particular emphasis on transmission pathways and intervention effectiveness.
Quantitative Evidence of One Health Efficacy
Global Assessment of OH Outcomes
A comprehensive scoping review of OH implementations has provided quantitative evidence supporting its effectiveness across various health challenges [115]. This analysis of 85 studies revealed that the majority reported positive or partially positive outcomes across both monetary and non-monetary metrics. The top biotic health issues addressed in these studies included rabies and malaria, while air pollution was the predominant abiotic focus [115]. The collaborations most frequently reported were between human and animal health sectors (n=42) and human and environmental disciplines (n=41) [115].
Monetary outcomes were typically expressed as cost-benefit or cost-utility ratios, while non-monetary outcomes were described using disease frequency or disease burden measurements such as Disability-Adjusted Life Years (DALYs) [115]. These quantitative assessments provide crucial evidence for policymakers and funding agencies regarding the value of cross-sectoral approaches to zoonotic disease control.
Efficacy in Intestinal Protozoa Control
Recent field studies demonstrate the practical efficacy of OH approaches in controlling intestinal protozoa across diverse geographical settings:
Table 1: One Health Efficacy in Intestinal Protozoa Control Across Geographic Settings
| Location | Human Prevalence | Animal Prevalence | Environmental Contamination | Key Protozoa Identified | Proposed Transmission Pathways |
|---|---|---|---|---|---|
| Rural Iraq [8] | Cryptosporidium spp. (12%), Blastocystis spp. (16%) | Cryptosporidium spp. (26%), Blastocystis spp. (78%) | Cryptosporidium spp. in water (15%), soil (5%); Blastocystis spp. in soil (45%) | Cryptosporidium parvum (zoonotic strain in humans and sheep), Blastocystis ST1 (humans), ST4/ST10 (sheep) | Zoonotic (C. parvum), environmental (water, soil) |
| Urban Chile [116] [117] | Overall parasites (28%), T. canis seropositivity (33%) | Owned dogs (26%), Stray dog feces (44%) | Soil contamination with zoonotic parasites (30.5% of park samples) | Giardia duodenalis, Blastocystis sp., Toxocara sp. | Environmental (soil contamination with dog feces) |
| Rural Madagascar [118] | Blastocystis spp. (74%) | Domesticated animals (various subtypes) | Not specified | Six distinct Blastocystis subtypes | Environmental (proximity to infected households), not social or zoonotic |
| Ecuadorian Coast [12] | Overall parasites (31.87%) | Domestic dogs (78%) | Not directly sampled | Entamoeba coli (18.13%), E. histolytica (10%) in humans; Ancylostoma caninum (53.6%) in dogs | Associated with water source, sanitation, barefoot walking, peridomiciliary dogs |
The Iraq study demonstrated molecular evidence of zoonotic transmission through the identification of identical Cryptosporidium parvum strains in both humans and sheep [8]. The urban Chile study highlighted the role of environmental contamination with zoonotic parasites in public spaces, finding that soil contamination with parasites such as Toxocara sp. and Trichuris vulpis reached 30.5% in park samples [116]. Notably, the Madagascar study utilized sophisticated network analysis to determine that environmental transmission (specifically, close proximity to infected households) was the most significant predictor of Blastocystis infection, with an odds ratio of 1.56 (95% CI: 1.33-1.82), while social contacts and zoonotic transmission from owned animals were not significant predictors [118].
Experimental Protocols for One Health Parasitology Studies
Integrated Sampling Methodologies
Implementing a comprehensive OH study requires systematic sampling across all three domains using standardized protocols:
Human Sampling Protocols:
- Stool Collection: Participants provide fecal samples collected in containers with PAF (Phenol, Alcohol, and Formaldehyde) fixative for parasitological analysis and in 70% ethanol for molecular analysis [116]. Multiple samples collected every other day increase detection sensitivity [116].
- Questionnaire Administration: Comprehensive surveys collecting data on socioeconomic conditions, water sources, sanitation facilities, animal contact, and hygiene behaviors [8] [116] [12].
- Serological Testing: Blood collection by venipuncture, centrifugation at 3,000 rpm for 5 minutes, with serum stored at -20°C until analysis by ELISA or similar methods [116].
Animal Sampling Protocols:
- Domestic Animal Sampling: Fecal collection from owned dogs, sheep, goats, or other relevant species with proper host identification [8] [116] [12].
