Thick Blood Smear Microscopy for Malaria Diagnosis: A Comprehensive Guide for Research and Clinical Application
This article provides a comprehensive analysis of thick blood smear (TBS) microscopy, the established gold standard for malaria diagnosis.
Thick Blood Smear Microscopy for Malaria Diagnosis: A Comprehensive Guide for Research and Clinical Application
Abstract
This article provides a comprehensive analysis of thick blood smear (TBS) microscopy, the established gold standard for malaria diagnosis. Tailored for researchers, scientists, and drug development professionals, it explores the foundational principles of TBS, detailed methodological protocols, and strategies for troubleshooting and optimization. It further examines the test's validation metrics and performance in comparison to alternative diagnostic modalities, including hematology thin smears, rapid diagnostic tests (RDTs), and molecular and AI-based methods. The content synthesizes current research and guidelines to serve as a critical resource for diagnostic refinement and the development of next-generation tools in malaria research and control.
The Gold Standard: Foundational Principles of Thick Blood Smear Microscopy
The Role of Thick Blood Smears as the Diagnostic Reference Standard
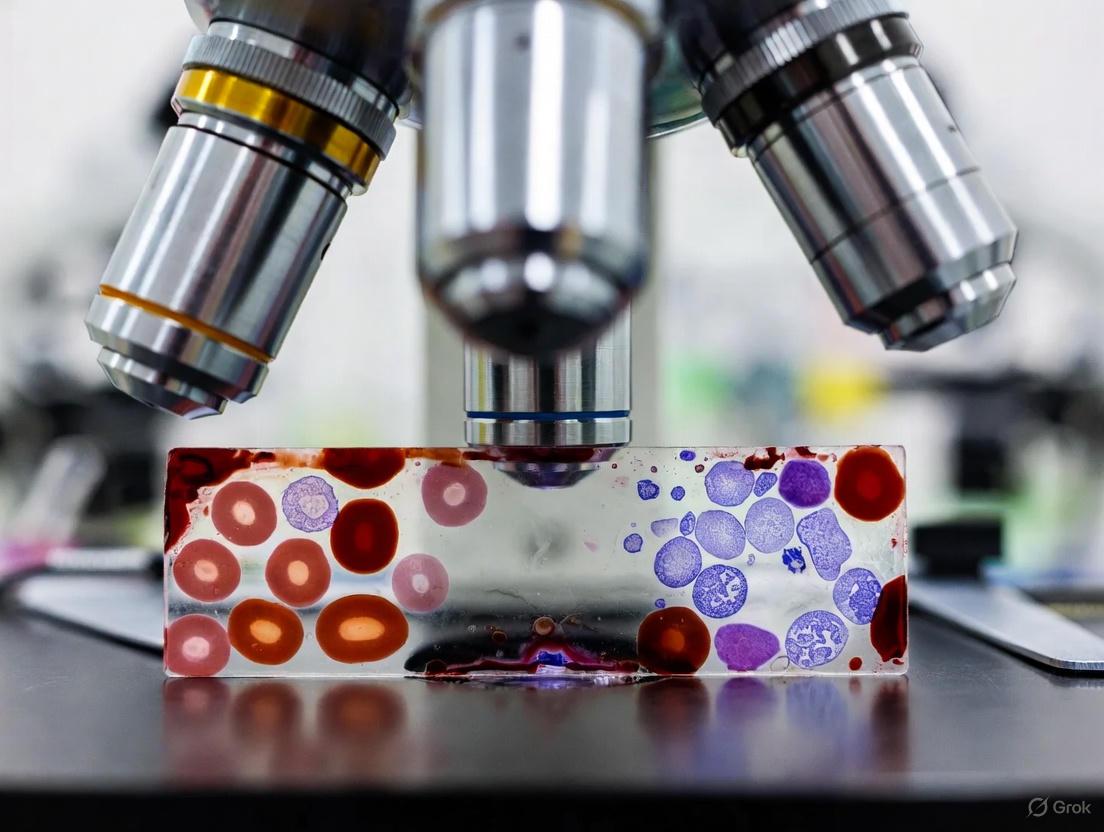
Within the framework of research on thick blood smear microscopy for malaria diagnosis, the thick blood smear maintains its status as the foundational reference standard for parasitological confirmation [1]. This method is critical for researchers and drug development professionals as it provides the benchmark against which new diagnostic technologies, such as rapid diagnostic tests (RDTs) and molecular assays, are validated [2]. Its unparalleled sensitivity in detecting low-level parasitemias is indispensable for clinical trials aiming to assess drug efficacy and for epidemiological studies monitoring transmission intensity in elimination settings [3] [1]. The thick smear's utility stems from the process of lysing red blood cells (RBCs) and concentrating the blood sample, allowing for the efficient scanning of a larger volume of blood—approximately 0.3 µL compared to 0.005 µL in a thin smear—which significantly enhances the probability of detecting parasites, especially in cases of low-level or early infections [4] [1].
Application Notes: Diagnostic Performance and Research Utility
Quantitative Diagnostic Performance of Thick Smears
The diagnostic performance of thick blood smear microscopy is well-documented, with its sensitivity making it the preferred initial test for malaria detection. The table below summarizes its key performance metrics and characteristics as established in the literature.
Table 1: Diagnostic Performance and Characteristics of Thick Blood Smears
| Parameter | Specification / Value | Context and Research Implication |
|---|---|---|
| Detection Sensitivity | 10-90 parasites/µL of blood [1] | Highly dependent on microscopist expertise; defines the lower limit of detection for field studies. |
| Concentration Factor | Approximately 30-fold compared to thin smears [4] | Enables examination of a larger blood volume, crucial for accurate parasite counts in vaccine trials. |
| Time to Results | 45-60 minutes (Giemsa staining) [1] | Slower than RDTs but provides quantifiable data; a key variable in workflow efficiency studies. |
| Parasite Density Calculation | Yes [1] | Essential for calculating parasitemia, a critical endpoint in antimalarial drug efficacy studies. |
| Species Identification | Limited reliability; requires thin smear confirmation [4] [1] | Fundamental limitation; necessitates paired thin smears for studies involving non-falciparum species. |
Comparative Analysis of Malaria Diagnostic Methods
For research and drug development, selecting the appropriate diagnostic tool is paramount. The following table provides a comparative overview of the primary methods available, positioning the thick smear within the modern diagnostic landscape.
Table 2: Comparison of Key Malaria Diagnostic Modalities for Research
| Method | Key Principle | Advantages for Research | Limitations for Research |
|---|---|---|---|
| Thick Blood Smear | Microscopic visualization of parasites in lysed RBCs. | Gold standard; quantifies parasitemia; low direct cost [1]. | Labor-intensive; subjective; requires expert training [3] [2]. |
| Thin Blood Smear | Microscopic visualization of parasites in a single RBC layer. | Enables species identification and parasite staging [3] [1]. | Lower sensitivity for detection; not suitable as a standalone primary test. |
| Rapid Diagnostic Tests (RDTs) | Immunochromatographic detection of parasite antigens. | High speed (minutes); ease of use; good sensitivity for P. falciparum [1]. | Cannot quantify parasites; false negatives possible with pfhrp2/3 gene deletions [1]. |
| Polymerase Chain Reaction (PCR) | Amplification of parasite nucleic acids. | High sensitivity (<10 parasites/µL); detects species and resistance markers [1]. | High cost; complex infrastructure; not for routine field diagnosis. |
| Advanced AI/Deep Learning | Automated image analysis and classification of smears. | High accuracy (>96%); potential for standardization and high-throughput screening [3] [2]. | Requires digitized smear images and computational resources; model generalizability can be a challenge [3]. |
Experimental Protocols
Detailed Protocol: Thick Blood Smear Preparation and Staining
This protocol is adapted from established CDC guidelines and is fundamental for ensuring reproducible and high-quality results in a research setting [4] [1].
A. Specimen Collection and Smear Preparation
- Collect venous blood into an EDTA tube. Note: Prepare smears as soon as possible (within 4 hours) to prevent morphological changes to the parasites [4] [1].
- Using a capillary tube or applicator stick, place a small drop of blood (approx. 10-15 µL) in the center of a pre-cleaned, labeled glass slide.
- Using the corner of a second slide, spread the drop in a circular pattern to achieve a uniform smear with a diameter of approximately 1.5 cm.
- Quality Control: A correctly prepared thick smear should be of a density that allows newsprint to be barely read through it when wet [4].
- Allow the smear to dry thoroughly. Drying must occur at room temperature (minimum 30 minutes, several hours recommended) or with the aid of a fan/cool hair dryer. Critical: Do not heat-fix thick smears [4].
B. Staining with Giemsa
- Briefly dip the dried thick smear in water to lyse any remaining hemoglobin. Do not use methanol or other fixatives [4].
- Place the slide in a Coplin jar containing 2.5% working Giemsa stain (pH 7.2) for 45-60 minutes. (A 10% Giemsa stain for 10 minutes is an alternative but yields lower quality) [1].
- Gently rinse the slide by immersing it in a jar of Giemsa buffer (pH 7.2) for a few moments.
- Place the slide upright in a rack to air-dry completely [1].
C. Microscopic Examination and Analysis
- Examine the stained smear under a 100x oil immersion objective.
- A minimum of 100 high-power fields should be examined before a slide is declared negative. For maximum sensitivity in research settings, 200-300 fields are recommended, especially for follow-up samples in drug trials [1].
- If parasites are detected, a corresponding thin smear must be examined for definitive species identification [4].
- Quantification: Parasite density can be calculated against white blood cells (WBCs) or RBCs. A common method is to count the number of parasites per 200 WBCs and, using an assumed WBC count (e.g., 8,000/µL), calculate parasites/µL [1].
Workflow Visualization: Thick Smear Diagnostic Pathway
The following diagram outlines the logical workflow for utilizing thick blood smears in a research and diagnostic context, highlighting key decision points and complementary techniques.
Diagram 1: Thick Smear Diagnostic Pathway
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents and materials required for the preparation, staining, and analysis of thick blood smears in a research environment.
Table 3: Essential Research Reagents and Materials for Thick Smear Microscopy
| Item | Specification / Function | Research Application Notes |
|---|---|---|
| Glass Microscope Slides | Pre-cleaned, frosted end for labeling. | Standard 75 x 25 mm size. Ensure they are grease-free to prevent smear detachment. |
| Giemsa Stain | Powder or liquid concentrate (e.g., Azure B, Eosin, Methylene Blue). | The metachromatic properties of Romanowsky-Giemsa stains allow differentiation of parasite nucleus (red/purple) and cytoplasm (blue) [1]. |
| Microscope | Binocular, with 10x, 40x, and 100x oil immersion objectives. | A quality microscope with a good light source is non-negotiable for accurate identification and counting. |
| Buffer Solution (pH 7.2) | Phosphate buffer. | Critical for correct staining; incorrect pH leads to poor color differentiation and misdiagnosis [1]. |
| Immersion Oil | Type A for light microscopy. | Essential for high-resolution imaging with the 100x objective. |
| EDTA Tubes | For venous blood collection and anticoagulation. | Prevents clotting; essential for standardized and delayed processing. |
| Capillary Tubes | For finger-prick capillary blood collection. | Standard for field studies and pediatric sampling. |
For over a century, the microscopic examination of Giemsa-stained thick blood smears has remained the cornerstone of malaria diagnosis in clinical and research settings. This technique continues to provide critical diagnostic information that informs both patient treatment and drug development research. Its enduring value lies in three fundamental advantages: exceptional analytical sensitivity for parasite detection, significant cost-effectiveness suitable for resource-limited settings, and the unique capability for direct parasite visualization that enables species identification and staging. Within the context of malaria drug development, thick smear microscopy serves as an indispensable tool for evaluating drug efficacy, monitoring parasite clearance kinetics, and detecting morphological changes indicative of drug pressure in both clinical trials and field studies. This protocol outlines standardized methodologies to leverage these advantages in research settings, ensuring reliable and reproducible data generation for antimalarial drug development.
Quantitative Advantage Analysis
The diagnostic performance and resource utilization of thick blood smear microscopy can be quantitatively compared against other common malaria diagnostic methods. The data in Table 1 highlight its strategic value for research and clinical applications.
Table 1: Comparative Analysis of Malaria Diagnostic Methods
| Diagnostic Method | Sensitivity (Parasites/μL) | Species Identification Capability | Relative Cost per Test | Infrastructure Requirements |
|---|---|---|---|---|
| Thick Blood Smear Microscopy | 10-90 [1] | Yes (with thin smear) [1] | Low [5] | Microscope, trained personnel [6] |
| Rapid Diagnostic Tests (RDTs) | ~100 [1] | Limited (HRP2/pLDH) [1] | Medium | Minimal |
| PCR Assay | 5-10 [1] | Excellent | High | Molecular biology lab |
| AI-Assisted Microscopy | Comparable to expert microscopy [7] | Yes (P. falciparum, P. vivax) [7] | Medium (initial hardware/software) [6] | Microscope, computer, AI model |
Experimental Protocols for Research-Grade Smear Analysis
Smear Preparation and Staining Protocol
This standardized protocol ensures optimal smear quality for reliable detection and quantification of malaria parasites.
Reagents and Materials:
- Microscope Slides: Clean, grease-free glass slides.
- Capillary Tubes: For finger-prick or venous blood collection.
- Giemsa Stain Stock Solution.
- Methanol (Absolute) for thin smear fixation.
- pH 7.2 Buffer Solution: Critical for correct staining contrast.
- Staining Jars (Coplin Jars).
Procedure:
- Thick Smear Preparation: Place 2-3 small drops of blood on a slide. Using the corner of another slide, spread the blood to form a uniform circle approximately 1 cm in diameter. The ideal thickness allows print to be read through the smear before staining. Air-dry the smear completely for 30-60 minutes at room temperature; do not apply heat [1].
- Thin Smear Preparation: Place a single small drop of blood near one end of a slide. Use a spreader slide to drag the blood across the surface to create a monolayer of cells. Air-dry completely [1].
- Fixation: Fix the thin smear only by immersing it in absolute methanol for 30-60 seconds. Do not fix the thick smear. Allow to dry [1].
- Staining: Immerse both thick and thin smears in a Coplin jar containing 2.5% Giemsa working solution (prepared in pH 7.2 buffer) for 45-60 minutes [1].
- Rinsing: Gently rinse the slides by dipping them 3-4 times in a jar of pH 7.2 buffer. Dry the slides upright in a rack [1].
Quality Control: Examine the staining quality under the microscope. White blood cell nuclei should be purple, and parasite cytoplasm blue. Red cells in the thin smear should appear pink. Poor staining requires destaining and restaining.
Microscopic Examination and Parasite Quantification
This protocol details the systematic examination of stained smears for accurate parasite detection and counting, a critical endpoint in drug efficacy trials.
Equipment:
- Light Microscope with 100x oil immersion objective.
- Immersion Oil.
Procedure:
- Start with the thick smear using the 100x oil immersion objective. Scan a minimum of 100 high-power fields before declaring a smear negative. The thick smear is used for initial detection and sensitivity assessment [1].
- If parasites are detected, switch to the thin smear for species identification and detailed staging based on parasite morphology.
- Parasite Density Calculation (Thick Smear): The parasite density can be estimated by counting the number of parasites per 200 white blood cells (WBCs) and multiplying by the patient's total WBC count. Alternatively, a standard WBC count of 8,000/μL is often assumed: Parasites/μL = (Number of Parasites Counted / Number of WBCs Counted) × 8,000.
- Parasitemia Calculation (Thin Smear): Examine the monolayer of red blood cells (RBCs) on the thin smear. Count the number of infected RBCs per at least 2,000 RBCs. Parasitemia (%) = (Number of Infected RBCs / Total RBCs Counted) × 100. This is crucial for assessing drug response in P. falciparum and P. knowlesi infections [1].
Data Recording: For research purposes, record species, stages present (ring, trophozoite, schizont, gametocyte), and parasite density/parasitemia for each time point (e.g., Day 0, and then every 6-12 hours to monitor parasite clearance).
Diagram 1: Blood smear analysis workflow for malaria diagnosis.
Advanced AI-Assisted Detection and Analysis
Recent advances in deep learning have augmented traditional microscopy, enhancing throughput and consistency for research applications.
Convolutional Neural Network (CNN) Protocol for Species Differentiation
Workflow:
- Image Acquisition: Capture high-resolution images of thick smear fields using a microscope coupled with a digital camera (e.g., 4K resolution) [5].
- Data Preprocessing: Extract individual Regions of Interest (ROIs) or cells from the larger microscope image. Apply preprocessing techniques such as feature enhancement and the Canny Algorithm to RGB channels to improve feature extraction [7].
- Model Training: Train a CNN model (e.g., a 7-channel input model) using a dataset of labeled cell images categorized as P. falciparum, P. vivax, or uninfected. Use a batch size of 256, 20 epochs, a learning rate of 0.0005, and the Adam optimizer [7].
