INVAPP and Paragon: Revolutionizing High-Throughput Chemical Screening for Parasite and Vector Control
This article details the INVertebrate Automated Phenotyping Platform (INVAPP) combined with the Paragon algorithm, a robust system transforming high-throughput screening for novel anthelmintics and larvicides.
INVAPP and Paragon: Revolutionizing High-Throughput Chemical Screening for Parasite and Vector Control
Abstract
This article details the INVertebrate Automated Phenotyping Platform (INVAPP) combined with the Paragon algorithm, a robust system transforming high-throughput screening for novel anthelmintics and larvicides. It explores the platform's foundational technology, which quantifies motility and growth in parasitic nematodes and mosquito larvae with a throughput of approximately 100 ninety-six-well plates per hour. The content covers its methodological application in library-scale chemical screening and resistance monitoring, provides insights for troubleshooting and optimizing the automated phenotyping workflow, and validates its performance against standard assays. Aimed at researchers and drug development professionals, this resource underscores INVAPP's capacity to accelerate the discovery of new therapeutic compounds against globally significant parasitic diseases and insect vectors.
Understanding INVAPP and Paragon: Core Technology and Its Role in Addressing Global Health Challenges
The Urgent Need for Novel Insecticides and Anthelmintics
Parasitic nematodes and resistant insect vectors pose a significant and growing threat to global health and food security. Infections caused by parasitic helminths affect hundreds of millions of people worldwide, while plant-parasitic nematodes and insect pests result in major crop damage and substantial economic losses in animal agriculture [1] [2]. The situation is exacerbated by the rapid and widespread development of resistance to existing chemical treatments. In livestock nematodes, multi-class resistance is now common, and the efficacy of mass drug administration programs for human helminthiases is increasingly compromised by anthelmintic resistance [1] [3]. Similarly, pyrethroid resistance in mosquito vectors threatens the remarkable progress made in reducing malaria mortality through insecticide-treated nets [4] [5]. This urgent scenario necessitates the accelerated discovery and development of novel chemical interventions with new mechanisms of action.
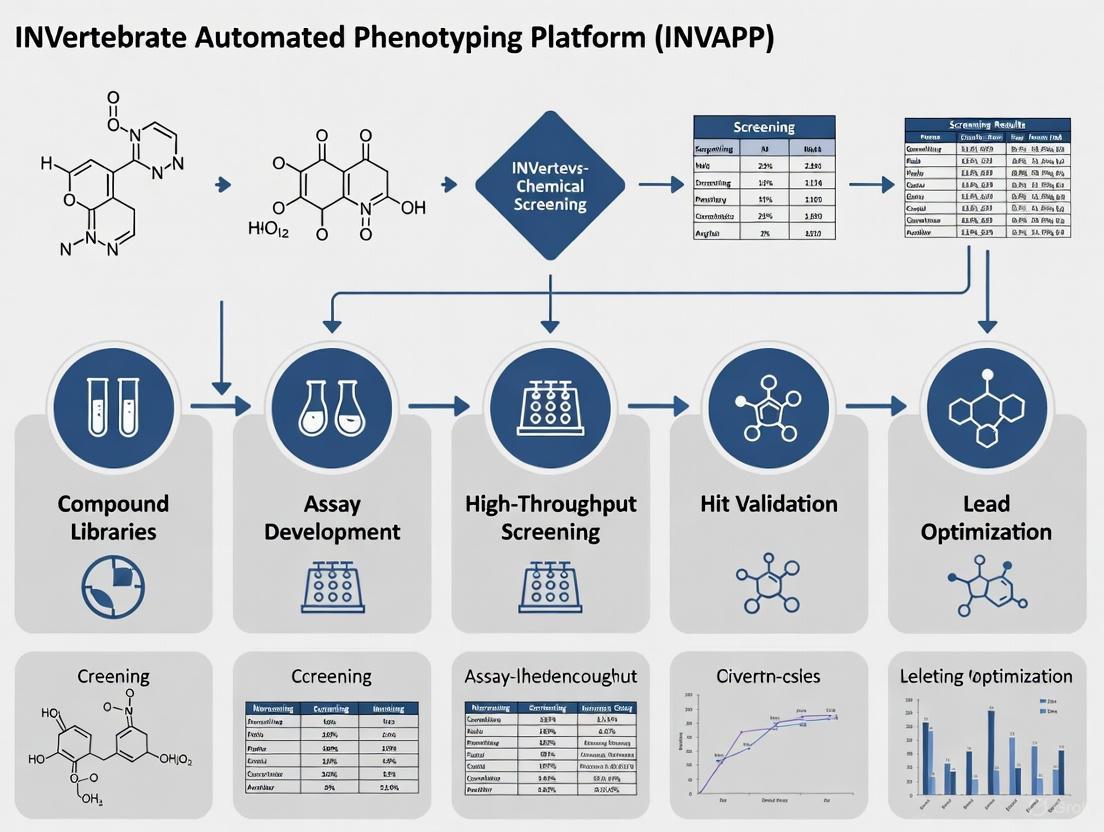
The INVertebrate Automated Phenotyping Platform (INVAPP) represents a transformative technological advancement for high-throughput chemical screening in the search for new insecticides and anthelmintics. When combined with the Paragon analysis algorithm, this system enables rapid, quantitative assessment of compound effects on invertebrate motility and development at a scale and speed previously unattainable with traditional manual methods [1] [4]. This application note details the implementation, validation, and application of the INVAPP/Paragon system for high-throughput phenotypic screening of chemical libraries to identify novel bioactive compounds against nematode and insect targets.
The INVAPP system utilizes a fast high-resolution camera (Andor Neo, resolution 2560×2160, maximum frame rate 100 frames per second) with a line-scan lens (Pentax YF3528) for imaging microtiter plates from below, with illumination provided by an LED panel with acrylic diffuser [1]. The system captures movies of invertebrate movement in multi-well plates, typically recording 200 frames at 25 frames per second for 8 seconds per well. The accompanying Paragon algorithm, implemented in MATLAB, analyzes these movies by calculating the variance through time for each pixel. Pixels whose variance exceeds a set threshold (typically those greater than one standard deviation away from the mean variance) are classified as 'motile' [1] [3]. These motile pixels are then assigned to individual wells and counted, generating a quantitative movement score for each well that serves as a robust metric of invertebrate viability and activity.