- Wildlife/Synanthropic Sampling: Trapping of relevant wildlife species (e.g., Norway rats in urban areas) with ethical euthanasia and collection of intestinal content preserved in 70-85% ethanol [119].
- Sample Processing: Similar processing as human samples to enable cross-species comparisons.
Environmental Sampling Protocols:
- Soil Sampling: Collection of 5g soil samples from a depth of 3-5 cm in relevant locations (public parks, household gardens, animal grazing areas) [8] [116]. Samples are dried for 48 hours at room temperature before processing.
- Water Sampling: Collection from various sources (drinking water, tap water, well water, river water) in sterile containers [8].
- Processing Techniques: Zinc sulfate flotation method for soil samples [116], filtration and concentration for water samples.
Laboratory Diagnostic Techniques
Modern OH parasitology studies employ integrated diagnostic approaches combining classical and molecular methods:
Table 2: Laboratory Methods for One Health Parasitology Studies
| Method Category | Specific Techniques | Applications | Advantages | Limitations |
|---|---|---|---|---|
| Microscopy | Direct wet mounts, iodine staining, concentration methods (flotation, sedimentation), Modified Burrows Method | Initial detection of parasitic forms in fecal and environmental samples | Low cost, equipment accessibility, can detect multiple parasites | Lower sensitivity, requires expertise, cannot differentiate species/subtypes |
| Molecular Detection | Multiplex PCR (Allplex Gastrointestinal Panel-Parasite Assay), qPCR, nested PCR, DNA sequencing (SSU rRNA, gp60, bg, tpi, ITS genes) | Species/subtype identification, zoonotic transmission tracking, genetic diversity assessment | High sensitivity and specificity, enables genotyping, detects non-viable organisms | Higher cost, technical expertise required, potential inhibition |
| Serological Assays | ELISA (e.g., NovaLisa for anti-Toxocara canis IgG) | Detection of exposure to parasitic infections, epidemiological studies | Identifies historical exposure, high throughput | Cannot distinguish current from past infections |
| Advanced Molecular | Next-Generation Sequencing (NGS) of target genes (18S rRNA, β-giardin) | Comprehensive subtype characterization, detection of mixed infections | High resolution, detects unexpected subtypes | High cost, bioinformatics expertise required |
The molecular identification of parasites using techniques such as multiplex PCR allows for the high-specificity detection necessary to understand transmission pathways across domains [114] [119]. For instance, the study in Barcelona utilized the Allplex Gastrointestinal Panel-Parasite Assay to simultaneously detect Blastocystis, Giardia duodenalis, Cryptosporidium spp., and Dientamoeba fragilis in urban rat populations [119]. Similarly, the Iraq study employed a combination of qPCR and nested PCR targeting various genetic markers (SSU rRNA, gp60, bg, tpi, ITS) to identify and subtype protozoan parasites across humans, livestock, and environmental samples [8].
Visualization of One Health Research Workflow
The following diagram illustrates the integrated experimental workflow for a comprehensive One Health study of intestinal protozoa:
One Health Research Workflow - This diagram illustrates the integrated, multi-domain approach required for comprehensive One Health studies of intestinal protozoa, from study design through to intervention development.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Essential Research Reagents and Materials for One Health Parasitology
| Category | Specific Items | Application & Function |
|---|---|---|
| Sample Collection & Preservation | PAF fixative (Phenol, Alcohol, Formaldehyde), 70% ethanol, sterile containers, blood collection tubes without anticoagulant | Preservation of parasitic forms in fecal samples, DNA stabilization for molecular analysis, serum separation for serological testing |
| Microscopy Supplies | Iodine stain, zinc sulfate, salt flotation solutions, slides, coverslips, Parasep SF fecal concentrator | Concentration and visualization of parasitic forms in fecal and environmental samples |
| Nucleic Acid Extraction | QIAamp DNA Stool Mini Kit, PureLink Microbiome Genomic DNA Purification Kit | Isolation of high-quality DNA from complex sample matrices (feces, soil) for molecular detection |
| PCR Reagents | Allplex Gastrointestinal Panel-Parasite Assay, primer sets (SSU rRNA, gp60, bg, tpi, ITS genes), PCR master mixes, positive and negative controls | Specific detection and differentiation of protozoan parasites, subtyping for transmission tracking |
| Sequencing & Analysis | Thermo Scientific GeneJET Gel Extraction Kit, sequencing reagents, bioinformatics software | Purification of PCR products, genetic characterization, phylogenetic analysis |
| Serological Assays | NovaLisa Toxocara canis ELISA kit, ELISA plate readers, conjugate solutions, substrate buffers | Detection of exposure to parasitic infections through antibody recognition |
| Specialized Equipment | CFX96 Real-Time PCR System, centrifuges, thermal cyclers, CO2 euthanasia chamber for ethical animal sampling | Precise temperature cycling for molecular assays, sample preparation, humane animal processing |
The efficacy of One Health approaches in managing intestinal protozoan infections is supported by growing quantitative evidence from diverse geographical settings. Successful implementation requires integrated sampling across human, animal, and environmental domains, combined with molecular epidemiological methods to elucidate transmission pathways. The studies reviewed demonstrate that OH approaches can identify unexpected transmission routes, such as the predominant environmental spread of Blastocystis in Madagascar [118] and the role of urban rats as reservoirs for zoonotic protozoa in Barcelona [119].