- Validation: Employ K-fold cross-validation (e.g., 5 folds) to robustly assess the model's generalization performance and calculate metrics like accuracy, precision, and recall [7].
Performance: One such model achieved an accuracy of 99.51%, a precision of 99.26%, and a recall of 99.26% in differentiating P. falciparum, P. vivax, and uninfected cells [7].
Diagram 2: AI-assisted detection workflow for malaria parasite classification.
The Scientist's Toolkit: Research Reagent Solutions
Successful implementation of thick smear microscopy relies on specific reagents and tools. Table 2 details essential items for the research laboratory.
Table 2: Essential Research Reagents and Materials for Thick Smear Microscopy
| Item | Specification/Function | Research Application |
|---|---|---|
| Giemsa Stain | Romanowsky-type stain; stains parasite chromatin purple and cytoplasm blue. | Enables visualization and differentiation of parasite structures from host cell components. |
| pH 7.2 Buffer | Phosphate buffer for stain dilution. | Critical: Maintaining correct pH ensures consistent staining quality and diagnostic reliability. |
| Microscope with Camera | 100x oil immersion objective; 4K camera for image capture. | Essential for high-resolution imaging and for digitizing samples for AI analysis or remote expert consultation [5]. |
| AI/CNN Model | Pre-trained convolutional neural network for parasite detection/classification. | Provides a second-opinion tool for species identification, reduces inter-observer variability, and increases throughput in large studies [6] [7]. |
Light microscopy of Giemsa-stained thick and thin blood smears remains the cornerstone of parasitological confirmation for malaria and a critical tool in research and drug development [8]. This method allows for the detection of various Plasmodium species (P. falciparum, P. vivax, P. malariae, P. ovale, and P. knowlesi), identification of parasite stages, and quantification of parasite density, which is essential for monitoring response to treatment in clinical trials [8]. The accuracy of this diagnostic standard, however, is contingent upon a rigorous and standardized protocol from blood collection to staining. This application note provides a detailed methodology for preparing and staining blood smears, framed within the context of quality-assured microscopy for research settings.
Materials and Reagents
Research Reagent Solutions
The following table details the essential materials required for the blood smear preparation and staining protocol.
Table 1: Key Research Reagents and Materials for Blood Smear Microscopy
| Item | Function/Application | Specifications & Notes |
|---|---|---|
| Microscope Slides | Support for blood films. | Cleaned, degreased, and stored dust-free [9]. |
| Giemsa Stock Solution | Primary stain for visualizing malaria parasites. | Prepared from Giemsa powder; quality control is essential [9]. |
| Buffered Water (pH 7.2) | Diluent for Giemsa working solution. | Critical for correct Romanowsky staining; requires pH verification [9]. |
| Absolute Methanol | Fixative for thin blood films. | Ensures cellular adhesion and preservation before staining. |
| EDTA Blood Collection Tubes | Anticoagulant for venous blood samples. | Lavender-top (EDTA) tubes; ensures sample integrity [10]. |
| Microscope with Oil Immersion | Visualization of stained parasites. | 100x objective required for species identification and staging [8]. |
Experimental Protocols
Blood Collection and Smear Preparation
Principle: The accurate diagnosis of malaria relies on the quality of the blood smear. Both finger-prick and venous blood can be used, with the former being ideal for immediate smear preparation and the latter allowing for multiple tests [9] [10].
Methodology:
- Collection: For a finger-prick, clean the site and use a sterile lancet. Wipe away the first drop of blood. For venous blood, collect 3 mL into an EDTA tube via venipuncture [10].
- Thick Smear Preparation: Touch a drop of blood to a clean slide and, using a corner of another slide, spread it in a circular pattern to cover an area approximately 1.5 cm in diameter. The ideal thick smear is thick enough that newsprint is just visible through it. Do not fix thick smears with methanol [9].
- Thin Smear Preparation: Place a small drop of blood near one end of a second slide. Use a spreader slide at a 30-45° angle to draw the blood into a thin, uniform film. Immediately fix the thin smear by dipping it in absolute methanol for a few seconds once air-dried [9].
- Labeling and Drying: Clearly label slides with a unique identifier. Allow thick smears to dry thoroughly (至少 30 minutes in a humid environment); slow drying can cause lysis of red blood cells, which is necessary for the thick smear but must be controlled [9].
Giemsa Staining Protocol
Principle: Giemsa stain is a Romanowsky stain that binds to DNA and RNA, coloring the parasite chromatin red-purple and the cytoplasm blue, enabling differentiation from host blood cells [9].
Methodology:
- Prepare Giemsa Working Solution: Dilute Giemsa stock solution at a ratio of 1:10 to 1:20 with buffered water at pH 7.2. For example, add 3 mL of Giemsa stock to 57 mL of buffered water to make a 5% working solution [9].
- Stain the Smears: Place the air-dried, unfixed thick smear and the fixed thin smear on a staining rack. Flood the slides with the Giemsa working solution for 20-45 minutes. The longer staining time is typically used for thick smears to ensure adequate penetration [9].
- Rinse and Dry: Gently rinse the slides by immersing them in a container of buffered water (pH 7.2) or allowing a slow, thin stream of buffered water to flow over the slide surface. Avoid direct, forceful water jets. Stand the slides vertically to air-dry in a dust-free environment [9].
Table 2: Critical Staining Parameters and Timing
| Step | Parameter | Optimal Condition | Impact of Deviation |
|---|---|---|---|
| Staining Solution pH | pH of buffered water | 7.2 | Incorrect pH leads to poor chromatic differentiation; cytoplasm may not stain blue [9]. |
| Staining Duration | Time in Giemsa working solution | 20-45 minutes | Under-staining: faint parasite appearance. Over-staining: excessive background precipitate. |
| Sample Age | Time from collection to staining | Within 4 hours for whole blood (for optimal morphology) [10] | Anticoagulant affects parasite morphology and staining after 4 hours, complicating speciation [10]. |
Workflow Diagram
The following diagram illustrates the end-to-end workflow for malaria diagnosis via thick and thin blood smear microscopy.
Diagram 1: Blood Smear Microscopy Workflow
Quality Assurance and Advanced Considerations
For research and drug development, the integrity of malaria microscopy data is paramount. An acceptable microscopy service must be part of a comprehensive quality management system [8]. This includes regular training and competency assessment of microscopists, a slide validation system with feedback, clear standard operating procedures (SOPs), and good microscope maintenance [8]. Stained blood films should be examined under oil immersion (1000x magnification). The thick film is scanned for the presence of parasites, while the thin film is used for species confirmation and parasite staging based on morphological characteristics.
While microscopy is the traditional gold standard, researchers should be aware of its limitations, particularly in detecting low-level parasitaemia, as seen in asymptomatic or pregnant populations [11]. Molecular techniques like multiplex qPCR offer higher sensitivity and are increasingly used as a more definitive reference in clinical trials and epidemiological studies [11]. Furthermore, novel technologies such as digital holographic microscopy and advanced AI-based classification models are emerging as promising tools for automated, label-free parasite detection, potentially enhancing throughput and objectivity in research settings [12] [3].
Within the framework of malaria research, thick blood smear microscopy serves as a fundamental diagnostic tool. However, its utility extends far beyond a simple positive or negative result. For researchers and drug development professionals, two advanced applications—precise parasite speciation and accurate parasitemia quantification—are critical for understanding disease dynamics, assessing treatment efficacy, and developing new therapeutics. This document details advanced protocols and application notes for these applications, leveraging both classical microscopy enhancements and modern computational and molecular techniques.
Quantitative Parasitemia Analysis
The accurate quantification of parasites per unit volume of blood is a cornerstone for assessing disease severity and treatment response in clinical trials.
Advanced Microscopy for Low Parasitemia Enumeration
The New Whole Field (NWF) counting method significantly improves the accuracy of manual parasitemia quantification, especially for low-frequency target cells like Plasmodium vivax or reticulocytes [13].
Experimental Protocol: NWF Counting Method
- Sample Preparation: Prepare thin smears from patient blood. Fix with 100% methanol and stain with Giemsa (e.g., 20 minutes) [13].
- Microscopy Setup: Use a microscope fitted with a Miller reticle in the eyepiece. Verify the reticle's area ratio (e.g., 1:4, 1:5, 1:9) by capturing an image through the eyepiece and calculating the ratio using image analysis software like ImageJ to prevent a critical source of error [13].
- Counting Procedure:
- Systematically scan the slide, ensuring cell distribution is even and without clots.
- For each field of view:
- Count and record all target cells (e.g., parasitized RBCs) found within the entire circular field.
- Count the total number of all RBCs located only within the small square of the Miller reticle. Adhere to the edge rule: cells touching the top and left borders are counted in; cells touching the bottom and right borders are counted out [13].
- Calculation: Calculate the parasitemia using the following formula [13]:
Parasitemia (%) = (Number of all target cells in whole field / Total number of all cells in small square) × Reticle Factor (RF)The RF is predetermined by calculating the ratio of the area of the whole field to the area of the small square:RF = (Area of Whole Visual Field) / (Area of Small Reticle Square).
Table 1: Comparison of Manual Parasitemia Quantification Methods
| Feature | Conventional Miller Reticle | New Whole Field (NWF) Method |
|---|---|---|
| Counting Area for Target Cells | Large reticle square only | Entire microscopic field |
| Counting Area for Total RBCs | Small reticle square | Small reticle square |
| Key Advantage | Standardized counting area | Greatly increased numerator count, enhancing accuracy for low parasitemia |
| Time Efficiency | Standard | Reduced, as fewer fields need to be examined to achieve high accuracy |
| Inter-technician Discrepancy | Higher | Significantly lower [13] |
Molecular Quantification Techniques
Molecular methods provide superior sensitivity and precision for quantifying parasite density, especially at sub-microscopic levels.
Experimental Protocol: Droplet Digital PCR (ddPCR) for Absolute Quantification
ddPCR allows for absolute quantification of parasite density without the need for a standard curve, increasing comparability across laboratories [14].
- Sample & DNA Preparation: Extract genomic DNA from a known volume of patient whole blood (e.g., 200 μL) using a commercial kit (e.g., QIAamp DNA Blood Mini Kit) [15] [14].
- Reaction Setup:
- Prepare the ddPCR reaction mix containing primers and probes targeting the Plasmodium 18S rRNA gene [14].
- Partition the reaction mixture into approximately 15,000 nanoliter-sized droplets using a droplet generator.
- PCR Amplification: Perform end-point PCR amplification on the droplet emulsion.
- Data Analysis: Read the plate on a droplet reader. Count the number of fluorescence-positive droplets. The parasite density is calculated directly from the fraction of positive droplets using Poisson distribution statistics, providing an absolute count of DNA copies per microliter of blood [14].
Table 2: Comparison of Parasitemia Quantification Technologies
| Method | Mechanism | Sensitivity | Key Advantage | Key Disadvantage |
|---|---|---|---|---|
| Light Microscopy | Visual count of parasites on stained smear [16] | ~50-100 parasites/μL [14] | Low cost; provides species & stage info [16] | Labor-intensive; sensitivity depends on tech skill [17] |
| Quantitative PCR (qPCR) | Amplification of parasite DNA with a standard curve [15] | Higher than microscopy [15] | High sensitivity; good for high-throughput | Requires external standard; results vary between labs [14] |
| Droplet Digital PCR (ddPCR) | Partitioning & end-point counting of DNA molecules [14] | Higher than qPCR for P. falciparum [14] | Absolute quantification without standard curve; high reproducibility [14] | Higher cost; specialized equipment |
Advanced Parasite Speciation and Staging
Determining the Plasmodium species and the parasite's life cycle stage is vital for species-specific research and understanding pathogenesis.
Deep Learning for Automated Speciation and Staging
Convolutional Neural Networks (CNNs) and hybrid models can automate the classification of parasite species and life-cycle stages from blood smear images with high accuracy.
Experimental Protocol: CNN Model for Multiclass Species Identification
- Dataset Curation: Collect a large set of thick or thin blood smear images from clinical samples [7]. Annotate individual cells within regions of interest (ROIs) into categories: P. falciparum, P. vivax, uninfected white blood cells, etc. [7].
- Image Preprocessing: Apply techniques to enhance model performance, such as:
- Multi-channel input: Using seven-channel input tensors by enhancing RGB channels and applying algorithms like the Canny filter to extract richer features [7].
- Image Cropping & Resizing: For object detection models, crop high-resolution source images into smaller tiles and resize them to the model's required input dimensions (e.g., 416x416 pixels for YOLOv3), using padding to avoid distortion [18].
- Model Training & Validation:
- Split data into training, validation, and test sets (e.g., 80:10:10) [7].
- Train a CNN model using a residual network architecture to prevent vanishing gradients. Use an optimizer (e.g., Adam) and a loss function (e.g., cross-entropy) [7].
- Validate performance using k-fold cross-validation (e.g., 5 folds) to ensure robustness [7].
Table 3: Performance of a Deep Learning Model for Malaria Speciation
| Performance Metric | Reported Result |
|---|---|
| Accuracy | 99.51% [7] |
| Precision | 99.26% [7] |
| Recall | 99.26% [7] |
| Specificity | 99.63% [7] |
| F1-Score | 99.26% [7] |
The Hybrid Capsule Network for Interpretable Analysis
The Hybrid Capsule Network (Hybrid CapNet) architecture addresses limitations of standard CNNs by preserving spatial hierarchies, improving interpretability, and excelling at life-cycle stage classification [3].
Experimental Protocol: Life-Cycle Stage Classification with Hybrid CapNet
- Model Architecture: The model combines CNN layers for feature extraction with capsule layers that capture pose and deformation information, crucial for distinguishing subtle morphological differences between stages (ring, trophozoite, schizont, gametocyte) [3].
- Composite Loss Function: Employ a novel loss function integrating:
- Margin loss: For class existence.
- Focal loss: To handle class imbalance.
- Reconstruction loss: To encourage the model to retain all necessary information for reconstructing the input.
- Regression loss: For spatial localization of parasites [3].
- Model Output: The model provides classification of parasite species and life-cycle stage. Its interpretability is validated using Grad-CAM visualizations, which highlight the biologically relevant regions in the image the model used for decision-making [3].
Integrated Workflow for Research Applications
The following diagram illustrates a potential integrated workflow combining the described methodologies for a comprehensive research analysis.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Reagents and Materials
| Item | Function/Application | Example/Note |
|---|---|---|
| Giemsa Stain | Stains parasite chromatin and cytoplasm; allows for morphological differentiation of species and stages [19] [20] | Standard for blood smear staining; pH critical for quality [17] [19] |
| Miller Reticle | Calibrated eyepiece graticule for standardized manual cell counting on smears [13] | Verify area ratio (1:4, 1:5, etc.) to prevent calculation errors [13] |
| DNA Extraction Kit | Isolation of high-quality genomic DNA from whole blood for molecular assays [15] [14] | QIAamp DNA Blood Mini Kit [15] |
| 18S rRNA Primers/Probes | Target for qPCR/ddPCR assays; highly conserved for genus Plasmodium with species-specific regions [15] [14] | Enables sensitive detection and quantification [15] [14] |
| Fluorescent Dyes (Acridine Orange) | Nucleic acid staining for fluorescent microscopy methods (e.g., QBC technique) [17] [20] | Increases diagnostic sensitivity but not species-specific [17] |
| Annotated Image Datasets | Training and validation data for developing AI/ML models for parasite detection and classification [3] [7] | Publicly available datasets include MP-IDB, IML-Malaria [3] |
Mastering the Technique: A Step-by-Step Protocol for Optimal Smear Preparation and Analysis
Standardized Protocol for Thick Smear Preparation and Giemsa Staining
Within malaria research and drug development, the microscopic examination of Giemsa-stained thick blood smears remains the cornerstone technique for the definitive diagnosis of malaria. This protocol is engineered to deliver maximum sensitivity for parasite detection, enabling researchers to accurately identify and quantify Plasmodium species. The fundamental principle of the thick smear is the lysis of red blood cells during the staining process, which allows for the examination of a larger volume of blood concentrated into a small area. This results in a ~30-fold increase in analytical sensitivity compared to a thin smear, making it indispensable for detecting low-level parasitemias, a critical factor in clinical trials and epidemiological studies assessing drug efficacy [4]. While species identification is often confirmed via thin smear or molecular methods, the thick smear is the primary tool for determining parasite density and monitoring response to experimental therapeutic interventions [8] [21].
Materials and Equipment
Research Reagent Solutions
The following table details the essential materials required for the execution of this protocol.