Table 1: Key Technical Specifications of the INVAPP/Paragon System
| Parameter | Specification | Application Benefit |
|---|---|---|
| Throughput | ~100 ninety-six-well plates/hour | Enables library-scale chemical screening |
| Imaging Resolution | 2560×2160 pixels | Precise detection of small organism movement |
| Assay Readout | Quantitative motility scores | Objective, reproducible phenotypic data |
| Organisms Validated | C. elegans, parasitic nematodes, mosquito larvae | Broad applicability across invertebrate targets |
| Data Analysis | Automated Paragon algorithm | Eliminates subjective manual scoring |
Application Note 1: Anthelmintic Discovery Using C. elegans
Protocol: High-Throughput Screening of Chemical Libraries
Experimental Workflow:
Step-by-Step Methodology:
C. elegans Maintenance and Synchronization:
- Maintain C. elegans (Bristol N2 strain) at 20°C on nematode growth medium (NGM) agar seeded with E. coli OP50 [6] [1].
- Prepare synchronized L1 populations by bleaching mixed-stage cultures using bleaching mix (1.5 mL 4M NaOH, 2.4 mL NaOCl, 2.1 mL water) for 4 minutes to release embryos [1].
- Wash liberated embryos three times with S-basal medium and incubate in S-basal at 20°C to obtain synchronized L1 larvae.
- For L4 synchronization, filter worms through 100 μm filters to capture L4 stage larvae while removing earlier stages [3].
Assay Plate Preparation and Optimization:
- Use clear, flat-bottomed 96-well polystyrene plates for optimal imaging.
- Spot 1 μL of test compounds in DMSO into each well. Include DMSO-only controls (1% final concentration).
- Add approximately 70 L4 larvae in 100 μL S medium per well. This worm density provides optimal dynamic range while maintaining assay economy [6].
- Maintain final DMSO concentration at 1% to balance compound solubility with minimal solvent effects on worm motility [6].
INVAPP Data Acquisition and Analysis:
- Capture movies using μManager software, acquiring 200 frames at 25 frames per second for 8 seconds per well [1] [3].
- Analyze movies using the Paragon algorithm, which calculates pixel variance through time and identifies motile pixels exceeding one standard deviation from mean variance.
- Normalize motility scores to DMSO controls (100% motility) and identify hits as compounds reducing motility to ≤25% of control values [6].
Hit Validation and Concentration-Response Analysis:
- For confirmed hits, perform concentration-response assays using serial compound dilutions (typically 9 concentrations from 0.005 μM to 40 μM) [6].
- Calculate half-maximal effective concentration (EC50) values using non-linear sigmoidal four-parameter logistic curve fitting in Prism GraphPad.
- Counter-screen against mammalian cells (e.g., HEK293 cytotoxicity assay) to assess selectivity indices [6].
Representative Data: MMV Library Screening
Screening of the Medicines for Malaria Venture (MMV) COVID Box and Global Health Priority Box (400 compounds total) identified multiple compounds with significant anthelmintic activity [6] [3]. The following table summarizes representative hits from these screens:
Table 2: Anthelmintic Hits Identified from MMV Library Screens Using INVAPP
| Compound | EC50 (μM) | Motility Reduction | Known Activity | Cytotoxicity (HEK293) |
|---|---|---|---|---|
| Tolfenpyrad | Not reported | 0.26% (vs control) | Insecticide (Complex I inhibitor) | Varying toxicity [6] |
| Flufenerim | 0.211 - 23.174 | Significant | Novel anthelmintic | CC50: 0.453 to >100 μM [6] |
| Flucofuron | 0.211 - 23.174 | Significant | Novel anthelmintic | CC50: 0.453 to >100 μM [6] |
| Indomethacin | 0.211 - 23.174 | Significant | NSAID | CC50: 0.453 to >100 μM [6] |
| Vorapaxar | 0.57 | Significant | PAR-1 antagonist | Not reported [3] |
| Macrocyclic Lactones | Varying | 0.28-13.19% | Known anthelmintics | Validation compounds [6] |
Application Note 2: Larvicide Screening in Mosquitoes
Protocol: Mosquito Larval Motility Assay
Experimental Workflow:
Step-by-Step Methodology:
Mosquito Rearing and Larval Preparation:
Compound Exposure and Motility Recording:
- Add test compounds in DMSO, maintaining appropriate solvent controls.
- Capture larval motility using INVAPP as described for nematodes.
- Compare results to standard WHO larvicidal toxicity assays for validation [5].
Data Analysis and Hit Identification:
- Analyze concentration-dependent effects on larval motility.
- Calculate EC50 values for active compounds.
- Compare responses in insecticide-resistant and sensitive strains to identify potential resistance mechanisms.
Representative Data: Pathogen Box Screening
Screening of the MMV Pathogen Box library using the INVAPP/Paragon system identified tolfenpyrad as a potent larvicide, demonstrating the platform's utility for discovering new insecticide chemotypes [5]. The system also detected differential responses in larval progeny from deltamethrin-resistant and sensitive mosquitoes, indicating its potential for resistance monitoring. The assay provides results faster than standard WHO larval assays and offers quantitative motility data rather than binary mortality endpoints [5].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for INVAPP-Based Chemical Screening
| Reagent/Resource | Specification | Application | Source/Reference |
|---|---|---|---|
| INVAPP/Paragon System | High-speed camera + MATLAB algorithm | Automated phenotyping of motility | [1] |
| C. elegans N2 | Wild-type strain | Primary screening model | [6] [1] |
| Mosquito Larvae | An. gambiae, Ae. aegypti | Larvicide screening | [4] [5] |
| MMV Chemical Libraries | Pandemic, Pathogen, COVID Boxes | Source of drug-like compounds | [6] [3] |
| S Complete Buffer | Nematode maintenance medium | Liquid culture assays | [1] |
| 96-well Plates | Clear, flat-bottomed | Optimal for imaging | [6] |
| WMicroTracker | Alternative motility system | Validation studies | [6] |
Technical Considerations and Optimization
Assay Optimization Parameters:
- Worm/Larval Density: 70 L4 C. elegans per well provides optimal signal-to-noise ratio while maintaining throughput [6].