Future directions for enhancing OH efficacy include the development of standardized evaluation metrics such as the proposed Global One Health Index (GOHI) [113], which could enable systematic comparison of OH performance across different regions and interventions. Additionally, greater integration of social science methodologies with biological sampling will be crucial for understanding and addressing the socioeconomic drivers of parasitic disease transmission [114] [12]. As climate change, urbanization, and agricultural intensification continue to alter parasite transmission dynamics [116] [119], the OH approach provides an essential framework for developing effective, sustainable strategies to reduce the burden of intestinal protozoan infections in human and animal populations.
Zoonotic protozoal diseases represent a critical challenge at the intersection of human, animal, and environmental health. These diseases, caused by protozoan parasites transmissible between animals and humans, impose substantial economic burdens on global health systems, agricultural productivity, and livelihoods, particularly in low- and middle-income countries (LMICs) [120]. Intestinal protozoa such as Entamoeba histolytica, Giardia duodenalis, Cryptosporidium spp., and Blastocystis spp. affect billions of people worldwide, with an estimated 3.5 billion people affected by intestinal protozoal infections globally [18]. The economic impact extends beyond direct healthcare costs to include reduced labor productivity, diminished agricultural output, and long-term developmental consequences in endemic regions. Understanding and quantifying this burden is essential for researchers, scientists, and drug development professionals working to develop effective interventions and policies within the broader context of zoonotic transmission pathways.
Quantifying the Economic Burden
The economic impact of zoonotic diseases is multifaceted, affecting healthcare systems, agricultural productivity, and national economies. While comprehensive cost analyses specific to protozoal diseases remain limited, available data reveals substantial economic losses across different regions and sectors.
Global and Regional Economic Losses
Table 1: Documented Economic Impacts of Selected Zoonotic Disease Outbreaks
| Country/Region | Disease(s) | Economic Impact | Context |
|---|---|---|---|
| Global | Soil-transmitted helminths (STH) | Affects ~1.5 billion people [121] | Prevalence data indicating scale of burden |
| Global | Lymphatic filariasis (LF) | 657 million in at-risk areas [121] | Potential for chronic disability and economic loss |
| India | Plague (1994) | USD 600 million - USD 2 billion [122] | Outbreak-related economic losses |
| Multiple Asian Countries | Highly Pathogenic Avian Influenza (2004-2009) | USD 10 billion [122] | Agricultural and control costs |
| Malaysia | Nipah virus encephalitis (1998-1999) | USD 617 million [122] | Outbreak impact on economy |
| China, Taiwan, Hong Kong, Singapore | Severe Acute Respiratory Syndrome (SARS) | USD 13 billion [122] | Economic impact of major outbreak |
The economic burden falls disproportionately on livestock farmers, especially in developing countries where zoonotic outbreaks can reduce market opportunities, increase veterinary costs, and disrupt supply chains. In some areas, zoonotic disease outbreaks have reduced milk consumption by up to 64%, causing severe financial hardship for dairy farmers [122]. This demonstrates how zoonotic diseases create ripple effects beyond direct health impacts, threatening food security and economic stability in vulnerable communities.
Productivity and Healthcare Costs
The economic burden of specific protozoal diseases manifests through multiple pathways:
- Giardiasis affects approximately 200 million people annually, causing productivity losses and healthcare expenditures, though it causes diarrheal diseases less frequently compared to cryptosporidiosis [18] [48].
- Amoebiasis caused by Entamoeba histolytica results in 40,000-100,000 deaths annually, ranking second to malaria in parasitic disease morbidity and mortality [48].