Table 1: Essential Research Reagents and Materials
| Item | Function/Explanation in Research Context |
|---|---|
| Capillary Blood | Preferred specimen; collected via fingerstick to ensure optimal parasite morphology and staining characteristics [4]. |
| Microscope Slides | Pre-cleaned, high-quality slides with frosted ends for labeling are essential for consistent sample processing and data tracking. |
| Giemsa Stain | The recommended Romanowsky-type stain; differentiates nuclear (purple) and cytoplasmic (blue) material of parasites, allowing for staging and speciation [8]. |
| Buffered Water (pH 7.2) | Critical for accurate stain performance; incorrect pH leads to poor color contrast and potential misidentification of parasites [22]. |
| Absolute Methanol | Used for fixing thin smears only; thick smears must not be fixed to allow for dehemoglobinization [4]. |
| Microscope (Oil Immersion) | Equipped with 10x, 40x, and 100x oil immersion objectives for scanning and detailed parasite analysis. |
Specimen Collection and Handling
Venous blood collected in EDTA is an acceptable alternative to capillary blood. However, smears must be prepared as soon as possible after collection (ideally within one hour) to avoid deteriorative changes in parasite morphology and staining characteristics that could compromise research data [4]. The use of fresh blood is paramount for superior cell morphology and minimal artifact introduction [22].
Step-by-Step Experimental Protocol
The following diagram outlines the complete workflow from specimen collection to microscopic analysis.
Detailed Procedural Steps
Thick Smear Preparation
- Slide Labeling: Label a pre-cleaned microscope slide with a unique patient or sample identifier using a graphite pencil or ethanol-resistant pen.
- Blood Application: For each sample, place a single small drop of blood (approximately 4-6 µL) in the center of the slide [21].
- Smear Spreading: Using the corner of a separate slide or an applicator stick, spread the drop in a circular pattern to achieve a uniform smear with a diameter of approximately 1.5 cm² (the size of a dime).
- Quality Check: A correctly prepared thick smear should be of a density that, when placed wet over newsprint, allows the text to be barely legible [4].
Drying: Lay the slides flat on a slide tray and allow them to dry thoroughly at room temperature. This requires a minimum of 30 minutes, but several hours is preferable to prevent the smear from detaching during staining. Protect the slides from dust, insects, and excessive heat. Do not heat-fix or chemically fix the thick smear [4].
Alternative Scratch Method for Improved Adherence: An alternate method involves using the edge of a glass slide to create small scratches in the underlying slide while spreading the blood. This enhances adherence, allowing for staining within 20-30 minutes of preparation, thereby accelerating research throughput [4].
Giemsa Staining Protocol
Staining must be performed with standardized timing and protocols to ensure consistent, reproducible results across experiments [22].
Table 2: Giemsa Staining Parameters
| Parameter | Specification | Rationale |
|---|---|---|
| Stain Type | Giemsa azure B type stain [8] | Provides definitive staining of parasite chromatin and cytoplasm. |
| Working Solution | Freshly diluted, 2-10% in buffered water [8] | Prevents stain precipitation and ensures optimal staining. |
| Buffer pH | 7.2 (using phosphate buffer) [8] | Critical for correct Romanowsky staining effect. |
| Staining Duration | 20-45 minutes [8] | Allows for adequate penetration and differentiation. |
| Rinse Solution | Buffered or tap water (pH 7.2) | Removes excess stain without causing precipitate. |
The detailed staining procedure is as follows:
- Prepare Stain: Dilute Giemsa stock solution to the desired concentration (e.g., 3%) using pH 7.2 buffered water.
- Stain Application: Place the thoroughly dried thick smear horizontally on a staining rack. Flood the slide with the diluted Giemsa stain, ensuring the entire thick smear is covered.
- Incubation: Allow the stain to act for the predetermined time (e.g., 30 minutes).
- Rinsing: After staining, gently pour off the stain and gently rinse the slide by applying a slow, steady stream of buffered water (pH 7.2) to the slide, allowing it to flow over the smear. Avoid directing a forceful stream directly onto the smear, as this can dislodge it.
- Drying: Stand the slide on end (vertically) in a draining rack and allow it to air-dry completely in a dust-free environment [4] [22].
Quality Assurance and Data Analysis
Microscopic Examination and Parasite Quantification
- Systematic Examination: Begin with a low-power (10x) scan of the smear to assess quality and locate the optimal monolayer for examination. Switch to the 100x oil immersion objective for detailed analysis [23].
- Parasite Counting: The gold standard method for quantifying parasitemia involves counting the number of asexual parasites against a set number of white blood cells (WBCs) or red blood cells (RBCs). The WHO method is recommended [21].
- Count the number of parasites (asexual and sexual) per 200 WBCs (or per 500 WBCs for low parasitemia).
- Calculate the parasite density/µL using the formula: Parasites/µL = (Number of parasites counted / Number of WBCs counted) × Total WBC count/µL
- If an actual WBC count is unavailable, a standard value of 8,000 WBCs/µL is often used for estimation [21].
Critical Quality Control Measures
A robust quality management system is non-negotiable for research integrity. Key requirements include [8]:
- Central Coordination: Oversight of the quality assurance program.
- Training and Competency: Regular, competency-based training and assessment of microscopists, supported by a validated reference slide bank.
- Slide Validation: A sustainable system for re-checking a percentage of slides with feedback and corrective actions for inadequate performance.
- Reagent Management: Proper storage and regular replacement of stains and buffers to prevent degradation and ensure consistent staining quality [22].
- Standard Operating Procedures (SOPs): Clear, written SOPs for all steps, from smear preparation to microscopy, must be available and adhered to at all levels [8].
Light microscopy of thick and thin blood smears remains the cornerstone of parasitological confirmation for malaria and the diagnostic standard against which other methods are compared [8]. It is the only method that allows for the detection of all five human-infecting Plasmodium species, differentiation of their various life-cycle stages, and quantification of parasite density to monitor response to treatment [8] [3]. This protocol details the procedures for screening and confirming parasite presence via thick blood smear microscopy, providing researchers and clinicians with a standardized methodology essential for reliable malaria diagnosis within a research context focused on improving diagnostic accuracy.
Materials and Reagents
Research Reagent Solutions
The following reagents and materials are essential for preparing and examining blood smears for malaria diagnosis.
| Item | Function/Explanation |
|---|---|
| Giemsa Stain | The recommended Romanowsky-type stain for malaria microscopy. It differentially stains parasite chromatin (red-purple) and cytoplasm (blue), providing critical contrast for identifying parasites within red blood cells [8]. |
| Microscope | A standard light microscope with 100x oil immersion objective is mandatory for visualizing malaria parasites. Accuracy is heavily dependent on microscope quality and proper maintenance [8]. |
| Microscope Slides | Clean, grease-free glass slides for preparing thin and thick blood films [8]. |
| Immersion Oil | Required for use with the 100x oil immersion objective to achieve the high resolution necessary to see detailed parasite morphology. |
| pH 7.2 Buffer Solution | Used to dilute Giemsa stain. The pH is critical for correct staining characteristics; a pH that is too high or too low will lead to poor staining and potential misdiagnosis [8]. |
Experimental Protocol: Thick Smear Microscopy Workflow
Procedure
Blood Smear Preparation:
- Place a small drop of capillary or venous blood (collected with appropriate ethical consent) onto a clean microscope slide.
- Using the corner of a second slide, spread the drop in a circular pattern to create a thick film with a diameter of approximately 1 cm. The correct thickness allows one to barely read newsprint through it before it dries.
- Allow the thick smear to air dry completely without fixation. Do not apply heat.
Staining (Giemsa Stain):
- Prepare a 10% Giemsa working solution by diluting the stock Giemsa stain in pH 7.2 buffer solution.
- Flood the air-dried thick smear with the 10% Giemsa stain for 20-30 minutes.
- Gently rinse the stained slide by immersing it in a container of pH 7.2 buffer or slow-running tap water to remove excess stain.
- Place the slide vertically on a drying rack to air dry completely.
Microscopic Examination:
- Place the dried smear on the microscope stage.
- Begin examination with the 10x or 40x objective to locate a well-stained area of the smear where the blood cells are evenly distributed and not overlapping excessively.
- Apply a drop of immersion oil directly onto the smear and carefully switch to the 100x oil immersion objective for detailed observation.
- Systematically scan the smear. A minimum of 100 microscopic fields should be examined before declaring a sample negative for parasites.
Parasite Detection and Quantification:
- Identify parasites based on their staining characteristics and morphology.
- For parasite quantification, the number of parasites is typically counted against a specified number of white blood cells (e.g., parasites/200 WBCs) or against a volume of blood [3].
Workflow Visualization
The following diagram illustrates the logical workflow for the microscopic examination of thick blood smears for malaria diagnosis.
Performance Data and Diagnostic Context
Comparative Diagnostic Performance
The table below summarizes the performance of microscopy in recent studies, providing a quantitative context for its diagnostic capabilities, especially when compared to Rapid Diagnostic Tests (RDTs) and molecular methods.
| Diagnostic Method | Sensitivity | Specificity | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Light Microscopy | 48.14% - 97.48%* [25] | Varies with quality assurance [8] | Gold standard; identifies species & stages; quantifies parasitemia [8] | Labor-intensive; requires expert skill; sensitivity variable [3] |
| Rapid Diagnostic Tests (RDTs) | 81.48% - 93.37% (for P. falciparum) [25] | 68.50% [25] | Rapid, easy to use, minimal infrastructure [24] | Cannot quantify; species limitations; false negatives (hrp2/3 deletions) [26] |
| Quantitative PCR (qPCR) | Highest (Reference) [25] | Highest (Reference) [25] | Detects low parasitemia; definitive species identification [25] | Expensive; complex; not for point-of-care [25] |
*The wide sensitivity range for microscopy (48.14% vs. 97.48%) highlights its dependency on technician expertise. The lower value (48.14%) comes from a direct comparison with a more sensitive qPCR test, while the higher value (97.48%) represents the proportion of detected infections that were P. falciparum in a setting where it is predominant [25].
Quality Assurance in Research Settings
A functioning quality assurance (QA) system is critical for generating reliable research data. Key components of an effective QA program for malaria microscopy include [8]:
- Standard Operating Procedures (SOPs): Clear, written protocols for all steps from smear preparation to result reporting.
- Training and Competency Assessment: Initial comprehensive training based on competency and regular re-assessment of skills [8].
- Slide Validation: A system for re-checking a random sample of slides, plus all positive and a subset of negative slides, by a senior microscopist.
- Reference Slide Bank: Access to a validated set of reference slides for ongoing training and proficiency testing [8].
Adherence to the detailed protocols for thick smear preparation, staining, and systematic examination outlined in this document is fundamental for accurate screening and confirmation of malaria parasites. The reliability of microscopy as a research tool is highly dependent on rigorous training and a robust quality assurance program. While microscopy remains the foundational method for parasite detection and species identification, researchers must be aware of its performance characteristics and variability compared to other diagnostic modalities like RDTs and molecular methods. A standardized approach ensures the generation of high-quality, reproducible data crucial for advancing malaria diagnostics and drug development.
Within malaria research and drug development, the accurate quantification of parasitemia—the percentage of red blood cells (RBCs) infected with Plasmodium parasites—is a critical metric. It is indispensable for assessing infection severity, monitoring patient response to treatment, and evaluating the efficacy of novel antimalarial compounds in development [27] [1]. While molecular techniques are emerging, light microscopy of Giemsa-stained thick and thin blood smears remains the widely accessible gold standard for parasite identification and quantification [27]. This protocol details the essential methodologies for calculating percent parasitemia, framed within the context of thick blood smear microscopy for malaria diagnosis research.
Core Principles of Parasitemia Quantification
The calculation of percent parasitemia is fundamentally a ratio of infected red blood cells to total red blood cells, expressed as a percentage. The formula for this calculation is:
% Parasitemia = (Number of Parasitized RBCs / Total Number of RBCs Counted) × 100 [20]
This quantitative data provides researchers with a direct measure of parasite burden in the bloodstream. Two primary microscopic approaches are employed, each with distinct advantages and applications in the research setting. The choice between them depends on the required balance of speed and precision.
Table 1: Comparison of Primary Microscopic Quantification Methods
| Method | Procedure | Application | Key Assumptions |
|---|---|---|---|
| Thin Smear (RBC Ratio) [20] | Count parasitized RBCs among 500 to 2,000 RBCs on a thin smear. | Preferred for precise quantification and species identification. | None for the ratio itself; conversion to parasites/μL requires an assumed total RBC count (e.g., 4-5 million/μL). |
| Thick Smear (WBC Reference) [20] [28] | Tally parasites against White Blood Cells (WBCs) until 500 parasites or 1,000 WBCs are counted. | Used for screening and rapid estimation, especially at low parasitemia. | Assumes a standard WBC count (e.g., 8,000/μL) to convert the parasite/WBC ratio to parasites/μL. |
Detailed Experimental Protocols
Protocol 1: Quantification via Thin Blood Smear
This method is recommended for obtaining precise percent parasitemia and for definitive species identification, which is crucial in drug development studies where the parasite species can influence compound efficacy [29] [30].
- Smear Preparation: Create a thin blood film on a clean, grease-free glass slide. The film should have a "feathered edge" where RBCs form a monolayer, free of overlap [27] [30].
- Fixation and Staining: Fix the thin smear by briefly immersing it in absolute methanol. Allow it to air-dry completely before staining with Giemsa stain (e.g., 2.5% for 45-60 minutes) [1] [30].
- Microscopic Examination: Examine the smear using a 100x oil immersion objective. Focus on the feathered edge where RBCs are undistorted and in a single layer [1].
- Systematic Counting:
- Select a region of the monolayer with well-stained and well-distributed RBCs.
- Systematically count the number of parasitized RBCs and the total number of RBCs in contiguous fields of view.
- The number of RBCs to count depends on the parasitemia level [20] [30]:
- It is critical to count asexual blood stage parasites and gametocytes separately, as only the former are clinically relevant for assessing active disease and response to most drug therapies [20].
- Calculation: Apply the standard formula using the collected counts.
Protocol 2: Quantification via Thick Blood Smear
The thick smear is used for rapid screening and estimation of parasite density, as it allows for the examination of a larger volume of blood, thereby increasing sensitivity for detecting low-level infections [20] [1].
- Smear Preparation: Place a large drop of blood (approximately 6-20 µL) on a slide and spread it in a circle of 1-2 cm diameter. Do not fix the smear before staining [27] [1].
- Staining: Stain the unfixed thick smear directly with Giemsa. The dehemoglobinization process occurs during staining, leaving the parasites concentrated and visible [1].
- Microscopic Examination: Screen the entire smear at low magnification (10x or 20x objective) to detect large parasites. Then, examine using the 100x oil immersion objective [20].
- Parasite and WBC Tally:
- Tally the number of parasites against the number of WBCs in each field of view.
- Continue counting until you have counted 500 parasites or 1,000 WBCs, whichever comes first [20].
- Calculation:
- If the patient's WBC count is known, use the following formula: Parasites/µL blood = (Parasites Tallied / WBCs Tallied) × Patient's WBC count/µL
- If the WBC count is unknown, the World Health Organization (WHO) recommends assuming 8,000 WBCs/µL of blood [20] [28]: Parasites/µL blood = (Parasites Tallied / WBCs Tallied) × 8,000
Advanced & Alternative Methodologies
While microscopy is the cornerstone, researchers should be aware of advanced and alternative methods that offer varying degrees of precision, sensitivity, and throughput.
Table 2: Advanced and Alternative Quantification Methods
| Method | Principle | Research Application | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Flow Cytometry [31] | Fluorescent staining of parasite DNA (e.g., with PicoGreen) and automated cell analysis. | High-throughput drug screening; growth assays of low-parasitemia field isolates. | Superior speed and reproducibility; low intra- and inter-observer variability. | Requires specialized, expensive equipment; complex sample preparation. |
| Fluorescent Microscopy [20] [32] | Staining with nucleic acid-binding dyes (e.g., Acridine Orange, SYBR Green I) and examination under fluorescence. | Automated parasitemia quantification; research in low-resource settings. | Enhanced signal-to-noise ratio; facilitates automated image analysis. | Requires a fluorescence microscope; dyes may be non-specific. |
| Quantitative Buffy Coat (QBC) [20] | Centrifugation of blood in acridine orange-coated capillary tubes, concentrating parasites. | Rapid diagnostic screening in field research. | Parasite concentration improves detection sensitivity. | Limited utility for species identification and precise quantification. |
| Molecular Methods (qPCR) [31] [33] | Amplification and detection of parasite-specific DNA sequences (e.g., 18S rRNA, varATS). | Gold standard for sensitivity in clinical trials; detecting submicroscopic infections; evaluating drug efficacy. | Extremely high sensitivity (can detect <5 parasites/µL); excellent for detecting mixed infections. | High cost and complexity; not suitable for rapid clinical diagnosis or quantification in high parasitemia. |
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful quantification of parasitemia relies on a suite of specific reagents and materials. The following table details key solutions and their functions in the experimental workflow.