- DMSO Concentration: Maintain at ≤1% final concentration to minimize solvent effects on motility [6].
- Imaging Parameters: 200 frames at 25 frames per second captures sufficient movement data for robust quantification [1].
- Temperature Control: Maintain consistent temperature during imaging (25±1°C for C. elegans) [6].
Validation and Secondary Assays:
- Confirm activity against parasitic nematodes (Haemonchus contortus, Teladorsagia circumcincta) following primary C. elegans screens [1].
- Perform cytotoxicity counter-screens using mammalian cell lines (HEK293) to determine selectivity indices [6].
- Compare results with established reference anthelmintics (macrocyclic lactones, benzimidazoles) for benchmarking [6].
The INVAPP/Paragon platform represents a significant advancement in high-throughput phenotypic screening for anthelmintic and insecticide discovery. Its ability to provide quantitative, reproducible motility data for diverse invertebrates enables rapid identification of novel bioactive compounds from chemical libraries. The system's validation against known anthelmintics and larvicides, combined with its successful application to MMV chemical libraries, demonstrates its robust performance for drug discovery campaigns. Furthermore, the adaptability of the platform for smartphone-based imaging offers potential for field-based resistance monitoring in resource-limited settings [5]. As resistance to existing insecticides and anthelmintics continues to escalate, this automated phenotyping approach provides a powerful tool for accelerating the discovery of next-generation chemical interventions.
The INVertebrate Automated Phenotyping Platform (INVAPP), coupled with the Paragon algorithm, represents a technological advancement in whole-organism phenotypic screening for chemical compound discovery [1]. This automated system was developed to address urgent needs in global health, particularly the accelerating threat of anthelmintic resistance in parasitic nematodes and insecticide resistance in mosquito vectors of deadly diseases [1] [7]. Traditional manual methods for assessing invertebrate viability and motility are labor-intensive, low-throughput, and subject to observer bias, creating bottlenecks in drug discovery pipelines [8]. INVAPP/Paragon overcomes these limitations by providing a robust, quantitative system for measuring motility and growth in microscopic nematodes and mosquito larvae with significantly higher throughput than conventional methods [1] [7]. By enabling rapid screening of large chemical libraries, this platform accelerates the identification of novel anthelmintic and larvicidal compounds, offering new hope for controlling neglected tropical diseases and combating resistance [3].
The INVAPP/Paragon system integrates specialized hardware for image acquisition with sophisticated software for motion analysis, creating a complete solution for automated phenotypic screening.
INVAPP Hardware Components
The INVAPP hardware platform is designed specifically for high-resolution imaging of small invertebrates in multi-well plate formats [1]. The core components include:
- A fast high-resolution camera (Andor Neo, resolution 2560×2160 pixels) capable of capturing up to 100 frames per second [1]
- A line-scan lens (Pentax YF3528) optimized for clear imaging of small, moving organisms [1]
- An LED panel with acrylic diffuser providing uniform illumination for consistent imaging conditions [1]
- A custom-built cabinet with microtiter plate holder for stable imaging from below [1]
- Adaptability to smartphone cameras for potential field applications, demonstrated in mosquito larvae studies [7] [5]
Paragon Analysis Algorithm
The Paragon algorithm analyzes acquired image data to quantify motility through a variance-based thresholding approach [1] [3]. The processing workflow involves:
- Movie capture: Typically 200 frames at 25 frames per second for 8 seconds using μManager software [3]
- Pixel variance calculation: Determining variance through time for each pixel [1]
- Threshold application: Identifying "motile pixels" as those with variance greater than one standard deviation from the mean variance [1]
- Pixel counting: Assigning motile pixels by well and generating a movement score for each well [1]
This algorithm has been released as open-source software under the MIT license, making it accessible to the research community [1].
Applications in Anthelmintic Discovery
Validation with Known Anthelmintics
The INVAPP/Paragon system was initially validated by quantifying the effects of established anthelmintic compounds on various nematode species, including Caenorhabditis elegans, Haemonchus contortus, Teladorsagia circumcincta, and Trichuris muris [1]. These validation studies demonstrated the system's ability to reliably detect concentration-dependent effects on nematode motility and growth, confirming its utility for anthelmintic discovery [1].
Table 1: Key Performance Metrics for INVAPP/Paragon in Nematode Screening
| Parameter | Specification | Experimental Context |
|---|---|---|
| Throughput | ~100 ninety-six-well plates per hour [1] | High-throughput chemical screening |
| Assay Types | L1-L4 growth/motility, L4 motility [3] | C. elegans developmental assays |
| Key Output | Movement index (based on motile pixels) [1] | Quantification of nematode viability |
| Validation | Known anthelmintics (e.g., mebendazole) [1] | Concentration-response curves |
Chemical Library Screening
The system's capacity for library-scale screening was demonstrated through blinded screening of the Medicines for Malaria Venture (MMV) Pathogen Box library [1]. This effort successfully identified:
- Compounds with known anthelmintic or anti-parasitic activity (tolfenpyrad, auranofin, mebendazole) [1]
- Fourteen compounds previously not described as anthelmintics, including benzoxaborole and isoxazole chemotypes [1]
Subsequent screening of the MMV Pandemic Response Box library identified six active compounds against C. elegans, with vorapaxar (MMV1593515) showing particularly high potency (EC~50~ of 5.7×10^-7^ M) [3].
Disease Modeling
Beyond chemical screening, INVAPP/Paragon has been applied to characterize phenotypic effects in nematode disease models. A transgenic C. elegans strain expressing the human D76N β~2~-microglobulin variant associated with systemic amyloidosis showed significantly reduced growth and motility compared to control strains [9]. This model provides a platform for screening compounds targeting amyloidosis, with INVAPP/Paragon enabling quantitative assessment of candidate drug effects [9].