- Cryptosporidiosis particularly affects children under five and immunocompromised individuals, contributing significantly to global diarrheal disease burden with approximately 58 million cases annually attributed to intestinal protozoa [18] [48].
The true economic burden is likely underestimated due to underreporting, inadequate surveillance systems, and the chronic nature of many protozoal infections that may not cause acute outbreaks but nevertheless reduce productivity and quality of life [120] [40].
Experimental Methodologies for Detection and Analysis
Advanced diagnostic and research methodologies are crucial for understanding zoonotic protozoal transmission pathways and quantifying their burden. The following experimental protocols represent current best practices in the field.
Molecular Detection Using qPCR
Real-time Polymerase Chain Reaction (qPCR) has emerged as a superior method for detecting intestinal protozoa compared to traditional microscopy, offering higher sensitivity, specificity, and the ability to distinguish morphologically identical species [48].
Protocol: Duplex qPCR for Detection of Entamoeba Species and Cryptosporidium
Sample Preparation: Extract genomic DNA from approximately 200 mg of fecal samples using commercial stool DNA kits (e.g., TIANamp Stool DNA Kit). Store extracted DNA at -20°C until processing [48] [123].
Primer and Probe Design:
- For novel targets like Chilomastix mesnili, identify suitable primers and probes by retrieving partial sequences for the small ribosomal subunit from NCBI using BLASTN [48].
- Check for highly conserved regions and compare with the NCBI database to assess specificity.
- Select primers and probes with GC content of approximately 50%, length between 20-24 bases, and estimated melting temperature (Tₘ) of ~58°C [48].
Reaction Setup: Implement two duplex qPCR assays:
- Assay 1: Entamoeba dispar + Entamoeba histolytica
- Assay 2: Cryptosporidium spp. + Chilomastix mesnili
- Use 10 µL reaction volumes to reduce reagent costs [48].
Amplification Conditions:
Data Analysis: Use CFX Maestro or similar software for analysis. Validate PCR products by agarose gel electrophoresis. For species confirmation, purify positive products, clone into appropriate vectors, transform into competent cells, and sequence [123].
Epidemiological Network Analysis
Understanding transmission pathways requires sophisticated epidemiological approaches that integrate multiple data sources.
Protocol: Transmission Pathway Network Analysis [124]
Data Collection:
- Obtain survey data on social contacts, agricultural activities, and sociodemographic characteristics
- Collect household GPS coordinates for spatial analysis
- Gather fecal samples for pathogen detection using DNA metabarcoding
- Include domesticated animals in sampling to assess zoonotic potential
Laboratory Analysis:
- Screen human and animal fecal samples for protozoa using DNA metabarcoding
- Identify distinct subtypes through genetic analysis
- Record co-infections and subtype distributions
Network Construction:
- Create multiple networks reflecting different transmission pathways:
- Social networks (time spent together)
- Spatial networks (household proximity)
- Zoonotic networks (human-animal contact)
- Calculate network metrics and associations
- Create multiple networks reflecting different transmission pathways:
Statistical Analysis:
- Employ network autocorrelation models (NAMs)
- Use permutation tests (network k-test)
- Calculate odds ratios with 95% confidence intervals
- Perform model averaging to identify most informative predictors
Risk Factor Analysis
Identifying determinants of infection is crucial for targeted control strategies.