Table 3: Essential Research Reagents and Materials
| Research Reagent / Material | Function / Application in Parasitemia Quantification |
|---|---|
| Giemsa Stain | The standard Romanowsky-type stain used to differentiate parasite chromatin (red-purple) and cytoplasm (blue) within red blood cells, enabling identification and counting [1] [30]. |
| EDTA (Lavender Top) Tubes | Preferred anticoagulant for blood collection; prevents clotting while best preserving parasite morphology for several hours, though smears should be made ASAP [27] [30]. |
| Acridine Orange / SYBR Green I | Fluorescent nucleic acid dyes used in flow cytometry and fluorescent microscopy protocols to stain parasite DNA/RNA, allowing for automated detection and counting [20] [31] [32]. |
| PicoGreen dsDNA Dye | A highly sensitive fluorescent dye specifically binding double-stranded DNA, used in flow cytometric methods to maximize precision for low-parasitemia samples and drug assays [31]. |
| RNase Solution | Used in flow cytometry protocols to digest RNA, reducing background noise and enhancing the signal-to-noise ratio of DNA-specific staining for more precise parasitemia determination [31]. |
The accurate calculation of percent parasitemia is a foundational technique in malaria research. The method of choice—whether the precise thin smear count, the sensitive thick smear estimate, or a more advanced molecular or flow cytometry approach—must be aligned with the specific research objectives, available resources, and required level of sensitivity. Adherence to standardized protocols for smear preparation, staining, and systematic counting is paramount for generating reliable, reproducible data that can drive drug development and deepen our understanding of malaria pathogenesis.
Critical Role in Species Identification and Mixed Infection Detection
Within the framework of malaria diagnosis research, thick blood smear microscopy remains a cornerstone technique, particularly for its capacity to identify Plasmodium species and detect mixed infections. In endemic regions, co-infections with multiple Plasmodium species are not uncommon, making accurate speciation a critical determinant for administering effective, species-specific drug regimens and for conducting robust epidemiological surveillance [17]. Although molecular techniques offer high sensitivity, light microscopy of thick and thin blood smears persists as the "gold standard" method, recommended by the World Health Organization (WHO) for point-of-care diagnosis and speciation [17]. This protocol outlines the detailed application of thick smear microscopy for these specific, high-value diagnostic applications, contextualizing its role within a broader research environment that may include complementary molecular assays.
Performance Characteristics and Quantitative Data
The utility of thick blood smears for detection and thin smears for definitive species identification is well-established. The table below summarizes the comparative performance and specifications of these techniques.
Table 1: Performance Characteristics of Blood Smear Microscopy for Malaria Diagnosis
| Parameter | Thick Smear | Thin Smear |
|---|---|---|
| Primary Application | Initial screening for parasite detection; sensitive for mixed infections [20] | Species identification; parasite morphology assessment [17] |
| Sensitivity (Theoretical) | ~4 parasites/μL (after examining 100 fields with ~20 WBCs/field) [20] | Lower than thick smear due to smaller blood volume examined [17] |
| Sensitivity (Practical, vs. PCR) | 81% at >10,000 parasites/mL; 29% at 1,000-10,000 parasites/mL [34] | Not typically used for sensitivity assessment at low parasitemia [34] |
| Parasite Density Estimation | Tally parasites against WBCs; report as parasites/μL [20] | Count parasitized RBCs among 500-2,000 RBCs; report as % parasitemia [20] |
| Time to Result | Minutes to hours, post-staining | Minutes to hours, post-staining |
| Key Limitation | Underestimates parasite density; parasites may be lost during staining [34] | Less sensitive for initial detection, especially at low parasitemia [17] |
Detailed Experimental Protocol for Thick Smear Analysis
Specimen Collection and Smear Preparation
- Specimen Type: Collect 3-5 mL of venous blood into an EDTA (lavender-top) tube. If venous blood is unavailable, a finger-prick capillary blood sample is acceptable [10].
- Smear Preparation:
- Place a small drop (approx. 10-20 μL) of well-mixed blood onto a clean microscope slide.
- Using the corner of another slide, spread the drop in a circular pattern to create a uniform smear approximately 1-2 cm in diameter. The ideal thickness allows newsprint to be barely legible through the smear.
- Handling and Stability: EDTA blood samples for smear microscopy should be processed within 4 hours of collection for reliable species identification, as parasite morphology degrades with prolonged exposure to anticoagulant [10]. Prepare smears immediately and air-dry them completely before staining.
Staining Procedures
- Giemsa Staining (Recommended for species identification):
- Rapid Staining (Field's Stain):
- Dip the air-dried slide in Field's Stain A (methylene blue) for 3 seconds.
- Rinse by gently dipping in tap water.
- Dip the slide in Field's Stain B (eosin) for 3 seconds.
- Rinse gently in tap water and air-dry completely [34].
Microscopic Examination and Species Identification Workflow
The following diagram outlines the systematic process for examining blood smears, from initial screening to final diagnosis and reporting.
Diagram 1: Microscopic Examination Workflow
- Step 1: Low Magnification Screening: Screen the entire thick smear at low power (10x or 20x objective) to detect large parasites and identify areas that are well-stained and free of debris [20].
- Step 2: High-Power Examination:
- Switch to the 100x oil immersion objective.
- Systematically examine a minimum of 100 microscopic fields, each containing approximately 20 White Blood Cells (WBCs), before declaring a smear negative. For higher sensitivity in non-immune patients, screening 200-300 fields is recommended [20].
- The presence of any parasites indicates a positive diagnosis.
- Step 3: Species Identification and Mixed Infection Detection:
- Tentative Identification on Thick Smear: Observe parasite morphology. While challenging, experienced microscopists can differentiate species on thick smears based on size, chromatin dot appearance, and cytoplasmic staining [17].
- Definitive Identification via Thin Smear: If parasites are detected, the corresponding thin smear must be examined for conclusive species identification. Key discriminators include:
- Infected Red Blood Cell (RBC) morphology: Size and shape.
- Parasite stages present.
- Schizont and gametocyte morphology, which are species-specific.
- Detecting Mixed Infections: Carefully scrutinize the thin smear for the presence of more than one species-specific morphological pattern. Mixed infections are frequently underreported due to oversight or the dominance of one species [17].
Parasite Quantification
- On Thick Smear: Tally parasites against WBCs until 500 parasites or 1,000 WBCs are counted. Calculate parasites/μL using the patient's known WBC count or an assumed count of 8,000 WBCs/μL: Parasites/μL = (parasites counted / WBCs counted) × WBC count per μL [20].
- On Thin Smear: Count parasitized RBCs among 500-2,000 total RBCs. Calculate the percent parasitemia: % Parasitemia = (parasitized RBCs / total RBCs) × 100 [20].
Research Reagent Solutions
The following table details essential materials and reagents required for conducting high-quality thick smear microscopy.
Table 2: Essential Research Reagents and Materials for Thick Smear Microscopy
| Item | Function/Application | Specifications & Notes |
|---|---|---|
| EDTA Tubes | Anticoagulant for venous blood collection. | Prevents clotting. Morphology is best preserved if smears are made within 4 hours of collection [10]. |
| Giemsa Stain | Differential staining of parasite chromatin (blue) and cytoplasm (blue/pink). | 3-5% solution, pH 7.2. Gold standard for morphological detail and species ID [17] [35]. |
| Field's Stain | Rapid two-step staining for field use. | Provides quicker results than Giemsa but may offer less morphological detail [34]. |
| Microscope Slides & Coverslips | Platform for blood smear preparation. | Must be clean and grease-free to ensure even spreading of blood. |
| Light Microscope | Visualization of stained blood smears. | Must be equipped with 10x, 20x, 40x, and 100x oil immersion objectives [20]. |
| Immersion Oil | Used with 100x objective to increase resolution. | Essential for detailed observation of parasite internal morphology. |
| Acridine Orange | Fluorescent dye that stains nucleic acids. | Used in techniques like QBC for potentially more sensitive detection, but less common for species ID [17] [20]. |
Ensuring Diagnostic Accuracy: Troubleshooting Common Pitfalls and Quality Control
Identifying and Resolving Common Staining and Preparation Artifacts
In the context of thick blood smear microscopy for malaria diagnosis, the accuracy of results is fundamentally dependent on the quality of the smear preparation and staining processes. Artifacts introduced during these stages can significantly impede automated analysis, confound the identification of parasites by AI models, and reduce the reliability of diagnostic data for research and drug development [36]. These artifacts, which can range from stain precipitate to improper smear thickness, are a major source of error, leading to misdiagnosis and compromising the integrity of research findings. This application note provides a detailed guide to identifying, troubleshooting, and preventing these common issues, ensuring the production of high-quality smear images suitable for robust computational analysis.
Common Artifacts and Their Impact on Analysis
Artifacts in blood smear images can be mistakenly identified as parasites by both human technicians and deep learning models, directly impacting the performance of computer-aided diagnostic (CAD) systems. The following table summarizes the primary artifacts, their causes, and consequences for research and diagnosis.
Table 1: Common Staining and Preparation Artifacts in Thick Blood Smear Microscopy
| Artifact Type | Visual Description | Common Causes | Impact on Research & Analysis |
|---|---|---|---|
| Stain Precipitate | Irregular, crystalline deposits that can be various sizes [36]. | Inadequate filtration of stain, over-concentration of stain solution, improper pH balance. | High false positive rates in object detection models (e.g., YOLO); can be misclassified as parasites or hemozoin [36]. |
| Inadequate Smear Thickness | Improperly thick smears prevent lysis of RBCs; thin smears have uneven cell distribution [36]. | Incorrect blood volume or spreading technique/angle. | Prevents accurate parasite detection; algorithms cannot standardize cell count per field of view [36]. |
| Bacteria/Fungi Contamination | Small, budding, or filamentous structures not conforming to parasite morphology [36]. | Contaminated stain, water, or slides; poor storage conditions. | Introduces non-malarial biological objects that challenge classification models, reducing specificity [36]. |
| Cell Debris & Dirt | Irregular, non-structured particles scattered across the slide [36]. | Dirty slides, dust settling on wet smears, use of non-lint-free materials. | Increases background "noise," complicating image segmentation and feature extraction for machine learning [36]. |
| Fixation Artifacts | Creation of "holes" or clear spots in the blood film; distorted cellular morphology. | Prolonged fixation or fixation of thick smears (which should not be fixed). | Obscures parasites and distorts red blood cells, leading to loss of diagnostic information and erroneous data points. |
| Uneven Staining | Areas of over-staining (excessively dark) or under-staining (excessively light) [3]. | Insufficient staining time, expired stain, uneven application. | Reduces contrast between parasite and background, lowering the confidence of AI in classification tasks [3]. |
Experimental Protocols for Artifact Assessment and Mitigation
Protocol: Standardized Giemsa Staining for Thick Smears
This protocol is designed to minimize the introduction of artifacts during the staining of thick blood smears, which is critical for generating consistent, high-quality data for computational models [36].
- Principle: Giemsa stain differentiates malaria parasites from host red blood cells by binding to nuclear DNA and cytoplasmic RNA. A standardized process ensures consistent color and contrast for machine learning applications.
- Materials:
- Methanol-free (absolute) Giemsa stain concentrate
- pH-buffered water (e.g., phosphate buffer, pH 7.2)
- Staining Coplin jars or a dedicated staining rack
- pH meter
- Filter paper or a syringe filter (0.45 µm)
- Procedure:
- Preparation: Prepare a 10% Giemsa working solution by diluting the concentrate with pH-buffered water. Filter the working solution immediately before use to remove crystalline precipitate [36].
- Staining: Place the unfixed thick smear horizontally on a staining rack. Gently flood the slide with the filtered 10% Giemsa stain for 20-30 minutes.
- Rinsing: Tilt the slide and gently rinse with a slow, steady stream of pH-buffered water (or clean tap water of neutral pH) to remove excess stain. Do not pour water directly onto the smear.
- Drying: Place the slide vertically in a draining rack and allow it to air-dry completely in a dust-free environment.
- Troubleshooting:
- Excessive Blue Tint: Check the pH of the buffer and water; a pH that is too high will cause this.
- Excessive Precipitate: Always filter the stain and ensure slides are not left in stain for excessively long periods.
- Pale Staining: Confirm the stain is not expired and that the dilution ratio is correct.
Protocol: Quality Control and Image Validation
This methodology outlines the steps for manually validating blood smear images, a crucial process for curating training datasets for deep learning models and ensuring their reliability [36].
- Principle: To establish a "ground truth" for smear quality by identifying and excluding images with significant artifacts that would compromise algorithmic training or diagnostic accuracy.
- Materials:
- Prepared and stained thick blood smears
- Microscope with 100x oil immersion objective
- Digital camera or smartphone attached via an adapter for image capture [36]
- Procedure:
- Systematic Slide Examination: Systematically scan the slide using a standardized pattern (e.g., a grid) to assess overall staining quality and artifact prevalence.
- Image Capture: Capture images from multiple, random fields of view. The use of a 3D-printed adapter to couple a smartphone to the microscope eyepiece standardizes this process across devices [36].
- Annotation and Exclusion: Annotate images using a consistent schema. Tags should include specific artifacts present (e.g., "stainprecipitate," "debris," "unevenstain"). Images with overwhelming artifacts that obscure the field of view should be excluded from research datasets [36].
- Dataset Curation: Maintain a balanced dataset that includes high-quality images of all parasite stages and common artifacts to improve model robustness.
The following workflow diagrams the integrated process of smear preparation, quality control, and computational analysis, highlighting key decision points for artifact management.
Diagram 1: Blood smear preparation and quality control workflow for AI-based diagnosis.
Computational Approaches to Mitigate Artifact Impact
Even with optimized protocols, some artifacts are inevitable. Advanced computational methods can be integrated into the analysis pipeline to enhance robustness.
- Deep Learning with Attention Mechanisms: Modern object detection frameworks, such as the YOLO-Para series, integrate attention mechanisms that help the model focus on the most relevant parts of an image, improving its ability to distinguish true parasites from distracting artifacts [37].
- Composite Loss Functions: Architectures like the Hybrid Capsule Network (Hybrid CapNet) employ composite loss functions that integrate margin, focal, and reconstruction losses. This enhances the model's robustness to annotation noise and class imbalance, often caused by mislabeled artifacts [3].
- Phase-Based Classification: For label-free imaging techniques like digital holographic microscopy (DHM), analysis based on the optical phase of the cell, rather than traditional morphological features, has proven more reliable. Phase information undergoes significant alterations due to the parasite, while being less sensitive to staining artifacts or minor segmentation errors [12].
The following diagram illustrates how a deep learning model can be structured to improve resilience against artifacts commonly found in smear images.
Diagram 2: Deep learning architecture for artifact-resilient parasite detection.
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key materials and their specific functions in ensuring the preparation of artifact-free smears for research.
Table 2: Essential Reagents and Materials for High-Quality Smear Preparation
| Item | Specification/Function | Research Application Notes |
|---|---|---|
| Giemsa Stain | Commercial concentrate; requires dilution and buffering to pH 7.2. | Filtering the working solution through a 0.45 µm syringe filter is a critical step to remove precipitate, a major source of artifact [36]. |
| pH Buffers | Phosphate buffer, pH 7.2. | Maintaining correct pH is essential for proper color balance (chromatin - red; cytoplasm - blue), which is critical for algorithm performance [3]. |
| Microscope Slides | Pre-cleaned, 75 x 25 mm, 1.0-1.2 mm thickness. | Using certified pre-cleaned slides minimizes the presence of dirt, dust, and grease that create artifacts [36]. |
| Smartphone & 3D-Printed Adapter | Standardizes image capture from microscope eyepiece [36]. | Enables reproducible digitization of slides across different locations and hardware, crucial for building large, diverse datasets [36]. |
| Spatial Filter | A pinhole (e.g., 50 µm) for lensless inline digital holographic microscopy (LiDHM) [12]. | Creates a clean, coherent illumination beam in LiDHM setups, which are emerging as label-free, portable alternatives to stained microscopy [12]. |
The Impact of Coloration Quality on Diagnostic Accuracy and Automated Systems
Within the framework of research on thick blood smear (TBS) microscopy for malaria diagnosis, coloration quality is a critical pre-analytical variable. The staining process directly influences the ability of microscopists and automated systems to accurately identify and quantify Plasmodium parasites. This Application Note details the methodologies for evaluating staining quality and its profound impact on both manual diagnostic accuracy and the performance of deep learning-based automated detection systems. In resource-poor settings, where factors such as reagent quality, pH, and sample preparation can vary significantly, standardized assessment of coloration quality is not merely beneficial but essential for reliable diagnosis [38].
The following tables consolidate key quantitative findings from recent studies on coloration quality analysis and automated parasite detection.