Applications in Mosquito Larvicide Screening
System Validation and Comparative Performance
INVAPP/Paragon has been adapted for screening mosquito larvicides, addressing the need for new insecticides to combat pyrethroid resistance [7] [5]. The system offers significant advantages over the WHO standard larval assay:
- Faster results than the 24-48 hour WHO protocol [7]
- Higher throughput enabled by 96-well plate format [5]
- Objective, quantitative endpoints versus subjective mortality scoring [7]
- Ability to detect sublethal effects through motility quantification [5]
Table 2: INVAPP/Paragon Applications in Mosquito Larval Screening
| Application | Species Tested | Key Findings |
|---|---|---|
| Temephos validation | Aedes aegypti, Anopheles gambiae [7] | Reliable quantification of time- and concentration-dependent effects |
| Pathogen Box screening | An. gambiae, An. coluzzii [7] [10] | Identification of tolfenpyrad as effective larvicide |
| Resistance detection | Deltamethrin-resistant and sensitive strains [7] | Differential responses in larval progeny from resistant adults |
| Smartphone adaptation | Ae. aegypti, An. gambiae [7] | Potential for portable field assays with geo-located data |
Chemical Screening and Resistance Monitoring
Screening the MMV Pathogen Box library identified tolfenpyrad as a potent larvicide, demonstrating the system's utility for discovering new insecticide chemotypes [7] [10]. The platform also detected differential responses in larval progeny from deltamethrin-resistant and sensitive mosquito strains, suggesting potential for monitoring emerging resistance [7]. Furthermore, the system's adaptability to smartphone-based imaging offers promise for developing portable field assays that could provide real-time, geo-located resistance data to identify hotspots [7] [5].
Detailed Experimental Protocols
C. elegans Growth/Motility Assay
This protocol assesses compound effects on C. elegans development from L1 to L4 larval stages [3].
Step-by-Step Procedure [3]:
- Worm maintenance: Maintain C. elegans N2 strain at 20°C on NGM agar seeded with E. coli OP50
- Synchronization:
- Wash mixed-stage populations from NGM plates with S-basal medium
- Centrifuge at 3000 × g for 4 minutes and repeat washes
- Filter through 100 μm then 40 μm sieves to obtain synchronized L1 larvae
- Assay preparation:
- Dilute synchronized L1s to approximately 15-25 worms per 50 μL in S-complete buffer with 1% w/v HB101 E. coli
- Dispense 99 μL of L1 suspension per well in 96-well plate
- Add 1 μL of test compound per well (1% v/v DMSO final concentration for controls)
- Incubation: Incubate plates at 25°C for 3 days
- Imaging:
- Capture 200-frame movies at 25 frames per second for 8 seconds using INVAPP
- Analyze with Paragon algorithm to determine movement index
Mosquito Larvicide Screening Assay
This protocol measures compound effects on larval motility in Anopheles and Aedes mosquitoes [7] [5].
Step-by-Step Procedure [7] [5]:
- Mosquito rearing:
- Anopheles: Hatch eggs in deoxygenated water with 0.001% pond salt, maintain at 25°C, test on days 5-6
- Aedes: Hatch eggs with crushed yeast tablet, test on days 3-4
- Larval preparation:
- Pass larval culture through 100 μm nylon mesh strainer to concentrate
- Dilute concentrated suspension until 100 μL contains 5-10 larvae
- Dispense 100 μL aliquots to each well of 96-well plate using pipette with cut-back tip
- Compound application:
- Add 100 μL of test compound dissolved in water (from 10^-2^ M DMSO stock) to yield required final concentration
- Include DMSO-only controls for normalization
- Motility assessment:
- Film larvae using INVAPP before compound addition to establish baseline
- Incubate for 240 minutes (or other appropriate duration)
- Film larvae again after incubation
- Normalize post-treatment movement index to baseline to account for well-to-well larval number variation
Research Reagent Solutions
Table 3: Essential Research Reagents for INVAPP/Paragon Screening
| Reagent/Resource | Specification | Function/Application |
|---|---|---|
| C. elegans strain | Wild-type N2 [3] | Primary nematode model for anthelmintic screening |
| Mosquito strains | An. gambiae G3 (sensitive), An. coluzzii Tiassale (resistant) [7] | Larvicide screening and resistance monitoring |
| Chemical libraries | MMV Pathogen Box, Pandemic Response Box [1] [3] | Sources of novel chemical starting points |
| Culture media | S-complete buffer with HB101 E. coli [3] | C. elegans liquid culture medium |
| Analysis software | MATLAB scripts for Paragon [1] | Open-source motility analysis |
| Imaging software | μManager [1] | Movie capture and microscope control |
The INVAPP/Paragon system represents a significant advancement in phenotypic screening technology, offering unprecedented throughput and quantitative precision for invertebrate-based chemical discovery. Its applications span anthelmintic discovery, larvicide screening, and disease modeling, demonstrating exceptional versatility across different organisms and research contexts. The system's open-source software, protocol adaptability, and proven success in identifying novel bioactive compounds make it a valuable resource for researchers addressing some of the most pressing challenges in global health. With potential for further development, including smartphone-based field deployment, INVAPP/Paragon stands to continue accelerating early-stage drug discovery for neglected tropical diseases and beyond.
The INVertebrate Automated Phenotyping Platform (INVAPP) is an integrated system designed for high-throughput chemical screening against parasitic nematodes and insect larvae. Its core technological strength lies in the combination of automated, high-resolution imaging with sophisticated motility analysis via the Paragon algorithm. This system addresses critical bottlenecks in parasitology and vector control research by enabling rapid, quantitative assessment of invertebrate behavior and development in response to chemical compounds, significantly accelerating the discovery of novel anthelmintics and larvicides [11] [7].
This document details the core methodologies for utilizing INVAPP and Paragon, providing application notes and standardized protocols for researchers in drug development and vector control.
Core System Components & Workflow
The INVAPP system automates the process of filming invertebrates in multi-well plates, and the Paragon algorithm translates the recorded video data into quantitative metrics of motility and development.
Key Research Reagent Solutions
The table below lists essential materials and reagents used in a typical INVAPP-based screening experiment.
| Item | Function/Description |
|---|---|
| 96-Well Plates | Standard platform for housing nematode/larvae samples during filming, enabling high-throughput screening [7]. |
| Chemical Libraries | Source of novel compounds for screening; the Pathogen Box from Medicines for Malaria Venture has been successfully used [11] [7]. |
| DMSO | Common solvent for preparing stock solutions of chemical compounds to be tested [7]. |
| Pond Guardian Tonic Salt | Added to hatching water to maintain osmolarity and health of nematode or mosquito larvae cultures [7]. |
| Nylon Mesh Cell Strainer | Used to concentrate and handle delicate invertebrate larvae during transfer to multi-well plates [7]. |
| Known Anthelmintics/Larvicides | Compounds like temephos and mebendazole used as positive controls for system validation [11] [7]. |
The following diagram illustrates the complete experimental workflow, from sample preparation to data analysis.