Protocol: Multivariate Risk Factor Analysis [123]
Data Collection:
- Record host factors: age, sex, neutering status, deworming history
- Document clinical symptoms: diarrhea, weight loss, vomiting, anorexia
- Note environmental factors: location, sanitation, animal contact
Laboratory Confirmation:
- Combine microscopy with PCR for maximum detection sensitivity
- Perform genotyping through sequence analysis
- Conduct phylogenetic analysis to identify zoonotic potential
Statistical Analysis:
- Use Fisher's exact test to compare infection rates across categorical variables
- Calculate odds ratios (OR) with 95% confidence intervals (CI)
- Set statistical significance at p < 0.05
- Employ two-tailed tests and address potential biases from small sample sizes
Research Reagent Solutions
Table 2: Essential Research Reagents and Their Applications
| Reagent/Kits | Specific Product Examples | Research Application | Technical Function |
|---|---|---|---|
| DNA Extraction Kits | TIANamp Stool DNA Kit (TIANGEN) [123] | Genomic DNA isolation from fecal samples | Efficient DNA purification from complex stool matrices |
| Cloning Vectors | pCE2 TA/Blunt-Zero Vector (5 min TA/Blunt-Zero Cloning Kit) [123] | PCR product cloning for sequencing | Rapid ligation and transformation of amplified products |
| Competent Cells | E. coli DH5α competent cells (Tolobio) [123] | Plasmid propagation | High-efficiency transformation for sequence verification |
| Gel Extraction Kits | FastPure Gel DNA Extraction Mini Kit (Vazyme) [123] | Purification of PCR products | Recovery of DNA fragments from agarose gels |
| qPCR Reagents | Custom primers/probes (Microsynth) [48] | Protozoan detection and differentiation | Target-specific amplification with fluorescence detection |
| Microscopy Reagents | Phosphate-buffered saline (PBS) [123] | Direct fecal smear preparation | Sample dilution and preservation of parasite morphology |
Discussion and Future Directions
The economic burden of zoonotic protozoal diseases extends far beyond direct healthcare costs, encompassing significant impacts on agricultural productivity, livelihoods, and economic development, particularly in LMICs. Research findings from northeast Madagascar demonstrate that proximity to infected households was the most significant predictor of Blastocystis infection (OR: 1.56, 95% CI: 1.33-1.82), while spending free time with infected participants was not informative (OR: 0.95, 95% CI: 0.82-1.10), suggesting environmental transmission pathways dominate over social ones [124]. This has important implications for control strategies, indicating that interventions should focus on environmental sanitation rather than social distancing measures.
The One Health approach, which integrates human, animal, and environmental health perspectives, is essential for addressing these complex challenges [120] [40]. Future research should prioritize:
- Enhanced Surveillance Systems: Integrating molecular diagnostics like qPCR with traditional surveillance to improve detection sensitivity and species differentiation [48].
- Economic Impact Studies: More comprehensive analyses of the true economic burden of protozoal diseases, including indirect costs and long-term impacts.
- Intervention Development: Targeted environmental interventions informed by transmission pathway analyses, such as improved water sanitation and animal management practices.
- Novel Therapeutic Approaches: Development of new treatment options, such as drug repurposing strategies, though recent research indicates emodepside showed no significant efficacy against protozoal infections [48].
The strategic application of emerging technologies such as genomics, artificial intelligence, and precision medicine can enhance diagnostic capacity, facilitate real-time data sharing, enable predictive modeling, and support evidence-based policy decisions [120]. Collaborative efforts among national and international partners, grounded in the One Health framework, are essential for achieving sustainable disease control and prevention, ultimately reducing the substantial economic burden of zoonotic protozoal diseases worldwide.
Conclusion
The zoonotic transmission of intestinal protozoa presents a complex challenge that necessitates a multifaceted, One Health approach. Foundational knowledge of diverse transmission pathways, from direct fecal-oral routes to mechanical vectors, is crucial. While advanced diagnostic and modeling tools offer powerful means for detection and prediction, significant hurdles remain, including drug resistance and diagnostic limitations. Future efforts must prioritize the development of novel therapeutics, the validation of integrated control strategies across human and animal populations, and the enhancement of real-time surveillance systems to effectively reduce the global burden of these diseases.
References
- 1. An Overview of Mucosa-Associated Protozoa: Challenges ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2022.860442/full]
- 2. Genotype‐Specific Small EVs Released by Giardia lamblia ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12399883/]