Table 1: Image-Based Features for Assessing Coloration Quality in Thick Blood Smears [38]
| Feature Category | Specific Feature Vector | Performance (Classification Rate) | Color Space Used |
|---|---|---|---|
| Background Segmentation | Thresholding on S (Saturation) and V (Value) components | Robust segmentation independent of coloration quality | HSV |
| Colour Histogram | 15-bins histogram of the H (Hue) component | > 97% | HSV |
| Other Features Tested | PCA, Correlation, Variance, Other Histograms | Lower performance compared to Hue histogram | RGB/HSV |
Table 2: Performance of Deep Learning Models for Malaria Detection in Thick Smears [39]
| Detection Target | Algorithm Used | Reported Accuracy | Key Function |
|---|---|---|---|
| Malaria Parasites | YOLOv8 | 95% | Detection and counting |
| Leukocytes (White Blood Cells) | YOLOv8 | 98% | Detection and counting |
| Model Application | Parasite/L blood count | Significantly faster than expert manual count | Supports parasite density calculation per WHO guidelines |
Experimental Protocols
Protocol 1: Image-Based Assessment of Staining Coloration Quality
This protocol is designed to objectively evaluate the coloration quality of thick blood smears stained with Romanowsky-type stains, based on the methodology of [38].
I. Materials and Equipment
- LED-illuminated optical microscope (e.g., Axio Zeiss Scope.A1)
- 100x oil immersion objective lens
- Digital camera for microscope image acquisition
- Light meter (e.g., Extech Model 407026)
- Computer with image processing software (e.g., Python with OpenCV, MATLAB)
II. Procedure
- Microscope Standardization:
- Set the reflector insert and condenser aperture diaphragm to the center position.
- Open the field diaphragm to its maximum.
- Adjust the condenser vertically to be as close to the thick blood smear as possible.
- Using a light meter, calibrate the light intensity through the eyepieces to a standardized 22.4 lux [38].
Image Acquisition:
- For each TBS sample, capture 10 images from different fields of view near the center of the smear to ensure uniformity.
- Ensure image resolution and format are consistent across all acquisitions.
Background Segmentation:
- Convert the acquired RGB image to the HSV (Hue, Saturation, Value) color space.
- Apply a thresholding-based segmentation algorithm to the Saturation (S) and Value (V) components to separate the background from the foreground (leukocytes, platelets, parasites). This step is robust to variations in coloration quality [38].
Feature Extraction and Classification:
- From the segmented background, extract the Hue (H) component.
- Construct a 15-bin histogram of the Hue values.
- Use this histogram as the feature vector to classify the smear's coloration quality as "Good" or "Bad" using a pre-trained classifier.
Protocol 2: Preparation of Thick Blood Smears for Optimal Staining
Proper smear preparation is foundational for achieving high coloration quality. This protocol is adapted from CDC guidelines and best practices [4] [40].
I. Materials
- Pre-cleaned, frosted-end microscope slides
- Capillary blood from fingerstick or EDTA-anticoagulated venous blood
- Applicator stick or corner of another slide
- Methanol (for thin smears only)
- Romanowsky-type stain (e.g., Giemsa, Wright-Giemsa)
- pH-buffered water
II. Procedure for Thick Smear Preparation
- Placement: Place a small drop of blood (approximately 2-3 µl) in the center of the slide.
- Spreading: Using the corner of a second slide or an applicator stick, spread the drop in a circular pattern to achieve a uniform smear approximately 1.5 cm in diameter.
- Quality Check: A smear of correct density should allow newsprint to be barely readable through it when wet [4].
- Drying: Lay the slide flat and allow it to dry thoroughly at room temperature for a minimum of 30 minutes. Protect from dust and insects. Avoid heat-fixing.
- Staining: Do not fix thick smears with methanol. Stain according to the specific protocol for the Romanowsky stain being used, typically involving dilution in pH-buffered water (e.g., pH 7.2) and a specified staining duration. Rinse gently with buffered water.
Technical Adjustments for Hematocrit Variations:
- High Hematocrit (Thicker blood): Push the spreader slide more slowly, use a larger drop of blood, and decrease the pusher slide angle to less than 30° [40].
- Low Hematocrit (Thinner blood): Push the spreader slide faster, use a smaller drop of blood, and increase the pusher slide angle to up to 45° [40].
Visual Workflows
The following diagram illustrates the logical workflow for the integrated assessment of coloration quality and its role in automated malaria diagnosis.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Thick Blood Smear Malaria Research
| Item | Function/Application | Specification Notes |
|---|---|---|
| Modified Romanowsky Stain | Staining of cellular components in blood smears. Differentiates parasites, WBCs, and platelets from the background. | Inexpensive, alcohol-free, recommended by WHO for TBS [38]. |
| pH-Buffered Water | Diluting stain and rinsing smears. Critical for consistent coloration and preventing precipitate formation. | Typically pH 7.2; precise pH ensures correct staining characteristics [38]. |
| LED-Illuminated Microscope | High-quality, consistent visualization and digital image capture of blood smears. | LED illumination eliminates need for a blue filter; enables light calibration [38]. |
| YOLOv8 Algorithm | Deep learning framework for object detection. Used for simultaneous detection of parasites and leukocytes. | Enables rapid, automated counting and calculation of parasite density [39]. |
| Standardized Image Database | Training and validation of image analysis and machine learning algorithms. | Should include images of TBS with "Good" and "Bad" coloration quality from defined fields of view [38]. |
In the context of global malaria control and elimination, the accurate diagnosis of malaria via thick blood smear microscopy remains a cornerstone of effective patient management and surveillance. Despite advancements in molecular techniques, microscopy persists due to its cost-effectiveness, accessibility, and ability to provide critical information on parasite species and density. However, the diagnostic performance of microscopy is highly dependent on several technical variables whose optimization is crucial for reliable results. This application note delineates the impact of three critical technical variables—stain pH, drying time, and microscopist expertise—on the quality and reliability of thick blood smear microscopy for malaria diagnosis. Framed within broader thesis research on optimizing diagnostic protocols, this document provides detailed methodologies and data analysis to guide researchers, scientists, and drug development professionals in standardizing procedures to enhance diagnostic accuracy and reproducibility in both clinical and research settings.
The Impact of Key Technical Variables on Smear Quality
The diagnostic sensitivity and specificity of thick blood smear microscopy are profoundly influenced by pre-analytical and analytical factors. Understanding and controlling these variables is fundamental to any research aiming to improve diagnostic protocols or evaluate new antimalarial compounds.
Stain pH: The pH of the Giemsa working buffer is a critical determinant of staining quality. A pH of 7.2 is specifically recommended for optimal staining of malaria parasites [41]. Deviations from this pH can lead to poor chromatic differentiation, where parasite chromatin (DNA) may not stain a distinct violet-blue and cytoplasm may not appear a clear blue [41]. This impairs the detection and species identification of Plasmodium parasites, particularly in cases of low parasitaemia or mixed infections.
Drying Time: Thick smears require sufficient and controlled drying to prevent detachment during staining. Insufficient drying can cause the entire smear to wash off the slide [4]. Drying times can range from 30 minutes to several hours at room temperature and are highly dependent on local humidity [4] [42]. The use of a fan or cooler in a hair dryer can accelerate this process to approximately 30 minutes, but heat must be avoided as it can fix the blood film, preventing the lysis of red blood cells and making the smear too dense to examine effectively [4] [42].
Microscopist Expertise: The skill of the laboratory professional is perhaps the most variable factor. Research demonstrates that targeted training interventions can lead to substantial improvements in diagnostic competency. A refresher training program in Ghana resulted in a significant increase in the median scores for parasite detection (from 64% to 87%), species identification (from 17% to 78%), and parasite quantification (from 20% to 50%) [43]. Furthermore, a study in Kenya found that recent refresher training and participation in a quality assurance program were significantly associated with accurate malaria diagnosis [44].
Table 1: Summary of Key Technical Variables and Their Optimal Specifications
| Technical Variable | Optimal Specification | Impact of Deviation from Optimal | Primary Reference |
|---|---|---|---|
| Giemsa Stain pH | 7.2 | Poor chromatic differentiation; impaired parasite detection and species ID. | [41] |
| Thick Smear Drying Time | 30 min to several hours (room temperature); ≥1 hour recommended. | Smear sloughing (if too short); potential heat-fixation (if accelerated with heat). | [4] [42] |
| Microscopist Training | Structured, refresher training with QA. | Low sensitivity/specificity; errors in species ID and quantification. | [43] [44] |
Detailed Experimental Protocols
Protocol 1: Giemsa Staining and pH Verification
This protocol ensures consistent, high-quality staining of thick blood smears for malaria parasite detection, as derived from established CDC guidelines [41].
Research Reagent Solutions:
- Working Giemsa Buffer (0.0067M, pH 7.2): Prepared by diluting 10 mL of autoclaved 0.67 M stock buffer (59.24 g Na₂HPO₄, 36.38 g NaH₂PO₄·H₂O in 1L deionized water) with 990 mL deionized water. The pH must be verified with a calibrated pH meter before use [41].
- Working Giemsa Stain (2.5%): Prepared fresh for each staining batch by mixing 1 mL of filtered stock Giemsa stain with 39 mL of working Giemsa buffer and adding 2 drops of 5% Triton X-100 to enhance staining [41].
- 5% Triton X-100: Prepared by slowly adding 5 mL of Triton X-100 to 95 mL of warmed deionized water (56°C) while swirling to mix [41].
Procedure:
- Preparation: Prepare fresh working Giemsa stain and buffer in staining jars. A second jar should contain 40 mL of working Giemsa buffer with 2 drops of Triton X-100 for rinsing [41].
- Staining: Place the thoroughly dried, unfixed thick smears into the working Giemsa stain for 45-60 minutes [41].
- Rinsing:
- For thin smears: Remove from stain and rinse by dipping 3-4 times in the Giemsa buffer/Triton X-100 solution.
- For thick smears: After staining, transfer them to the Giemsa buffer/Triton X-100 jar and leave for 3-5 minutes for gentle dehemoglobinization and rinsing [41].
- Drying: Remove slides and place them upright in a rack to air-dry completely [41].
- Quality Control: A known positive control smear (e.g., from a patient sample with adequate parasite density, stored at -70°C) should be included with each staining batch to ensure proper staining results have been achieved [41].
Protocol 2: Standardized Thick Smear Preparation with Scratch Method
This protocol details the scratch method, an evidence-based technique for preparing thick smears that minimizes sloughing and allows for faster processing, crucial for high-throughput research environments [45].
Procedure:
- Slide Preparation: Use pre-cleaned, labeled glass slides.
- Blood Application: Place a small drop of capillary or EDTA-anticoagulated venous blood (approx. 5-10 µL) in the center of the slide. Venous blood should be used within 2 hours of collection to preserve parasite morphology [4].
- Spreading via Scratch Method: Using the corner of a second glass slide, spread the blood in a circular pattern while applying firm, moderate pressure to create microscopic scratches on the underlying slide. Spread the drop to a diameter of 1.5-2 cm (the size of a dime) [4] [45].
- Drying: Lay the slides flat on a level surface and allow them to dry thoroughly. Drying must occur without the application of heat. Using a fan or cool-air hair dryer can reduce drying time to 20-30 minutes. A properly dried thick film should allow newsprint to be barely read through it when placed underneath [4] [45] [42].
- Storage (if not staining immediately): To prevent sloughing during future staining, briefly dip the dried thick smear in water to lyse the red blood cells, then allow it to dry again [4].
Protocol 3: Assessing Microscopist Competency
This protocol, modeled on the WHO's External Competency Assessment for Malaria Microscopists (ECAMM) framework, provides a standardized method for evaluating and quantifying microscopist performance in a research context [46].
Procedure:
- Panel Preparation: Create a validated slide panel comprising at least 20 slides. The panel should include:
- Negative smears: Blood from healthy individuals.
- Positive smears: Samples with varying parasite densities (low and high) and different Plasmodium species (P. falciparum, P. vivax), including mixed infections if possible [46] [47].
- Validation: All slides must be validated by a consensus of WHO-certified or similarly qualified expert microscopists, with molecular confirmation as a gold standard [46] [44].
- Pre-Test Assessment: Participants examine the slide panel and report on parasite detection (positive/negative), species identification, and, if required, parasite quantification. A time limit of 10 minutes per slide is recommended to simulate routine diagnostic pressure [46].
- Intervention: Conduct a structured training intervention over several days, incorporating theoretical lectures and intensive, supervised practical sessions on smear preparation, staining, parasite detection, species identification, and quantification [43] [46].
- Post-Test Assessment: Following the training, participants examine a different but equivalently validated slide panel under the same conditions as the pre-test.
- Data Analysis: Compare pre- and post-test results against the reference standard to calculate performance metrics, including sensitivity, specificity, species identification accuracy, and quantification precision. Inter-reader agreement can be expressed using kappa (κ) statistics [44] [47].
Table 2: Exemplar Data from Microscopist Training Efficacy Studies
| Performance Metric | Pre-Training Median Score | Post-Training Median Score | p-value | Study Context |
|---|---|---|---|---|
| Parasite Detection | 64% | 87% | < 0.001 | Ghana, 765 lab professionals [43] |
| Species Identification | 17% | 78% | < 0.001 | Ghana, 765 lab professionals [43] |
| Parasite Quantification | 20% | 50% | < 0.001 | Ghana, 765 lab professionals [43] |
| Sensitivity | 69% | 97% | < 0.01 | Kenya, QA-pilot facilities [44] |
| Specificity | 93% | 100% | < 0.01 | Kenya, QA-pilot facilities [44] |
Integrated Workflow and Data Analysis
The following diagram synthesizes the protocols and variables discussed into a cohesive research workflow for evaluating technical variables in thick smear microscopy. This integrated approach allows for the systematic assessment of how stain pH, drying time, and training interventions collectively impact diagnostic outcomes.
Research Workflow for Technical Variables
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Thick Blood Smear Research
| Item | Specification / Function | Research Context |
|---|---|---|
| Giemsa Stain Powder | Certified for consistency. Stains parasite chromatin and cytoplasm for visual identification. | Critical for standardizing staining across experimental batches [41]. |
| pH Meter | Calibrated, for verifying working buffer pH of 7.2. | Ensures optimal staining chromaticity, a key dependent variable [41]. |
| Phosphate Salts | Na₂HPO₄ & NaH₂PO₄·H₂O for 0.67M stock buffer. | Maintains stable pH for reproducible staining results [41]. |
| Triton X-100 (5%) | Non-ionic surfactant. Added to stain and rinse buffer to reduce sloughing and improve stain penetration. | Key reagent for improving thick film adherence and quality [41]. |
| Validated Blood Smear Panel | Slides with confirmed species, density, and negative samples. | Serves as the reference standard for assessing microscopist competency and test accuracy [46] [47]. |
| Microscope with Oil Immersion | 100x objective lens for parasite examination. | Essential tool for endpoint analysis [20]. |
| Reference Training Materials | WHO basic malaria microscopy guide, ECAMM resources. | Foundation for designing and implementing effective training interventions [43] [46]. |
In the context of malaria diagnosis, quality assurance (QA) through proficiency testing (PT) and competency assessment is not merely a supplementary activity but a fundamental component of reliable surveillance and effective treatment strategies. The remarkable achievement of malaria elimination in China and its ongoing vulnerability to imported cases underscores the critical importance of maintaining diagnostic proficiency among laboratory personnel [46]. As countries progress toward elimination, opportunities for practical experience with malaria microscopy decline, creating a paradoxical situation where diagnostic skills deteriorate precisely when accurate detection becomes most crucial for preventing reintroduction [46]. This application note provides researchers, scientists, and drug development professionals with detailed protocols and frameworks for implementing robust QA systems specifically for thick blood smear microscopy in malaria research and diagnosis.
Theoretical Framework and Current Landscape
Proficiency testing, a form of external quality assessment (EQA), involves sending panels of characterized samples to participating laboratories to verify their testing reliability and accuracy [48]. In malaria diagnosis, PT serves as an objective measure of a laboratory's capability to correctly identify and quantify Plasmodium species—a critical competency given that different species require distinct treatment approaches and have varying public health implications.
The decline in malaria diagnostic proficiency is a widespread concern. In China, 75% of participants in a recent study had received no formal malaria microscopy training in the preceding five years, creating significant gaps in diagnostic capabilities despite the country's malaria-free certification [46]. Similarly, in Ethiopia, the transition of malaria diagnostic responsibilities from specialized microscopists to general laboratory technicians has raised concerns about proficiency, particularly in species differentiation and parasite quantification [49].
Table 1: Performance Metrics in Malaria Proficiency Testing Across Regions
| Region | Parasite Detection Accuracy | Species Identification Accuracy | Quantification Proficiency | Key Challenges |
|---|---|---|---|---|
| Hunan Province, China | 25.52% improvement post-training [46] | 48.96% improvement in species differentiation [46] | Not formally assessed (75% lacked baseline training) [46] | Interspecies confusion, particularly P. ovale as P. malariae (20.8% of errors) [46] |
| West Oromia, Ethiopia | 95.7% sensitivity, 96.2% specificity [49] | 69% agreement (kappa = 0.52, moderate) [49] | 3.4% within expected range [49] | Educational level, lack of recent training [49] |
| Nigeria | Variable performance across PT rounds [48] | Not specifically reported | Not specifically reported | Gross deficiencies in quality of laboratory services [48] |
Experimental Protocols and Methodologies
Protocol 1: External Competency Assessment for Malaria Microscopists (ECAMM)
The WHO's ECAMM framework provides a standardized approach for assessing and improving malaria microscopy competency. The following protocol adapts this framework for implementation in research settings and diagnostic laboratories [46].