Detailed Experimental Protocols
Protocol 1: INVAPP-Based Screening of Chemical Libraries on Nematodes
This protocol is adapted from the screening of the Pathogen Box against C. elegans and parasitic nematodes [11].
Objective: To identify novel anthelmintic compounds by quantifying their effect on nematode motility and growth.
Materials:
- Biological Material: Synchronized populations of C. elegans, Haemonchus contortus, or other parasitic nematodes.
- Chemicals: Chemical library compounds (e.g., Pathogen Box), dissolved in DMSO.
- Equipment: INVAPP system, 96-well plates, cell strainers (100 μm), Gilson pipettes with cut-back tips.
Method:
- Sample Preparation: Harvest synchronized nematodes and concentrate them using a 100 μm nylon mesh cell strainer. Adjust the concentration so that a 100 μL aliquot contains 5-10 nematodes.
- Plate Loading: Dispense 100 μL of the nematode suspension into each well of a 96-well plate. Use cut-back pipette tips to prevent damage to the organisms.
- Compound Addition: Add 100 μL of the test compound to each well. Compounds are typically prepared from a 10 mM DMSO stock, diluted in water to a final test concentration of 10-100 μM. Include control wells containing DMSO alone at the same final concentration.
- Automated Phenotyping: Place the 96-well plate into the INVAPP system. Initiate automated filming according to the manufacturer's instructions. The recording duration may vary but typically spans several hours to capture time-dependent effects.
- Data Analysis: Process the recorded videos using the Paragon algorithm to generate quantitative motility and development metrics for each well.
Protocol 2: Larvicide Screening on Mosquito Larvae
This protocol is used for high-throughput screening of larvicides against Anopheles gambiae and Aedes aegypti [7].
Objective: To rapidly identify and characterize novel larvicidal compounds and monitor behavioral resistance.
Materials:
- Biological Material: First or early second instar larvae of Anopheles gambiae or Aedes aegypti.
- Chemicals: Test compounds (e.g., from MMV Pathogen Box), temephos (positive control).
- Equipment: INVAPP system, 96-well plates, cell strainers, deoxygenated water.
Method:
- Larval Hatching: Hatch mosquito eggs in deoxygenated, cooled tap water supplemented with a crushed yeast tablet or a small amount of cat food.
- Larval Harvesting: On days 3-4 post-hatching for Aedes or days 5-6 for Anopheles, concentrate the larvae using a cell strainer.
- Assay Setup: Dispense 5-10 larvae in 100 μL of water into each well of a 96-well plate. Add 100 μL of the test compound solution. The final DMSO concentration should be normalized across all wells and controls.
- Imaging & Analysis: Film the plates using INVAPP and analyze larval motility with the Paragon algorithm. The system can detect sub-lethal effects on motility faster than traditional WHO mortality assays.
Quantitative Data Output and Analysis
The Paragon algorithm processes video data to produce quantitative metrics. The following table summarizes sample validation data from screening campaigns.
Table: Representative Screening Data from INVAPP/Paragon Validation Studies
| Organism | Test Compound | Measured Effect (Metric) | Result | Reference |
|---|---|---|---|---|
| C. elegans, Parasitic Nematodes | Known anthelmintics (e.g., mebendazole) | Motility inhibition, Developmental delay | System efficacy validated; EC₅₀ values determined [11] | [11] |
| C. elegans, Parasitic Nematodes | Pathogen Box Library (400 compounds) | Motility & Growth | 14 novel anthelmintic hits identified (e.g., benzoxaborole, isoxazole) [11] | [11] |
| Ae. aegypti, An. gambiae | Temephos (larvicide) | Larval motility inhibition | Rapid, concentration-dependent decrease in motility observed [7] | [7] |
| Ae. aegypti, An. coluzzii | Tolfenpyrad (from Pathogen Box) | Larval motility inhibition | Confirmed as a potent larvicide in proof-of-principle screen [7] | [7] |
The Paragon Motility Algorithm: Core Logic
The Paragon algorithm converts raw video of moving invertebrates into a quantitative motility score. Its core logic involves background subtraction, object identification, and movement tracking.
Implementing INVAPP: Protocols for High-Throughput Screening and Resistance Monitoring
This application note details a standardized protocol for preparing mosquito larvae for high-throughput chemical screening using the INVertebrate Automated Phenotyping Platform (INVAPP). The procedures below support the automated, motility-based phenotyping of larvae to accelerate the discovery of novel larvicides and the assessment of insecticide resistance [5] [7].
Research Reagent Solutions and Essential Materials
The following table lists key materials required for the larval harvesting and plate setup workflow.
| Item | Function/Application in the Protocol |
|---|---|
| Anopheles gambiae (G3) & Aedes aegypti (New Orleans) | Pyrethroid-sensitive mosquito strains used as reference populations for establishing baseline susceptibility [7]. |
| Anopheles coluzzii (Tiassale) & Aedes aegypti (Cayman) | Deltamethrin-resistant mosquito strains for comparative studies on insecticide resistance [7]. |
| 96-Well Plates | Standard platform for high-throughput larval assays and chemical screening [5] [7]. |
| Pond Guardian Tonic Salt & Yeast Tablet | Used in hatching and maintenance water for Anopheles and Aedes larvae, respectively [7]. |
| 100 µm Nylon Mesh Cell Strainer | For concentrating and harvesting larvae from their maintenance water during the transfer process [7]. |
| INVAPP with Paragon Algorithm | Automated platform and software for recording and analyzing larval motility from video data [5] [7]. |
| Temephos | Established organophosphate larvicide used for protocol validation and as a positive control [7]. |
| Tolfenpyrad | Insecticide identified through high-throughput library screening using the INVAPP system [5] [7]. |
Larval Rearing, Harvesting, and Plate Setup Protocol
Larval Strains and Rearing
This protocol is optimized for larvae of malaria vectors (Anopheles gambiae and Anopheles coluzzii) and dengue/Zika vectors (Aedes aegypti) [7].