- 3. Cryo-EM structure of ribosome from pathogenic protozoa ... [https://www.nature.com/articles/s41467-025-62767-x]
- 4. Epidemiology and transmission patterns of Cryptosporidium ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12088920/]
- 5. A review of recent Cryptosporidium hominis and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12304688/]
- 6. Book Review: Amebiasis. Biology and Pathogenesis of ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2016.00819/full]
- 7. Advances in Molecular Biology of Entamoeba histolytica [https://www.mdpi.com/journal/microorganisms/special_issues/30684370S7]
- 8. Intestinal Protozoa at the Human, Animal and Environment ... [https://www.preprints.org/manuscript/202505.1812]
- 9. Prevalence of Nitroimidazole-Refractory Giardiasis ... [https://wwwnc.cdc.gov/eid/article/31/6/25-0076_article]
- 10. Recent advances in in vitro models for the study of ... [https://www.sciencedirect.com/science/article/pii/S2667114X25000299]
- 11. Severity of waterborne diseases in developing countries and ... [https://bnrc.springeropen.com/articles/10.1186/s42269-023-01088-9]
- 12. Ecoepidemiology and zoonotic risk of intestinal parasites in ... [https://www.nature.com/articles/s41598-025-13886-4]
- 13. Predicting and preventing the next viral disease transmitted ... [https://www.sciencedirect.com/science/article/pii/S0740002025000620]
- 14. Common and Potential Emerging Foodborne Viruses [https://www.mdpi.com/2075-1729/14/2/190]
- 15. Molecular Characterization of Zoonotic Intestinal Protozoa ... [https://pubmed.ncbi.nlm.nih.gov/41239015/]
- 16. Transmission of Viruses from Restroom Use: A Quantitative ... [https://link.springer.com/article/10.1007/s12560-023-09580-1]
- 17. Persistence of Infectivity of Different Enteroviruses on a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388917/]
- 18. Epidemiological Characteristics of Intestinal Protozoal ... [https://www.researchprotocols.org/2025/1/e66350]
- 19. Extracellular vesicles in intestinal protozoa: hidden mediators ... [https://gutpathogens.biomedcentral.com/articles/10.1186/s13099-025-00747-8]
- 20. A Systematic Review of Zoonotic Enteric Parasites Carried ... [https://pubmed.ncbi.nlm.nih.gov/35056038/]
- 21. Prevalence and infestation dynamics of intestinal protozoa ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12533579/]
- 22. Mechanical Transmission of Human Protozoan Parasites ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC544177/]
- 23. Evaluating the role of synanthropic filth flies in the ... [https://bmcvetres.biomedcentral.com/articles/10.1186/s12917-025-04627-w]
- 24. Cockroaches as carriers of human intestinal parasites in ... [https://www.sciencedirect.com/science/article/abs/pii/S0035920308002265]
- 25. Cockroaches as carriers of human medically important ... [https://link.springer.com/article/10.1007/s00436-024-08146-9]
- 26. Waterborne Transmission Driving the Prevalence of ... [https://www.mdpi.com/2076-2607/13/7/1549]
- 27. Prevalence of Intestinal Protozoan Parasites and Associated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8639259/]
- 28. Environmental transmission of Toxoplasma gondii: Oocysts in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7033973/]
- 29. Zoonotic enteric protozoa [https://www.sciencedirect.com/science/article/abs/pii/S0304401711004857]
- 30. Prevalence of Protozoa Species in Drinking and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4348585/]
- 31. Detectability and Persistence of Cyclospora cayetanensis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12114183/]
- 32. Impact of Environmental Conditions on the Survival ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4087255/]
- 33. Seasonal Inactivation of Cryptosporidium parvum Oocysts ... [https://www.sciencedirect.com/science/article/pii/S0362028X25001693]
- 34. Response of soil protozoa to acid mine drainage in a ... [https://www.sciencedirect.com/science/article/abs/pii/S030438942101757X]
- 35. Transmission and Epidemiology of Zoonotic Protozoal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3553666/]
- 36. Molecular characteristics and zoonotic potential of enteric ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12595128/]
- 37. One Health in a globalized world: challenges and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12406317/]
- 38. Prevalence and infestation dynamics of intestinal protozoa ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2025.1635456/full]
- 39. Prevalence of Intestinal Protozoa Among Patients Living ... [https://www.mdpi.com/2414-6366/10/11/324]
- 40. Wild Warnings: The Rise of Animal-borne Diseases [https://advances.umn.edu/fall-2025/wild-warnings-the-rise-of-animal-borne-diseases/]
- 41. Emerging and reemerging transboundary and zoonotic ... [https://www.sciencedirect.com/science/article/pii/B9780443248122000038]
- 42. Diagnostic Methods of Common Intestinal Protozoa [https://pmc.ncbi.nlm.nih.gov/articles/PMC9606991/]
- 43. Molecular characteristics and zoonotic potential of enteric ... [https://www.sciencedirect.com/science/article/pii/S2405676625000435]
- 44. Molecular diagnosis of intestinal protozoa in young adults and ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0283824]