Participant Recruitment and Preparation
- Participant Selection: Recruit malaria microscopists from relevant institutions. In the Hunan Province study, participants were from CDC-affiliated institutions across 14 municipal cities and autonomous prefectures, with each institution dispatching 1–2 personnel responsible for malaria microscopy [46].
- Equipment Preparation: Secure high-resolution bright-field microscopes (one per participant), one digital camera-equipped microscope system for demonstration purposes, computerized imaging station with printer, and Giemsa-stained thick/thin blood smears [46].
- Blood Smear Validation: All blood smears must undergo rigorous validation through consensus review by three ECAMM Level 1-certified experts and confirmatory nucleic acid amplification testing before use in assessment [46].
Blood Smear Composition and Panel Design
The blood smear panel should include a balanced distribution of negative samples and positive infections representing different Plasmodium species at varying parasite densities.
Table 2: Recommended Blood Smear Composition for Proficiency Testing
| Slide Type | Quantity | Specifications | Validation Requirements |
|---|---|---|---|
| Negative blood smears | 20 | No parasites present; may include confounding factors (e.g., platelet clusters, stain precipitate) | Expert verification + molecular confirmation [46] |
| P. falciparum-positive | 12 | Various parasite densities (low, medium, high) | Expert verification + molecular confirmation [46] |
| P. vivax-positive | 8 | Various parasite densities and developmental stages | Expert verification + molecular confirmation [46] |
| P. malariae-positive | 4 | Confirm presence of characteristic forms | Expert verification + molecular confirmation [46] |
| P. ovale-positive | 4 | Focus on differentiation from P. malariae | Expert verification + molecular confirmation [46] |
| Mixed infections | 4 | Typically P. falciparum with another species | Expert verification + molecular confirmation [46] |
Assessment Implementation
- Pre-assessment: Conduct a 135-minute baseline skill evaluation followed by 30 minutes of expert feedback [46].
- Theoretical Training: Deliver four didactic lectures (420 minutes total) covering blood elements differentiation, pseudo-parasites/artifacts identification, parasite quantification, and quality assurance principles [46].
- Competency Verification: Conduct four 105-minute standardized assessments where participants examine validated slide panels [46].
- Examination Conditions: Participants must independently examine slides and submit diagnostic interpretations within a strictly enforced 10-minute examination window per slide. Inter-participant communication must be prohibited during testing [46].
- Stage Evaluation: Announce test results, review incorrect slides using a camera-integrated live tracking microscopy system, and conduct on-site Q&A sessions (240 minutes total) [46].
Protocol 2: Proficiency Testing for Resource-Limited Settings
This protocol, adapted from studies in Nigeria and Ethiopia, provides a more resource-efficient approach suitable for areas with limited infrastructure [48] [49].
PT Panel Preparation and Distribution
- Panel Composition: Prepare a proficiency panel consisting of 10 pre-validated slides: 5 positive and 5 negative, representing the most prevalent species in the region [49].
- Sample Characterization: In Benin, panels may consist of samples with 1000, 500, and 0 parasites/μL; in Liberia, panels may include one sample at 1000 parasites/μL, two at 500 parasites/μL, and one negative [50].
- Storage and Transportation: Store proficiency panels under refrigeration with reactivity known only to the study coordinator. Rehydrate samples immediately before testing and label with random identification numbers to maintain blinding [50].
- Validation: Panel slides should be prepared as smears and stained according to WHO guidelines, then validated by WHO Level 1 certified laboratory personnel [49].
On-site Evaluation and Mentoring
- Direct Observation: During PT panel testing by health center staff, the study coordinator should observe the technician and record correct and incorrect testing procedures using a checklist of critical steps in the testing procedure [50].
- Performance Recording: Complete a pass/fail form to indicate whether the technician read and recorded correct sample reactivity. Document errors irrespective of frequency across samples [50].
- Immediate Feedback: Provide feedback regarding health worker performance and offer corrections when needed [50]. This mentoring component has demonstrated significant impact on performance improvement.
Data Analysis and Interpretation Frameworks
Quantitative Assessment and Scoring
Implement standardized scoring systems to evaluate performance across multiple dimensions of competency:
- Parasite Detection: Score as correct identification of positive vs. negative samples.
- Species Identification: Evaluate correct classification of Plasmodium species.
- Quantification Accuracy: Assess parasite counting against established reference values.
- Composite Score: Calculate overall performance combining all dimensions.
In the Hunan Province study, non-parametric comparisons between pre-training assessments and post-training evaluations were conducted using Wilcoxon test, with competence scores expressed as median values and 95% confidence intervals [46].
Statistical Analysis Protocols
- Data Collection: Collect data from paper-based tests, questionnaires, and digital applications, then enter into statistical software packages [46].
- Correlation Analysis: Compute Kendall's tau-b correlation coefficients to examine relationships between participants' background information and diagnostic performance metrics [46].
- Significance Testing: Set statistical significance threshold at α = 0.05 (two-tailed), considering differences significant when p < 0.05 [46].
- Normality Testing: Assess normality of distribution using appropriate tests (Kolmogorov-Smirnov, Shapiro-Wilk) before selecting parametric vs. non-parametric tests [51].
- Psychometric Properties: Establish reliability and validity of assessment instruments, with Cronbach's alpha scores > 0.7 considered acceptable for internal consistency [51].
Implementation Workflow
The following diagram illustrates the core workflow for implementing a proficiency testing and competency assessment program:
Proficiency Testing and Competency Assessment Workflow
Research Reagent Solutions and Essential Materials
Table 3: Essential Materials for Malaria Microscopy Proficiency Testing
| Item | Specifications | Function/Application |
|---|---|---|
| High-resolution bright-field microscopes | 100x oil immersion objective [46] | Detailed visualization of parasite morphology |
| Giemsa stain | Commercial or prepared according to WHO specifications [46] | Differential staining of malaria parasites and blood components |
| Pre-validated blood smear panels | 48-52 slides including negatives and multiple species [46] | Standardized assessment of diagnostic competency |
| Digital microscope camera system | e.g., Leica DM2000 with imaging station [46] | Demonstration, training, and documentation of findings |
| Quality control slides | Known positive and negative samples [46] | Daily verification of staining and microscopy quality |
| Standardized data collection forms | Capture participant demographics and performance metrics [46] [49] | Systematic documentation of assessment results |
| Molecular validation tools | PCR or other NAAT methods [46] | Confirmatory testing of reference slides |
Technological Innovations and Future Directions
Artificial intelligence and machine learning approaches are emerging as valuable tools for enhancing quality assurance in malaria microscopy. The Hybrid Capsule Network (Hybrid CapNet) represents a promising development, combining CNN-based feature extraction with dynamic capsule routing for accurate parasite identification and life-cycle stage classification [3]. This lightweight architecture (1.35M parameters, 0.26 GFLOPs) achieves superior accuracy with significantly reduced computational cost, making it suitable for mobile diagnostic applications in resource-constrained settings [3].
These technological advancements may eventually complement human expertise, particularly in areas where skilled microscopists are scarce. However, current implementations still require validation against human performance, and traditional proficiency testing remains essential for establishing baseline competency and identifying specific areas for improvement.
Implementing robust proficiency testing and competency assessment programs is fundamental to maintaining diagnostic accuracy in malaria microscopy, particularly in post-elimination settings and regions with declining case numbers. The protocols outlined in this application note provide researchers and public health professionals with evidence-based frameworks for assessing and improving microscope competency. Regular participation in such programs, combined with structured training and mentoring, significantly enhances diagnostic performance and contributes to more effective malaria surveillance and patient management. Institutionalizing these approaches as certification programs, especially targeting primary healthcare facilities, will help sustain malaria diagnostic capacity and prevent the reintroduction of eliminated diseases.
Benchmarking Performance: Validation of Thick Smears Against Emerging Diagnostic Technologies
Within the framework of research on thick blood smear microscopy for malaria diagnosis, the validation of analytical sensitivity and specificity against molecular standards is a critical endeavor. This protocol outlines a standardized procedure for establishing the diagnostic accuracy of thick blood smear microscopy by using polymerase chain reaction (PCR) as a reference molecular standard. The persistent challenge in malaria diagnosis lies in the variable performance of routine diagnostic tests, particularly in resource-limited, high-transmission settings where false-positive rapid diagnostic test (RDT) results, often due to persistent antigenemia, can lead to overtreatment and undermine public health interventions [52]. This document provides researchers and drug development professionals with a detailed methodology for a rigorous, head-to-head comparison of microscopy and molecular diagnostics, enabling the generation of reliable performance metrics such as sensitivity, specificity, and predictive values.
Experimental Design and Comparative Framework
The core of this validation study is a cross-sectional comparison of diagnostic outcomes from thick blood smear microscopy against a molecular gold standard, specifically real-time PCR. This design is apt for concurrently assessing the sensitivity and specificity of the index test (microscopy) [52]. A minimum sample size of 270 participants is recommended to ensure sufficient statistical power for estimating sensitivity and specificity with precision, though this number may be adjusted based on expected disease prevalence and desired confidence intervals [52].
Key Considerations:
- Population: The study should enroll febrile patients or individuals with a clinical suspicion of malaria from the target transmission setting (e.g., high-transmission vs. non-endemic). Participant demographics and clinical characteristics, including age, sex, and fever history, should be recorded, as these can influence test performance [52].
- Index Test: Thick blood smear microscopy, performed and interpreted by experienced microscopists according to International Council for Standardization in Haematology (ICSH) guidelines [53].
- Reference Standard: Real-time PCR for the detection of Plasmodium species DNA. A composite microbial reference, potentially combining multiple test results, can also be employed to define a case of malaria infection [54] [55].
- Blinding: Personnel performing and interpreting the microscopy must be blinded to the PCR results and vice versa to prevent assessment bias.
Materials and Reagents
Research Reagent Solutions
Table 1: Essential Materials and Reagents for Thick Smear Microscopy and Molecular Validation
| Item | Function/Description |
|---|---|
| K3EDTA Blood Collection Tubes | Prevents coagulation for consistent smear preparation and nucleic acid preservation [53]. |
| Microscope Slides (Pre-cleaned) | Substrate for preparing thick blood films; must be grease-free for optimal adhesion [4]. |
| Giemsa Stain | Standard Romanowsky stain that highlights chromatin (purple) and cytoplasm (blue) of malaria parasites [56] [4]. |
| Absolute Methanol | Used for fixing thin blood smears only; thick smears must not be fixed [4]. |
| DNA Extraction Kit | For purifying parasite genomic DNA from dried blood spots (DBS) or whole blood [52]. |
| Real-time PCR Master Mix & Primers/Probes | Targets species-specific Plasmodium genes (e.g., for P. falciparum, P. vivax, P. ovale, P. malariae) for amplification and detection [52] [54]. |
| Dried Blood Spot (DBS) Filter Paper | A stable, simple medium for collecting, transporting, and storing blood samples for later PCR analysis [52]. |
Methodologies
Specimen Collection and Preparation
- Blood Collection: Draw venous blood into K3EDTA tubes. For capillary blood, clean the finger thoroughly and perform a fingerstick [4].
- Thick Smear Preparation:
- Place a small drop of blood (approx. 10-15 μL) in the center of a labeled slide [4].
- Using the corner of a second slide, spread the drop in a circular pattern to achieve a uniform diameter of 1.5-2.0 cm. The ideal density allows text to be barely legible through the wet smear [4].
- Scratch Method (Optional for Improved Adherence): Use the edge of the spreader slide to apply firm pressure, creating small scratches on the underlying slide as you spread the blood. This enhances adhesion and can reduce drying time to 20-30 minutes [4].
- Air-dry the smears thoroughly in a horizontal position, protected from dust and insects. Do not heat-fix or chemically fix thick smears [4].
- Dried Blood Spot (DBS) Collection: Apply a separate drop of blood from the patient onto a designated area of DBS filter paper. Allow it to dry completely at room temperature before storage in a sealed plastic bag with a desiccant [52].
Thick Smear Staining and Microscopy
- Staining: Stain the thoroughly dried thick smears with a 10% Giemsa solution (pH 7.2) for 10-15 minutes [56] [4].
- Microscopy Examination:
- Examine stained smears under an oil immersion objective (100x magnification) [16].
- Systematically scan the entire smear for a minimum of 100 fields before declaring a result negative [16].
- For positive samples, an experienced microscopist should examine at least 200 leukocytes to estimate parasite density and attempt species identification, though the latter is more reliably performed on a concomitant thin smear [53] [4].
Molecular Detection by PCR
- DNA Extraction: Extract genomic DNA from a 3-6 mm punch of the DBS or from 100-200 μL of whole blood using a commercial DNA extraction kit, following the manufacturer's protocol [52].
- Real-time PCR Setup: Perform real-time PCR amplification using species-specific primers and probes. Include appropriate controls (positive, negative, no-template) in each run [52] [54].
- Result Interpretation: Analyze amplification curves. A cycle threshold (Ct) value below a predetermined cut-off indicates a positive result for the specific Plasmodium species.
Diagram 1: Experimental validation workflow for comparing microscopy against PCR.
Data Analysis and Performance Metrics
Data Collection and 2x2 Contingency Table
Compile all results into a 2x2 contingency table, cross-tabulating the outcomes of the index test (thick smear microscopy) against the reference standard (PCR).
Table 2: 2x2 Contingency Table for Diagnostic Accuracy Calculation
| PCR Positive | PCR Negative | Total | |
|---|---|---|---|
| Microscopy Positive | True Positive (TP) | False Positive (FP) | TP + FP |
| Microscopy Negative | False Negative (FN) | True Negative (TN) | FN + TN |
| Total | TP + FN | FP + TN | N |
Calculation of Performance Metrics
Calculate the following key metrics with 95% confidence intervals (CI) to quantify diagnostic performance [52] [57] [55]:
- Analytical Sensitivity = TP / (TP + FN) × 100
- Analytical Specificity = TN / (TN + FP) × 100
- Positive Predictive Value (PPV) = TP / (TP + FP) × 100
- Negative Predictive Value (NPV) = TN / (TN + FN) × 100
Representative Quantitative Data
The following table summarizes performance data from published studies to provide benchmark values. Note that performance can vary significantly based on transmission setting and microscopist expertise.
Table 3: Representative Performance Metrics of Malaria Diagnostic Tests Against PCR
| Diagnostic Test | Sensitivity (%, 95% CI) | Specificity (%, 95% CI) | Context / Citation |
|---|---|---|---|
| Thick Smear Microscopy | 90.4% (CI: 84.5-94.2) | 100.0% (CI: 98.8-100.0) | Dakar, Senegal [55] |
| HRP2/pLDH RDT | 93.6% (CI: 82.5-98.7) | 99.4% (CI: 98.9-99.7) | Imported malaria in UK children [57] |
| HRP2/pLDH RDT | 99.4% (CI: 95.9-100.0) | 46.7% (CI: 37.7-55.9) | High-transmission setting, Uganda [52] |
| Laboratory HRP2 Antigen Detection | 97.9% (CI: 94.2-99.3) | 48.1% (CI: 42.8-53.4) | Dakar, Senegal [55] |
Advanced Applications: Two-Step Diagnostic Algorithms
Research in high-transmission settings has demonstrated that a single RDT can lack specificity. A validated solution is a two-step algorithm that uses a highly sensitive screening test followed by a specific confirmatory test. The workflow below illustrates this approach using RDT and microscopy, which has been shown to significantly improve specificity with minimal loss of sensitivity [52].
Diagram 2: Two-step diagnostic algorithm to improve specificity.
Discussion and Interpretation
Validation against molecular standards is indispensable for quantifying the true performance of thick smear microscopy. The data from comparative studies reveal critical insights:
- Microscopy vs. RDTs: While RDTs offer operational advantages, microscopy maintains a crucial role in species identification and parasite quantification, which are vital for treatment decisions and prognostic assessment [16]. The high specificity of microscopy, as shown in Table 3, makes it a reliable confirmatory test.
- Impact of Persisting Antigens: The low specificity of HRP2-based RDTs in high-transmission settings (46.7% in Uganda) is often attributed to the persistence of HRP2 antigenemia after successful treatment of an acute infection, leading to false-positive results [52]. This underscores the need for confirmatory testing in such contexts.
- Role of PCR: PCR offers superior sensitivity, especially at low parasite densities, and is the best tool for resolving discrepant results, confirming species, and detecting mixed infections [52] [16]. However, its turn-around time limits its utility for acute clinical diagnosis, positioning it primarily as a research and reference tool [16].
In conclusion, this protocol provides a standardized framework for validating thick blood smear microscopy. The resulting sensitivity and specificity metrics are essential for researchers and drug developers to critically evaluate diagnostic performance, optimize testing algorithms, and advance the development of novel diagnostic tools for malaria.