- Hatching: Ship eggs on moist filter paper. At the destination, wash eggs into a shallow dish containing deoxygenated (pre-boiled and cooled) water.
- For Anopheles, add 0.001% pond guardian tonic salt.
- For Aedes, add a crushed yeast tablet to the water to induce hatching.
- Maintenance: Maintain larvae at 25°C. Feed Anopheles larvae cat food pellets. Test Anopheles larvae on days 5-6 and Aedes larvae on days 3-4 post-hatching [7].
Larval Harvesting and 96-Well Plate Transfer
This critical step ensures the consistent, undamaged transfer of larvae to the assay plate.
- Concentration: Pass the water containing the swimming larvae through a 100 µm Nylon mesh cell strainer to concentrate them [7].
- Suspension Preparation: Dilute the concentrated larval suspension until a 100 µL aliquot contains between 5 to 10 larvae.
- Plate Loading: Using a standard pipette with the tip cut back to widen the orifice and prevent larval damage, transfer a 100 µL aliquot of the larval suspension to each well of a 96-well plate [7].
- Compound Application: Add 100 µL of the test compound to each well. For compounds dissolved in DMSO, prepare intermediate stocks and dilute in water so the final DMSO concentration is non-toxic (e.g., 1%). Wells with DMSO alone serve as negative controls [7].
Automated Phenotyping with INVAPP
- Filming: Place the prepared 96-well plate into the INVAPP system to record larval motility [7].
- Analysis: Use the Paragon algorithm to analyze the video recordings and quantify time- and concentration-dependent changes in larval motility. This provides a robust and fast readout of insecticide effect, surpassing the speed and objectivity of the manual WHO standard assay [5] [7].
Workflow and Experimental Logic
The following diagram illustrates the complete, standardized pathway from egg to data analysis.
Quantitative Protocol Performance and Outputs
The INVAPP platform, fed by this standardized workflow, generates robust quantitative data as shown in the summary tables below.
Table 1: Key Performance Timelines for the INVAPP Larval Assay
| Assay Stage | Key Metric | Performance/Output |
|---|---|---|
| Larval Loading | Plate Preparation Time | A 96-well plate can be loaded with larvae in minutes [7]. |
| Chemical Screening | Assay Speed | Provides a faster readout of insecticide action compared to the standard WHO larval assay [5]. |
| High-Throughput Capacity | Library Screening | Successfully applied to screen the ~400 compound Medicines for Malaria Venture (MMV) Pathogen Box library [5] [7]. |
Table 2: Representative Screening Results from the INVAPP Workflow
| Test Compound / Application | Quantified Result | Biological Significance |
|---|---|---|
| Temephos (Validation) | Reliable quantification of time- and concentration-dependent motility reduction [7]. | Confirms system effectiveness with an established larvicide [7]. |
| Tolfenpyrad (Discovery) | Identified as a hit from the MMV Pathogen Box screen [5] [7]. | Proof-of-principle for library-scale screening to find novel larvicides [5]. |
| Deltamethrin Resistance | Detection of differential motility responses in progeny from resistant vs. sensitive strains [7]. | Offers potential for a smartphone-based field assay to monitor resistance [5] [7]. |
This detailed protocol provides a reliable and efficient pathway from larval harvesting to 96-well plate setup, forming the foundation for robust, high-throughput chemical screening using the INVAPP platform. This standardized workflow is a critical tool for accelerating the discovery of new public health interventions and monitoring the spread of insecticide resistance.
The Paragon algorithm is a computational core component of the INVertebrate Automated Phenotyping Platform (INVAPP), designed to provide a robust, scalar readout of invertebrate motility for high-throughput chemical screening [11] [5]. This automated system addresses a critical bottleneck in parasitology and drug discovery by replacing labour-intensive, subjective manual observations with an unbiased, quantitative assay [11] [12]. The algorithm analyzes time-lapse video footage to quantify movement and, in some implementations, growth, in small invertebrates such as parasitic nematodes and mosquito larvae [11] [5] [13]. By converting complex animal behavior into a precise Movement Index, Paragon facilitates the rapid phenotypic screening of large chemical libraries for novel anthelmintics and larvicides [11] [5].
Its application is particularly vital given the urgent need for new compounds to control human parasitic nematodes, which infect hundreds of millions, and disease-transmitting mosquitoes, in the face of growing resistance to existing treatments [11] [5]. The Paragon algorithm, in conjunction with INVAPP, has been successfully validated against known anthelmintics and larvicides and deployed in screens that identified previously unknown active compounds, demonstrating its utility in accelerating the discovery of next-generation public health interventions [11] [5].
The Computational Basis of the Movement Score
Core Image Processing Workflow
The Paragon algorithm calculates its motility score by analyzing the temporal variance of individual pixels across a sequence of images, effectively capturing movement as changes in the visual field over time [13]. The core process can be broken down into several key computational stages.
Key Quantitative Outputs and Their Significance
The algorithm produces two primary quantitative metrics that serve as the foundation for phenotypic screening.
Table 1: Key Quantitative Outputs of the Paragon Algorithm
| Output Metric | Description | Computational Basis | Biological Interpretation |
|---|---|---|---|
| Movement Index | A primary scalar readout of motility [13]. | The proportion of pixels classified as "motile" based on variance thresholding [13]. | Direct measure of overall animal activity and health; suppressed by neuromuscular toxins [11] [5]. |
| Thrashing Rate | A measure of the frequency of specific movement patterns. | Quantified from the same image analysis used for the Movement Index [13]. | Particularly relevant for nematode and larval movement; indicates vitality and sublethal neurotoxicity [11]. |
Application Notes: Protocol for High-Throughput Chemical Screening
The following protocol details the application of INVAPP and the Paragon algorithm for a high-throughput screen of a chemical library for anthelmintic or larvicidal activity, as demonstrated with the Medicines for Malaria Venture (MMV) Pathogen Box [11] [5].
Experimental Workflow
The complete screening process, from plate preparation to data analysis, is designed for efficiency and reproducibility.