- 45. Molecular detection of intestinal protozoa and ... [https://www.sciencedirect.com/science/article/abs/pii/S0732889325003918]
- 46. A Deep Dive into 3 Revolutionary Molecular Diagnostic Tools [https://www.celnovte.com/solution/beyond-the-microscope-a-deep-dive-into-3-revolutionary-molecular-diagnostic-tools/]
- 47. Beyond Cultures: The Evolving Role of Molecular ... [https://www.mdpi.com/2077-0383/14/19/6886]
- 48. Implementation of real-time PCR assays for diagnosing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11978536/]
- 49. The impact of genetic diversity in protozoa on molecular ... [https://www.sciencedirect.com/science/article/abs/pii/S1471492210002345]
- 50. Diagnostic dilemma: application of real-time PCR assays for ... [https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-025-06730-1]
- 51. Pathogen quantitative efficacy of different spike-in internal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10714923/]
- 52. Molecular Methods for Pathogenic Bacteria Detection and ... [https://www.mdpi.com/2073-4441/13/24/3551]
- 53. A quantum mechanics-based framework for infectious ... [https://www.nature.com/articles/s41598-025-96817-7]
- 54. Mathematical Modeling of Regional Infectious Disease ... [https://www.mdpi.com/2079-3197/13/8/187]
- 55. Mathematical modelling of Toxoplasma gondii transmission [https://pmc.ncbi.nlm.nih.gov/articles/PMC7753131/]
- 56. Risk Evaluation of Pathogenic Intestinal Protozoa Infection ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2021.696568/full]
- 57. Intestinal Parasitic Infections in 2023 - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC10284646/]
- 58. A mathematical model for zoonotic transmission of malaria in ... [https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0008736]
- 59. Analysis and prediction of improved SEIR transmission ... [https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2023.1223039/full]
- 60. Modeling the effectiveness of One Health interventions ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10028197/]
- 61. From Wastewater to Epidemiological Insights: A Systematic ... [https://www.sciencedirect.com/science/article/pii/S0043135425018809]
- 62. the paradigm shift of spatial epidemiology in disease ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12558956/]
- 63. Top 6 Visualizations for Quantitative Data Analysis Methods [https://chartexpo.com/blog/quantitative-data-analysis-methods]
- 64. Drug Development to Protozoan Diseases - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3103878/]
- 65. Exploration of innovative drug repurposing strategies for ... [https://www.sciencedirect.com/science/article/pii/S2095177924001813]
- 66. Modern technologies and solutions to enhance ... [https://www.sciencedirect.com/science/article/pii/S2949704323000550]
- 67. STEM: An Open Source Tool for Disease Modeling - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6708268/]
- 68. STEM: An open source tool for disease modeling [https://research.ibm.com/publications/stem-an-open-source-tool-for-disease-modeling]
- 69. STEM Disease Models - Eclipsepedia [https://wiki.eclipse.org/STEM_Disease_Models]
- 70. Treatment-refractory giardiasis: challenges and solutions [https://pmc.ncbi.nlm.nih.gov/articles/PMC6207226/]
- 71. Risk factors for and management of metronidazole ... [https://www.sciencedirect.com/science/article/abs/pii/S1477893921001319]
- 72. Extracellular vesicles in intestinal protozoa [https://pmc.ncbi.nlm.nih.gov/articles/PMC12395738/]
- 73. Considerations for diagnostic COVID-19 tests [https://www.nature.com/articles/s41579-020-00461-z]
- 74. Factors that Influence the Reported Sensitivity of Rapid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8524138/]
- 75. Overview of Intestinal Protozoan and Microsporidia Infections [https://www.merckmanuals.com/professional/infectious-diseases/intestinal-protozoa-and-microsporidia/overview-of-intestinal-protozoan-and-microsporidia-infections]
- 76. Intestinal protozoa infections and associated factors among ... [https://www.sciencedirect.com/science/article/pii/S2405673125000017]
- 77. Quantification of Protozoa and Viruses from Small Water ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4515645/]
- 78. Enteric Protozoa in the Developed World: a Public Health ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3416492/]
- 79. Climate Change Contribution to the Emergence or Re ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5755797/]
- 80. Temperature, humidity may drive future transmission of ... [https://www.psu.edu/news/research/story/temperature-humidity-may-drive-future-transmission-parasitic-worm-infections]
- 81. The impact of climate change upon intestinal parasites in ... [https://www.sciencedirect.com/science/article/abs/pii/S0277379124002968]
- 82. Effect of sanitation and water treatment on intestinal ... [https://pubmed.ncbi.nlm.nih.gov/26404667/]
- 83. The association between the lack of safe drinking water and ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0237102]
- 84. Quantification of viable protozoan parasites on leafy ... [https://www.sciencedirect.com/science/article/abs/pii/S0740002021000812]