Within the framework of malaria diagnosis research, microscopy of peripheral blood smears remains a cornerstone. The gold standard for diagnosis has long been the parasitology thick and thin smear (PS), which combines the sensitivity of the thick smear with the species-identifying capability of the thin smear [58]. However, the diagnostic landscape is evolving. In many clinical settings, particularly outside high-endemicity regions, hematology thin smears (HS)—prepared routinely for complete blood count (CBC) analysis—are often the first to be examined. This Application Note provides a comparative analysis of these two methods, summarizing critical performance data and providing detailed protocols to guide researchers and scientists in their diagnostic and drug development efforts. The integration of new technologies, such as automated analyzers and artificial intelligence (AI), is also explored for its potential to augment traditional methods.
Comparative Performance Data
The diagnostic performance of Hematology Thin Smears (HS) versus the gold standard Parasitology Thick and Thin Smears (PS) has been quantitatively assessed in a recent clinical study. The data below summarizes the key findings from 529 paired tests [58].
Table 1: Diagnostic Performance of Hematology Thin Smears vs. Parasitology Smears
| Performance Metric | Value (%) |
|---|---|
| Sensitivity | 93.3 |
| Specificity | 99.8 |
| Positive Predictive Value (PPV) | 97.7 |
| Negative Predictive Value (NPV) | 99.4 |
Table 2: Concordance Analysis Between HS and PS
| Smear Type | Plasmodium Cases | Babesia Cases | Total PS+/HS+ Cases |
|---|---|---|---|
| Initial Diagnoses | 21 | 21 | 42 |
| Follow-up Tests | 4 discordant cases (3 PS+/HS-, 1 PS-/HS+) with parasitemia below quantification level |
Table 3: Operational and Practical Characteristics
| Characteristic | Parasitology Smears (PS) | Hematology Thin Smears (HS) |
|---|---|---|
| Primary Diagnostic Role | Gold standard for detection and species identification [4] | Initial, rapid assessment; widely available in laboratories performing CBCs [58] |
| Sensitivity | High (reference standard) | High (93.3%), 100% for initial diagnoses in study [58] |
| Species Identification | Possible with thin smear component [4] | Limited; reported as "Plasmodium or Babesia present" [58] |
| Typical Availability | Weekdays during business hours [58] | All hours of the day and week [58] |
Experimental Protocols
To ensure reproducible and accurate results, adherence to standardized protocols for smear preparation, staining, and examination is paramount. The following sections detail the methodologies for both parasitology and hematology smears as cited in the literature.
Protocol for Parasitology Thick and Thin Smears
This protocol, based on CDC guidelines, is considered the gold standard for malaria diagnosis [4].
Specimen Collection and Smear Preparation
- Specimen: Use venous blood collected in an EDTA tube or capillary blood from a fingerstick.
- Slide Preparation: Prepare at least two smears per patient on pre-cleaned, labeled slides.
- Thick Smear:
- Place a small drop of blood in the center of the slide.
- Using the corner of another slide or an applicator stick, spread the drop in a circular pattern to achieve a diameter of approximately 1.5 cm.
- The smear should be of a density that allows newsprint to be barely read through it when wet.
- Allow smears to dry thoroughly (30 minutes to several hours). Do not fix thick smears with methanol or heat.
- Thin Smear:
- Place a small drop of blood near the frosted end of the slide.
- Bring a second slide (spreader) at a 30-45° angle up to the drop, allowing the blood to spread along the contact line.
- Quickly push the spreader slide toward the unfrosted end of the lower slide to create a feathered edge.
- Allow the thin smear to air dry, then fix by dipping in absolute methanol.
Staining and Examination
- Staining: Stain thick and thin smears with 2.5% Giemsa stain for 45 minutes [58].
- Washing: After staining, gently dip thin smears in buffered water. Thick smears should be placed in buffered water for 3-5 minutes to decolorize.
- Microscopy:
- Examine slides at 10x, 20x, and 100x (oil immersion) objectives.
- Carefully examine the thin smear at 100x magnification for at least 300 fields to identify parasites and determine species based on morphological characteristics.
- The thick smear should be examined for the efficient detection of parasites.
Protocol for Hematology Thin Smears
This protocol is adapted from the methodology used in the comparative clinical study [58].
Smear Preparation and Staining
- Specimen: Use peripheral whole blood collected in an EDTA tube. Prepare smears within one hour of collection to preserve optimal morphology [22].
- Automated Preparation: Four thin smears can be prepared using an automated blood smearing instrument (e.g., HemaPrep) to ensure a consistent length of 1–1.5 inches with a feathered edge.
- Staining: Stain two thin smears using an automated stainer (e.g., Sysmex SP-10) with Wright-Giemsa stain and a suitable buffer (pH 6.8). Assess stain quality to ensure clear differentiation of nuclear and cytoplasmic material.
Microscopy and Parasitemia Quantification
- Examination: Two general technologists, trained to recognize Plasmodium and Babesia but not to differentiate species, independently examine the two stained smears.
- Screening: Use a brightfield microscope to screen at 10x, 20x, and 50x objectives for large parasites, and at 100x oil immersion for intraerythrocytic parasites. Examine 300 oil immersion fields on each of the two smears.
- Quantification of Parasitemia:
- Use a Miller disc reticle in the microscope ocular.
- Count parasitized red blood cells (RBCs) until at least 1,000 RBCs are assessed.
- Calculate the percentage of parasitemia using the formula:
(Number of parasitized RBCs in large square × 100) / (Total RBCs in small square × 9). - Repeat on the second smear; the two results should agree within two percentage points. Report the average, rounded to the nearest whole number.
Emerging and Automated Techniques
Automated Haematology Analyzers
- Technology: The Sysmex XN-31 analyzer is a flow cytometry-based device that uses a 405-nm laser to analyze RBCs treated with lysing and fluorescent staining reagents [59].
- Output: It provides the number and ratio of malaria-infected RBCs (MI-RBC), a complete blood count (CBC), and suggests species identification based on scattergram patterns, all within approximately one minute [59].
- Performance: Clinical testing showed a high correlation coefficient (>0.99) between XN-31 parasitemia results and manual microscopy counts, demonstrating strong agreement with the gold standard [59].
Artificial Intelligence (AI) and Deep Learning
- Technology: AI, particularly deep learning models like YOLOv3 (You Only Look Once), can be trained to detect parasites in digital images of thin blood smears [60].
- Process: Images of Giemsa-stained smears are captured, preprocessed, and used to train a convolutional neural network (CNN) to identify and classify infected RBCs [60] [61].
- Performance: One study reported an overall recognition accuracy of 94.41% for Plasmodium falciparum [60]. A broader review found AI models for malaria detection achieved a mean accuracy of 95.5% [61].
Workflow and Logical Diagrams
The following diagram illustrates the key decision points and pathways in the comparative diagnosis of malaria using thick, thin, and hematology smears.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents and Materials for Blood Smear Analysis
| Item | Function / Application |
|---|---|
| EDTA Blood Collection Tubes | Prevents coagulation to preserve blood cell morphology for analysis. |
| Glass Microscope Slides | Platform for preparing and examining blood smears. |
| Absolute Methanol | Fixative for thin blood smears; preserves cell structure before staining. |
| Giemsa Stain | Standard Romanowsky-type stain used to differentiate malaria parasite chromatin and cytoplasm. |
| Wright-Giemsa Stain | A variant used in automated hematology stainers for hematology thin smears. |
| Buffered Water (pH 6.8 or 7.2) | Used for diluting stain and rinsing slides; critical for consistent staining results. |
| Microscope with Oil Immersion | Essential for high-resolution (100x) examination of smears to identify intracellular parasites. |
| Miller Disc Reticle | Eyepiece graticule used for standardized manual counting of parasitized cells. |
| Lysercell M / Fluorocell M | Proprietary reagents for the XN-31 analyzer that lyse RBCs and stain nucleic acids, respectively [59]. |
| Digital Slide Scanner | Captures high-resolution whole-slide images for AI-based analysis and archiving [61]. |
Malaria remains a formidable global health challenge, with accurate diagnosis being a cornerstone of effective control, management, and elimination efforts. While thick blood smear microscopy has long been considered the gold standard for malaria diagnosis in clinical and research settings, the diagnostic landscape is rapidly evolving to incorporate more rapid, sensitive, and technologically advanced methods. These include Rapid Diagnostic Tests (RDTs), molecular techniques like Polymerase Chain Reaction (PCR), and emerging artificial intelligence (AI)-driven solutions.
Each diagnostic modality offers distinct advantages and limitations in terms of sensitivity, specificity, operational requirements, and applicability in different transmission settings. This article provides a comprehensive technical overview of these methods, focusing on their performance characteristics, standardized protocols, and implementation considerations within the context of malaria research and public health programs. The integration of these tools with traditional microscopy is creating a more robust, multi-faceted approach to malaria diagnosis and surveillance.
Performance Comparison of Diagnostic Modalities
The following tables summarize the performance characteristics of various malaria diagnostic methods based on recent clinical studies and evaluations.
Table 1: Overall Diagnostic Performance of Common Malaria Diagnostic Methods
| Diagnostic Method | Sensitivity Range | Specificity Range | Time to Result | Key Advantages | Major Limitations |
|---|---|---|---|---|---|
| Thick Blood Smear Microscopy | Varies with technician expertise [25] | Varies with technician expertise [25] | 30-60 minutes | Quantifies parasitemia, species identification [62] | Labor-intensive, requires expertise [62] |
| Rapid Diagnostic Tests (RDTs) | 87.6%-93.4% [25] [62] | 97.8%-100% [25] [62] | 15-20 minutes | Easy to use, minimal training [25] | Cannot quantify parasites [62] |
| PCR | High (reference standard) [25] [62] | High (reference standard) [25] [62] | Several hours | High sensitivity for low parasitemia [62] | Expensive, requires lab infrastructure [62] |
| Hemozoin-Based Detection (Hz-MOD) | 91.7%-94.2% [62] | 97.8%-98.6% [62] | ~3 minutes [62] | Rapid, detects hemozoin [62] | Emerging technology [62] |
| AI-Driven Microscopy Analysis | Up to 96.5% [2] | Up to 96.9% [2] | Minutes (after digitization) | Automated, high-throughput [3] | Requires digital infrastructure [3] |
Table 2: Recent Field Performance of Specific RDTs and Newer Technologies
| Assay | Study Location | Sensitivity vs. Reference | Specificity vs. Reference | Notes |
|---|---|---|---|---|
| Abbott Bioline Malaria Ag P.f/Pan RDT [25] | Central Cameroon | 93.4% (vs. Microscopy) [25] | 68.5% (vs. Microscopy) [25] | Recommended by national program; lower specificity noted [25] |
| Abbott Bioline Malaria Ag P.f/Pan RDT [62] | Southwestern India | 87.6% (vs. PCR) [62] | 98.9% (vs. PCR) [62] | Targets HRP-2 and pLDH [62] |
| Hemozoin-Based Magneto-Optical Detection (Hz-MOD) [62] | Southwestern India | 91.7% (vs. PCR) [62] | 97.8% (vs. PCR) [62] | Gazelle device; results in 3 minutes [62] |
| AI for RDT Interpretation (ConnDx System) [63] | Western Kenya | 96.1% (vs. Expert Panel) [63] | 98.0% (vs. Expert Panel) [63] | Uses smartphone app and cloud-based AI [63] |
Detailed Experimental Protocols
Protocol 1: Standardized Evaluation of RDT Performance Against Microscopy and PCR
This protocol outlines the procedure for assessing the diagnostic performance of a malaria RDT in a field setting, using both microscopy and PCR as reference standards [25] [62].
Materials and Equipment
- Test RDTs (e.g., Abbott Bioline Malaria Ag P.f/Pan)
- Microscopy supplies: Glass slides, Leishman's stain, immersion oil, light microscope
- Molecular biology supplies: Venous blood collection tubes (EDTA), DNA extraction kit (e.g., QIAamp DNA Blood Mini Kit), PCR reagents, nested PCR primers for 18S rRNA gene
- Personal protective equipment (PPE)
- Data collection forms
Procedural Steps
Study Participant Enrollment and Sample Collection:
Parallel Diagnostic Testing:
- RDT Performance: Perform RDT according to manufacturer's instructions using fresh whole blood. Interpret results within specified time window [25] [62].
- Blood Smear Preparation and Microscopy:
- Prepare thin and thick blood smears from the collected sample.
- Stain slides with Leishman's stain (or Giemsa).
- Examine smears under oil immersion (1000x magnification) by two experienced microscopists blinded to RDT and PCR results.
- Examine a minimum of 200 fields before declaring a slide negative [62].
- Calculate parasite density per μL based on assumed white blood cell count (e.g., 8,000/μL) [62].
- Molecular Confirmation by Nested PCR:
- Extract genomic DNA from a portion of the venous blood sample.
- Perform genus-specific PCR amplification followed by species-specific nested PCR targeting the 18S rRNA gene.
- Visualize amplification products on a 1.5% agarose gel with EtBr staining [62].
Data Analysis:
Protocol 2: AI-Assisted Analysis of Blood Smears Using a Hybrid Capsule Network
This protocol describes a methodology for training and validating a deep learning model for automated detection and classification of malaria parasites from thin blood smear images [3].
Materials and Equipment
- Microscopy system with digital camera or whole slide scanner
- Computational resources: GPU workstation
- Software: Python with deep learning libraries (e.g., TensorFlow, PyTorch)
- Datasets: Publicly available malaria image datasets (e.g., MP-IDB, IML-Malaria, MD-2019) [3]
Procedural Steps
Image Acquisition and Preprocessing:
- Create a digital library of thin blood smear images from Giemsa-stained slides.
- Annotate images for parasite species and life-cycle stages (ring, trophozoite, schizont, gametocyte) [3].
- Preprocess images: normalization, resizing, and data augmentation (rotation, flipping) to increase dataset size and variability.
Model Architecture and Training (Hybrid CapNet):
- Feature Extraction: Employ a lightweight Convolutional Neural Network (CNN) as a feature extractor from input images [3].
- Capsule Network Integration: Feed extracted features into a Capsule Network (CapsNet) to capture spatial hierarchies and relationships between features, improving robustness to orientation and pose variations [3].
- Composite Loss Function: Implement a novel loss function integrating margin loss, focal loss (for class imbalance), reconstruction loss, and regression loss to enhance classification accuracy and spatial localization [3].
- Model Training: Train the Hybrid CapNet model on the annotated dataset, using a standard split (e.g., 80% training, 20% validation).
Model Validation and Interpretation:
- Performance Evaluation: Test the model on a held-out test set and/or through cross-dataset validation. Report standard metrics: accuracy, sensitivity, specificity, precision, F1-score [3].
- Interpretability Analysis: Apply Grad-CAM (Gradient-weighted Class Activation Mapping) visualizations to confirm that the model focuses on biologically relevant regions of the parasite within the red blood cells [3].
The workflow for this AI-based diagnostic framework is summarized in the diagram below.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Malaria Diagnostic Research
| Item | Specification / Example | Primary Function in Research Context |
|---|---|---|
| Rapid Diagnostic Tests | Abbott Bioline Malaria Ag P.f/Pan [25] [62] | Field-based immunochromatographic detection of HRP-2 (P. falciparum) and pLDH (Pan/ P. vivax) antigens. |
| Microscopy Stains | Leishman's Stain, Giemsa Stain [62] | Staining of thin and thick blood films to visualize malaria parasite morphology and species identification. |
| DNA Extraction Kit | QIAamp DNA Blood Mini Kit [62] | Extraction of high-quality genomic DNA from whole blood samples for subsequent molecular analysis. |
| PCR Primers | 18S rRNA genus & species-specific primers [62] | Amplification of conserved Plasmodium gene regions for highly sensitive detection and species differentiation via nested PCR. |
| Hemozoin Detection Device | Gazelle (Hemex Health) [62] | Magneto-optical detection of hemozoin crystals for rapid, non-invasive malaria diagnosis. |
| AI/ML Analysis Platform | HealthPulse App & ConnDx System [63] | Mobile application and cloud-based AI for standardized interpretation of RDT images and real-time data aggregation. |
| Deep Learning Model | Hybrid Capsule Network (Hybrid CapNet) [3] | Advanced neural network for automated, high-accuracy classification of parasite species and life-cycle stages from digital smear images. |
The diagnostic landscape for malaria is undergoing a significant transformation, moving from a reliance on a single gold standard method toward an integrated, multi-tool approach. While thick blood smear microscopy remains an essential and irreplaceable tool, particularly for quantifying parasitemia and species identification, its limitations are being addressed by complementary technologies. RDTs offer rapid point-of-care testing, PCR provides ultra-sensitive confirmation, and AI-driven solutions bring new levels of automation, accuracy, and data connectivity to both RDT interpretation and smear analysis.
The future of malaria diagnosis and surveillance lies in the strategic deployment of these technologies based on the specific context—whether in a high-throughput research laboratory, a remote clinic, or a community-based screening program. The ongoing development and refinement of these tools, as evidenced by the recent research outlined in these application notes, are critical for achieving the global goals of malaria control and eventual elimination.