Step-by-Step Procedure
- Plate Preparation: Utilize a standard 96-well plate format. The system allows configuration of plate geometry via the
'plateColumns'(default: 12) and'plateRows'(default: 8) parameters in the analysis script [13]. - Organism Transfer:
- For mosquito larvae: Concentrate a suspension of first or early second-instar larvae (e.g., Anopheles gambiae or Aedes aegypti) and pipette 100 µL containing approximately 5-10 larvae into each well. To minimize damage, use a pipette tip with the end cut back [5] [7].
- For nematodes: Transfer a similar volume containing a synchronized population of C. elegans or parasitic nematodes like Haemonchus contortus [11].
- Compound Addition: Add 100 µL of the test compound to each well. Compounds are typically dissolved in DMSO and then serially diluted in water to achieve the desired final concentration (e.g., 10⁻⁴ M). Wells containing DMSO alone at the same concentration serve as negative (vehicle) controls [5] [7].
- Automated Filming: Place the 96-well plate into the INVAPP system. The platform automatically acquires time-lapse images of the entire plate at regular intervals over the desired incubation period (e.g., 24-80 hours), maintaining a consistent environment throughout [11] [5].
- Motility Analysis with Paragon:
- Execute the
invappParagonBatchfunction in MATLAB, specifying the path to the folder containing the acquired image files [13]. - The function processes the images, calculating the Movement Index and Thrashing Rate for each well over time [13].
- The default
'movementIndexThreshold'for classifying pixels as motile is 1 (mean plus one standard deviation of the pixel variance distribution), but this can be adjusted as an optional argument [13].
- Execute the
- Data Output and Hit Identification: The algorithm outputs a large table containing the motility metrics linked to the experimental filename. A significant reduction in the Movement Index compared to vehicle controls indicates a bioactive "hit" compound [11] [5].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagents and Materials for INVAPP-Paragon Screening
| Item | Function / Description | Example Use Case |
|---|---|---|
| INVAPP Hardware | An automated imaging system for 24/7 time-lapse image acquisition from multi-well plates under controlled environmental conditions [11] [5]. | Core platform for continuous, high-throughput monitoring of invertebrate motility. |
| Paragon Software | A MATLAB-based algorithm for analyzing time-lapse movies to calculate a Movement Index and thrashing rate [13]. | Converts raw video into quantitative, scalar motility data for statistical analysis. |
| Model Organisms | Invertebrates such as C. elegans, parasitic nematodes (H. contortus), or mosquito larvae (Ae. aegypti, An. gambiae) [11] [5]. | Biological sensors for detecting the phenotypic effects of chemical compounds. |
| Chemical Libraries | Collections of compounds for screening (e.g., MMV Pathogen Box) [11] [5]. | Source of potential novel anthelmintics or larvicides. |
| 96-Well Plates | Standard format microplates for housing organisms and compounds during assays [11] [13]. | Enables high-throughput testing by allowing many conditions to be run in parallel. |
| DMSO (Dimethyl Sulfoxide) | A common solvent for preparing stock solutions of chemical compounds [5] [7]. | Ensures compounds remain soluble when added to aqueous assay conditions. |
Validation and Key Findings from Library Screens
The INVAPP/Paragon system has been rigorously validated and deployed in successful library-scale screens, demonstrating its utility in accelerating drug discovery.
Table 3: Summary of Key Screening Validation and Results
| Experiment / Screen | Model Organism | Key Outcome | Reference |
|---|---|---|---|
| Validation with Known Anthelmintics | C. elegans, H. contortus, T. circumcincta, T. muris | Quantified the efficacy of a panel of known anthelmintics, validating the system's ability to detect motility impairment. | [11] |
| Screening of MMV Pathogen Box | C. elegans and parasitic nematodes | Identified compounds with known anthelmintic/anti-parasitic activity (e.g., tolfenpyrad, auranofin, mebendazole) and 14 compounds previously undescribed as anthelmintics. | [11] |
| Validation with Known Larvicide | Ae. aegypti, An. gambiae | Reliably quantified the time- and concentration-dependent effects of temephos (a standard larvicide) on larval motility. | [5] |
| Screening of MMV Pathogen Box | Ae. aegypti, An. gambiae | Identified tolfenpyrad as a potent larvicide in a proof-of-principle library screen. | [5] |
| Resistance Detection | Progeny of deltamethrin-resistant and sensitive mosquitoes | Detected differential motility responses in larvae, indicating potential for assessing behavioural resistance. | [5] |
The system's performance is evidenced by its ability to identify known active compounds like tolfenpyrad and mebendazole in blinded screens, while also discovering new potential leads, such as certain benzoxaborole and isoxazole chemotypes [11]. Furthermore, it can detect sublethal effects and differences in strains, such as the response of larvae from pyrethroid-resistant adult mosquitoes, offering a more nuanced picture of compound action and resistance than binary mortality assays [5].
Library-Scale Chemical Screening (e.g., MMV Pathogen Box)
The INVertebrate Automated Phenotyping Platform (INVAPP) is an automated, high-throughput system designed for plate-based chemical screening against nematodes, enabling the simultaneous quantification of motility and growth [11]. Coupled with the Paragon algorithm for data analysis, this integrated system allows researchers to efficiently screen for compounds that adversely affect worm development and movement, key indicators of anthelmintic activity [11].
The utility of INVAPP was robustly validated by quantifying the efficacy of a panel of established anthelmintic compounds against both model and parasitic nematodes, including Caenorhabditis elegans, Haemonchus contortus, Teladorsagia circumcincta, and Trichuris muris [11]. This validation confirmed the platform's sensitivity and reliability in detecting phenotypic changes, establishing it as a powerful tool for discovering novel therapeutics.
Quantitative Screening Data
The following tables summarize key quantitative results from the application of INVAPP in screening the Pathogen Box chemical library.