- 85. Exploration of innovative drug repurposing strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11786068/]
- 86. The potential of the antifungal nystatin to be repurposed ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1539629/full]
- 87. RepurposeDrugs: an interactive web-portal and predictive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11232279/]
- 88. Comparative Pathobiology of the Intestinal Protozoan ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6789772/]
- 89. Intestinal Protozoal Diseases [https://emedicine.medscape.com/article/999282-overview]
- 90. Therapeutic Potential of Natural Products as Innovative ... [https://www.mdpi.com/2673-6772/5/3/49]
- 91. Protozoa: Structure, Classification, Growth, and Development [https://www.ncbi.nlm.nih.gov/books/NBK8325/]
- 92. A Systematic Computational Drug Repurposing Approach ... [https://www.sciencedirect.com/science/article/pii/S3050787125000873]
- 93. A fused deep learning approach to transform drug ... [https://www.nature.com/articles/s42004-025-01712-x]
- 94. Advances in the Diagnosis of Intestinal Parasitic Infections ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2021.715406/full]
- 95. Detection of Intestinal Protozoa in the Clinical Laboratory - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3957779/]
- 96. Comparison of four rapid diagnostic tests, ELISA ... [https://www.sciencedirect.com/science/article/abs/pii/S0167701215000305]
- 97. Comparative analysis of commercial and "In-House ... [https://pubmed.ncbi.nlm.nih.gov/40751255/]
- 98. a prospective study on 3500 stool samples over 3 years [https://pubmed.ncbi.nlm.nih.gov/40162804/]
- 99. Implementation of real-time PCR assays for diagnosing ... [https://link.springer.com/article/10.1007/s00436-025-08483-3]
- 100. Comparative evaluation of real-time PCR and ELISA for the ... [https://www.nature.com/articles/s41598-024-54602-y]
- 101. Predictive Validation of an Influenza Spread Model | PLOS One [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0065459]
- 102. Validation of the Wild-type Influenza A Human Challenge ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4342672/]
- 103. Monitoring seasonal influenza epidemics by using internet ... [https://www.nature.com/articles/srep46469]
- 104. A Case Study of Measles in Lola Prefecture, Guinea - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5953353/]
- 105. Prediction of measles cases in US counties: A machine ... [https://www.sciencedirect.com/science/article/pii/S0264410X2400971X]
- 106. Modeling the Spread of Ebola - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4776269/]
- 107. Predictive Model for Estimating Annual Ebolavirus ... [https://wwwnc.cdc.gov/eid/article/31/4/24-1193_article]
- 108. Prevalence and risk factors of intestinal protozoal infections ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12425333/]
- 109. Prevalence and risk factors of intestinal protozoal infections ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0332218]
- 110. Identification of risk factors and high-risk areas for ... [https://www.nature.com/articles/s41598-025-11547-0]
- 111. Frequency of Intestinal Protozoa in Patients Receiving ... [https://www.turkiyeparazitolderg.org/articles/frequency-of-intestinal-protozoa-in-patients-receiving-treatment-at-van-special-physioactive-special-education-and-rehabilitation-center/doi/tpd.galenos.2025.69875]
- 112. Prevalence and molecular characterization of intestinal ... [https://www.sciencedirect.com/science/article/abs/pii/S0034528825000943]
- 113. Towards a global One Health index: a potential assessment ... [https://idpjournal.biomedcentral.com/articles/10.1186/s40249-022-00979-9]
- 114. Community-based surveillance of zoonotic parasites in a 'One ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5462647/]
- 115. Quantitative Outcomes of a One Health approach to Study ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6003973/]
- 116. High parasite prevalence driven by the human-animal ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2025.1536861/full]
- 117. a One Health study in an urban area in southern of Chile [https://pubmed.ncbi.nlm.nih.gov/40191077/]
- 118. Disentangling Social, Environmental, and Zoonotic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11495997/]
- 119. Molecular Detection of Intestinal Protozoans in an Urban ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7998591/]
- 120. Zoonotic Diseases in Low and Middle-Income Countries ... [https://www.sciencedirect.com/science/article/pii/S2950248925000823]
- 121. New combination of medicines to treat parasitic worm infections [https://www.ema.europa.eu/en/news/new-combination-medicines-treat-parasitic-worm-infections]
- 122. Economic burden of zoonotic and infectious diseases on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12083159/]
- 123. Frontiers | Gastrointestinal protozoa in pet cats from Anhui province... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2025.1522176/full]
- 124. Disentangling social, environmental, and zoonotic ... transmission [https://pubmed.ncbi.nlm.nih.gov/39287986/]