Despite being the gold standard for malaria diagnosis, thick blood smear microscopy faces significant challenges that can impact its effectiveness in both routine clinical practice and research settings. These limitations include high labor intensity, inherent subjectivity, and the emerging threat of Plasmodium falciparum genetic deletions that evade detection [64]. For researchers and drug development professionals, these constraints introduce variability in endpoint measurements and complicate the accurate assessment of parasite clearance in clinical trials. This application note provides a detailed framework for identifying, quantifying, and addressing these limitations through integrated protocols and quality control measures, ensuring the generation of reliable, reproducible data in malaria research.
The table below summarizes key performance metrics and the prevalence of factors that compromise thick blood smear microscopy and Rapid Diagnostic Tests (RDTs).
Table 1: Performance Metrics and Prevalence of Diagnostic Challenges
| Parameter | Value | Context / Location | Citation |
|---|---|---|---|
| Microscopy Sensitivity | 77% | Compared to species-specific qPCR in Nigeria | [65] |
| Microscopy Specificity | 44% | Compared to species-specific qPCR in Nigeria | [65] |
| HRP2-based RDT Sensitivity | 95% | Compared to species-specific qPCR in Nigeria | [65] |
| HRP2-based RDT Specificity | 68% | Compared to species-specific qPCR in Nigeria | [65] |
| False Negative HRP2-RDTs | 4.61% (16/347) | Pacific Coast, Colombia | [66] |
| Prevalence of pfhrp2 deletion | 7.29% | Cubal, Angola | [67] |
| Prevalence of pfhrp3 deletion | 11.46% | Cubal, Angola | [67] |
| Prevalence of pfhrp2/3 double deletion | 0% | Cubal, Angola | [67] |
| Countrywide pfhrp2/3 deletion prevalence | 22% | Ethiopia | [68] |
| WHO Threshold for RDT Change | >5% | Prevalence of pfhrp2/3 deletions | [66] [68] |
Protocol for Investigating pfhrp2/3 Gene Deletions
Background and Principle
Deletions in the pfhrp2 and pfhrp3 genes, which code for the histidine-rich proteins 2 and 3, allow P. falciparum parasites to escape detection by HRP2-based RDTs [67] [69]. This represents a significant threat to malaria control. The World Health Organization (WHO) recommends a threshold of 5% prevalence for pfhrp2/3 deletions to trigger a change in national diagnostic strategy from HRP2-based RDTs to alternatives [66] [68]. The following protocol follows WHO guidelines to investigate these deletions in a research context.
Materials and Equipment
- Patient Samples: Blood samples (via finger prick or venipuncture) from confirmed P. falciparum patients, collected on filter paper (Whatman 3MM) or in EDTA tubes [66] [68].
- Diagnostic Tools: Microscopy supplies (slides, Giemsa stain) and HRP2-based RDTs (e.g., STANDARD Q Malaria Pf/Pan Ag test) [66] [67].
- Molecular Biology Reagents:
- DNA extraction kit (e.g., QIAamp DNA Blood Kit, Qiagen) [70] [68].
- PCR reagents (primers, polymerase, dNTPs, buffer).
- Primers for pfhrp2 and pfhrp3 (targeting exons 1-2 and exon 2), and a control gene (e.g., P. falciparum serine tRNA ligase, pfcrt) [67] [68].
- Agarose gel electrophoresis equipment.
- (Optional) Droplet Digital PCR (ddPCR) system for quantitative analysis of mixed infections [68].
Step-by-Step Procedure
Sample Collection and Initial Diagnosis:
- Collect blood samples from patients with suspected malaria.
- Perform parallel diagnosis using both light microscopy and an HRP2-based RDT for all participants [66].
- Include only patients with microscopy-confirmed P. falciparum monoinfection and parasitemia ≥ 200 parasites/µL. Exclude mixed infections and cases of severe malaria [66].
Identification of Discordant Results:
- Identify samples where microscopy is positive for P. falciparum, but the HRP2-RDT gives a negative result (false-negative RDT) [66]. These samples are candidates for gene deletion analysis.
Molecular Confirmation and DNA Extraction:
- Confirm the presence of P. falciparum DNA in all discordant samples (and a subset of concordant positives for quality control) using a species-specific PCR [66].
- Extract genomic DNA from blood samples preserved on filter paper or in EDTA using a commercial DNA extraction kit, following the manufacturer's instructions [68].
PCR Amplification of Target Genes:
- Perform multiplex or singleplex PCR reactions to amplify regions of the pfhrp2 and pfhrp3 genes. A common approach is to amplify two regions: exon 1-2 and exon 2 for each gene [67].
- Include control reactions to amplify a conserved single-copy gene (e.g., pfcrt or serine tRNA ligase) to confirm the quality of the DNA and the presence of P. falciparum [67] [68].
- Use validated positive controls (e.g., strain 3D7 for wild-type genes) and negative controls (nuclease-free water) in each PCR run [68].
Analysis of PCR Products:
- Visualize PCR amplicons on a 1.5% agarose gel stained with an intercalating dye [67].
- Interpret the results as follows:
- Gene Present: A band of the expected size is observed.
- Gene Deleted: No band is observed, but the control gene amplifies successfully.
- A sample is considered to have a pfhrp2 or pfhrp3 deletion only if the control gene amplifies, confirming the presence of amplifiable parasite DNA [67].
Data Analysis and Reporting:
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Malaria Diagnostic Research
| Item | Function / Application | Example / Note |
|---|---|---|
| Giemsa Stain | Staining thin and thick blood films for microscopic visualization of parasites. | Standard for parasite species identification and staging [64]. |
| HRP2-based RDT | Rapid immunochromatographic detection of P. falciparum HRP2 antigen. | Used to identify false-negative results suggestive of pfhrp2/3 deletions (e.g., STANDARD Q Malaria Pf/Pan Ag) [66] [67]. |
| DNA Extraction Kit | Isolation of high-quality genomic DNA from whole blood or dried blood spots. | Critical for downstream molecular assays (e.g., QIAamp DNA Blood Kit) [70] [68]. |
| PCR Primers | Amplification of specific P. falciparum gene targets. | Targets include pfhrp2 (exons 1-2, exon 2), pfhrp3 (exons 1-2, exon 2), and control genes (pfcrt, tRNA ligase) [67] [68]. |
| Droplet Digital PCR (ddPCR) | Absolute quantification of gene copy number; detects deletions in polyclonal infections. | More accurately quantifies the proportion of deleted parasites in a sample than conventional PCR [68]. |
| Positive Control Genomic DNA | Control for PCR efficiency and specificity. | WHO-recommended strains: 3D7 (wild-type), Dd2 (Δpfhrp2), Hb3 (Δpfhrp3) [68]. |
Addressing Labor Intensity and Subjectivity
Automated Microscopy and AI
Labor-intensive manual reading and subjective interpretation are major bottlenecks. Deep learning (DL) and artificial intelligence (AI) models offer a solution by automating the detection and quantification of parasites in digital images of blood smears [64].
- Workflow: Digital images of Giemsa-stained thick and thin smears are captured using microscope-mounted cameras or smartphone adapters. These images are then analyzed by pre-trained convolutional neural networks (CNNs) that can identify infected erythrocytes and differentiate parasite stages [64].
- Impact: This automation provides a fast, low-cost diagnostic aid that requires less supervision, reduces inter-observer variability, and can operate in resource-poor settings via smartphone applications [64].
Quality Control in Microscopy
Implementing rigorous quality control (QC) procedures is essential to mitigate subjectivity.
- Procedure: A standard operating procedure should include:
- Slide Preparation: Standardized preparation of thick and thin films.
- Staining: Use of fresh, filtered Giemsa stain following WHO protocols [64].
- Examination: Examination of a minimum number of microscopic fields (e.g., 200 fields at 100x oil immersion) before declaring a sample negative [65].
- Cross-Checking: All slides should be read by two independent, trained microscopists blinded to each other's results and to the RDT outcome. A third reader serves as a tie-breaker in case of discordance [66] [65].
Conclusion
Thick blood smear microscopy remains an indispensable tool in malaria diagnosis and research, offering unparalleled sensitivity, the ability to quantify parasitemia, and confirm species identity—all critical for clinical management and drug efficacy studies. However, its value is maximized when integrated into a broader diagnostic ecosystem. The future of malaria diagnostics lies in hybrid models that leverage the proven utility of microscopy while incorporating the speed of RDTs and the exquisite sensitivity and specificity of molecular and AI-powered techniques. For researchers and drug developers, this synergy presents a pathway to more robust clinical trial endpoints, enhanced surveillance for drug resistance, and the creation of field-deployable, automated systems that preserve the diagnostic power of microscopy while overcoming its operational constraints.
References
- 1. Malaria Workup: Approach Considerations, Blood Smears, ... [https://emedicine.medscape.com/article/221134-workup]
- 2. Automated multi-model framework for malaria detection ... [https://www.nature.com/articles/s41598-025-04784-w]
- 3. Hybrid Capsule Network for precise and interpretable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12370641/]
- 4. Preparing Blood Smears [https://www.cdc.gov/dpdx/diagnosticprocedures/blood/specimenproc.html]
- 5. Blood smear imagery dataset for malaria parasite detection [https://pmc.ncbi.nlm.nih.gov/articles/PMC11648091/]
- 6. Malaria parasite detection in thick blood smear microscopic ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-021-04036-4]
- 7. Deep learning-based malaria parasite detection [https://www.nature.com/articles/s41598-025-87979-5]
- 8. Microscopy [https://www.who.int/teams/global-malaria-programme/case-management/diagnosis/microscopy]
- 9. Giemsa staining of malaria blood films [https://www.who.int/publications/i/item/HTM-GMP-MM-SOP-07a]
- 10. Malaria Exam Thick & Thin Smears and Immunoassay [https://dlmp.uw.edu/test-guide/view/MALP]
- 11. Comparative performance of microscopy, rapid diagnostic ... [https://malariajournal.biomedcentral.com/articles/10.1186/s12936-025-05256-2]
- 12. Enhanced detection of malaria infected red blood cells ... [https://www.nature.com/articles/s41598-025-12899-3]
- 13. Improved light microscopy counting method for accurately ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5814318/]
- 14. Sensitive and accurate quantification of human malaria ... [https://www.nature.com/articles/srep39183]
- 15. A validation study of microscopy versus quantitative PCR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6712708/]
- 16. Malaria Diagnostic Tests [https://www.cdc.gov/malaria/hcp/diagnosis-testing/malaria-diagnostic-tests.html]
- 17. Current methods for the detection of Plasmodium parasite ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9006665/]
- 18. Rapid and accurate recognition of erythrocytic stage ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1471436/full]
- 19. DPDx - Malaria [https://www.cdc.gov/dpdx/malaria/index.html]
- 20. Blood Specimens – Microscopic Examination [https://www.cdc.gov/dpdx/diagnosticprocedures/blood/microexam.html]
- 21. Malaria diagnostic update: From conventional to advanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8993657/]
- 22. 6 tips to optimize your staining in hematology [https://www.cellavision.com/about/news/6-tips-optimize-your-staining-hematology]
- 23. Smear examination [https://eclinpath.com/hematology/hemogram-basics/blood-smear-examination/]
- 24. Utility of rapid diagnostic tests and microscopy to detect ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12235921/]
- 25. Malaria infection prevalence and diagnostic performance ... [https://www.nature.com/articles/s41598-025-13688-8]
- 26. Performance and Challenges of Malaria Rapid Diagnostic ... [https://www.medrxiv.org/content/10.1101/2025.07.30.25331638v1.full-text]
- 27. Update on Malaria Diagnostics and Test Utilization - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5483902/]
- 28. Malaria parasite identification and quantification by ... [https://bio-protocol.org/exchange/minidetail?id=4085734&type=30]
- 29. How to identify the type of malaria on a blood smear [https://www.medmastery.com/guides/malaria-clinical-guide/how-identify-type-malaria-blood-smear?srsltid=AfmBOoromzvUaNuZ0JC5ER1wzY-t0ruC_AE0A0D_K-eMHcPZRPn5eeR8]
- 30. Rapid Malaria/Babesia Smear, Varies [https://pediatric.testcatalog.org/show/MAL]
- 31. Quantitative Determination of Plasmodium Parasitemia by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3468747/]
- 32. A portable brightfield and fluorescence microscope toward ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0266441]
- 33. comparing microscopy, RDT and a highly sensitive ... [https://malariajournal.biomedcentral.com/articles/10.1186/s12936-023-04506-5]
- 34. Thick blood film examination for Plasmodium falciparum ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1636647/]
- 35. P. vivax (malaria) infected human blood smears [https://bbbc.broadinstitute.org/BBBC041]
- 36. A dataset of blood slide images for AI-based diagnosis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11719325/]
- 37. A deep architecture based on attention mechanisms for ... [https://www.sciencedirect.com/science/article/pii/S001048252500054X]
- 38. Image features for quality analysis of thick blood smears ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8900337/]
- 39. Supporting Malaria Diagnosis Using Deep Learning and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11012121/]
- 40. Technique and Tips for Making Perfect Blood Smears [https://bitesizebio.com/48369/blood-smear/]
- 41. Staining Blood Smears [https://www.cdc.gov/dpdx/diagnosticprocedures/blood/staining.html]
- 42. Quality thick films for malaria diagnosis [https://thebiomedicalscientist.net/2018/11/01/quality-thick-films-malaria-diagnosis]
- 43. Impact of malaria diagnostic refresher training programme on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8186098/]
- 44. Factors associated with malaria microscopy diagnostic ... [https://malariajournal.biomedcentral.com/articles/10.1186/s12936-017-2018-2]
- 45. A method for reducing the sloughing of thick blood films for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3734161/]
- 46. Evaluating ECAMM-based training efficacy for malaria ... [https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2025.1598917/full]
- 47. Performance evaluation of laboratory professionals on malaria ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4255633/]
- 48. Proficiency testing for HIV, tuberculosis and malaria ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5637760/]
- 49. Performance Assessment of Laboratory Professionals on ... [https://www.researchsquare.com/article/rs-6922700/latest]
- 50. Proficiency testing (PT) [https://bio-protocol.org/exchange/minidetail?id=7047312&type=30]
- 51. Quantitative Data Quality Assurance, Analysis and Presentation [https://pmc.ncbi.nlm.nih.gov/articles/PMC12056448/]
- 52. Improving the Specificity of Plasmodium falciparum Malaria ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5405272/]
- 53. Validation rules for blood smear revision after automated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6817000/]
- 54. Clinical validation and evaluation of the EasyNAT Malaria ... [https://www.sciencedirect.com/science/article/pii/S1477893925000365]
- 55. Sensitivity and specificity for malaria classification of febrile ... [https://www.sciencedirect.com/science/article/pii/S1201971222002594]
- 56. A new approach for malaria diagnosis in thick blood smear ... [https://www.sciencedirect.com/science/article/abs/pii/S1746809422004347]
- 57. A Diagnostic Accuracy Study to Evaluate Standard Rapid ... [https://pubmed.ncbi.nlm.nih.gov/37070464/]
- 58. Hematology thin smears perform equally to parasitology ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12077176/]
- 59. Clinical performance testing of the automated haematology ... [https://malariajournal.biomedcentral.com/articles/10.1186/s12936-022-04247-x]
- 60. Rapid and accurate recognition of erythrocytic stage parasites ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12611930/]
- 61. A rapid review of artificial intelligence for the peripheral ... [https://www.sciencedirect.com/science/article/abs/pii/S0268960X23001054]
- 62. Diagnostic performance of hemozoin-based magneto ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12642893/]
- 63. Artificial intelligence (AI)-driven rapid diagnostic test ... [https://bmcdigitalhealth.biomedcentral.com/articles/10.1186/s44247-025-00197-x]
- 64. Advances and challenges in automated malaria diagnosis ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2022.1006659/full]
- 65. Evaluating Malaria Rapid Diagnostic Tests and Microscopy for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11066366/]
- 66. Prevalence of pfhrp2/pfhrp3 gene deletions among patients with Plasmodium falciparum malaria with false-negative in the HRP2-based rapid diagnostic test in Colombia - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12165712/]
- 67. Analysis of pfhrp2 and pfhrp3 gene deletions, and the structure and variability of PfHRP2 and PfHRP3 proteins: implications for the performance of malaria rapid diagnostic tests in Cubal, Angola | Malaria Journal | Full Text [https://malariajournal.biomedcentral.com/articles/10.1186/s12936-025-05523-2]
- 68. A Countrywide Survey of hrp2/3 Deletions and kelch13 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11646589/]
- 69. Global risk of selection and spread of Plasmodium falciparum histidine-rich protein 2 and 3 gene deletions | Nature Medicine [https://www.nature.com/articles/s41591-025-03974-3]
- 70. Challenges in Laboratory Diagnosis of Malaria in a Low ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8727160/]