Table 1: Hit Compounds Identified from Pathogen Box Screening
| Compound Category | Examples Identified | Key Findings/Activity |
|---|---|---|
| Compounds with known anthelmintic/anti-parasitic activity | Tolffenpyrad, Auranofin, Mebendazole | System successfully identified known active compounds, validating the screening approach [11]. |
| Novel anthelmintic compounds (previously undescribed) | Benzoxaborole, Isoxazole chemotypes (total of 14 compounds) | Discovery of new chemotypes with activity against nematodes, expanding potential lead compounds [11]. |
Table 2: Validation of INVAPP with Known Anthelmintics
| Nematode Species | Platform Application | Key Outcome |
|---|---|---|
| C. elegans, H. contortus, T. circumcincta, T. muris | Quantification of motility and growth inhibition | Platform reliably determined the efficacy of known anthelmintics, correlating with expected phenotypic effects [11]. |
Experimental Protocols
INVAPP-Based Phenotypic Screening Protocol
This protocol details the steps for using the INVAPP system for high-throughput phenotypic screening of chemical libraries against nematodes [11].
Organism Preparation:
- Culture synchronized populations of the target nematode (e.g., C. elegans, H. contortus, T. muris) using standard methods.
- Harvest and wash the worms in appropriate buffer solutions to remove media components.
Compound Library Plating:
- Dispense the chemical library (e.g., Pathogen Box compounds) into assay-compatible microtiter plates. The Pathogen Box is a 400-compound library provided by the Medicines for Malaria Venture.
- Include control wells on each plate: negative controls (vehicle only) and positive controls (known anthelmintics, e.g., mebendazole).
Assay Setup:
- Transfer a standardized volume and number of nematodes into each well of the compound plate.
- Seal the plates to prevent evaporation and incubate under conditions optimal for the nematode species for a defined period (e.g., 72-96 hours for growth assays).
Automated Phenotyping with INVAPP:
- Place the assay plates into the INVAPP system for automated, high-throughput imaging at defined time intervals.
- INVAPP captures video data of worm motility and static images for biomass assessment.
Data Analysis with Paragon Algorithm:
- Process the acquired images and videos using the Paragon algorithm.
- The algorithm quantifies key phenotypic parameters, including:
- Motility: Based on worm movement frequency and pattern.
- Growth: Estimated by measuring the total worm biomass or body size in each well.
- Normalize data against plate controls to calculate percent inhibition for motility and growth.
Hit Identification:
- Define hit thresholds based on statistical significance (e.g., >50% inhibition in motility or growth compared to negative control).
- Rank compounds based on their activity scores for further validation.
Supplementary Virtual Screening Protocol
For a comprehensive discovery pipeline, INVAPP-based phenotypic screening can be integrated with computational approaches. The following protocol outlines a fully local virtual screening pipeline using free software to prioritize compounds for phenotypic testing [14].
System and Software Setup:
- Use a Unix-like operating system (Linux or Windows Subsystem for Linux on Windows 11).
- Install required free software dependencies:
build-essential,openbabel,fpocket, andAutoDock Vina(or QuickVina 2). - Install the
jamdock-suitescripts (jamlib,jamreceptor,jamqvina,jamresume,jamrank) from the GitHub repository and add them to the system path [14].
Compound Library Generation (
jamlib):- Use
jamlibto generate a library of compounds in PDBQT format, suitable for docking. This can include custom molecule sets or pre-defined libraries like FDA-approved drugs from the ZINC database [14].
- Use
Receptor and Grid Preparation (
jamreceptor):- Obtain the 3D structure of the target protein (e.g., from the Protein Data Bank).
- Use
jamreceptorto convert the receptor file to PDBQT format. - Run
jamreceptorwithfpocketto identify potential binding pockets on the target protein. - Select the most relevant binding pocket (based on druggability score and biological knowledge) to automatically define the docking grid box coordinates [14].
Molecular Docking (
jamqvina):- Execute the docking calculation using
jamqvina(orjamvina), specifying the receptor, compound library, and grid box parameters. - Use
jamresumeto resume long-running jobs if needed [14].
- Execute the docking calculation using
Analysis of Docking Results (
jamrank):- After docking completion, use
jamrankto evaluate and rank the results based on docking scores. - Select the top-ranking compounds for subsequent experimental validation using the INVAPP phenotypic assay [14].
- After docking completion, use
Workflow and Pathway Diagrams
The Scientist's Toolkit
Table 3: Essential Research Reagents and Materials
| Item/Reagent | Function in Screening | Specification Notes |
|---|---|---|
| Pathogen Box Library | A curated set of ~400 chemical compounds with known or potential activity against pathogens [11]. | Serves as the primary source for chemical screening. Provided by the Medicines for Malaria Venture. |
| Nematode Strains | Biological targets for phenotypic screening. Include model (C. elegans) and parasitic (H. contortus, T. muris) species [11]. | Requires standard culturing protocols and synchronization for consistent assay results. |
| INVAPP Hardware | Automated, high-throughput platform for imaging nematodes in microtiter plates [11]. | Quantifies motility (via video) and growth (via biomass estimation). |
| Paragon Algorithm | Software for analyzing image and video data from INVAPP [11]. | Translates raw phenotypic data into quantitative metrics of motility and growth inhibition. |
| AutoDock Vina/QuickVina 2 | Molecular docking engine used for structure-based virtual screening [14]. | Predicts binding poses and scores of small molecules to a target protein. Free software. |
| ZINC/Files.Docking.org | Publicly accessible databases hosting chemical and structural information for millions of commercially available compounds [14]. | Source for building ultra-large virtual screening libraries. |
| jamdock-suite Scripts | A set of five modular Bash scripts (jamlib, jamreceptor, jamqvina, jamresume, jamrank) that automate the virtual screening pipeline [14]. | Lowers the access barrier for structure-based drug discovery; uses free, local software. |
The INVertebrate Automated Phenotyping Platform (INVAPP), in conjunction with the Paragon algorithm, provides a high-throughput, quantitative method for detecting insecticide resistance in mosquito larvae. This system overcomes limitations of traditional, labor-intensive World Health Organization (WHO) larval assays by automating the measurement of larval motility, a sublethal endpoint that can reveal resistance phenotypes faster and with greater objectivity [7]. This application note details the protocol for using INVAPP to quantify differential responses between insecticide-resistant and susceptible larval strains, a capability demonstrated on the progeny of pyrethroid-resistant adults of Anopheles coluzzii and Aedes aegypti [7]. This approach is vital for monitoring the emergence and spread of resistance in field populations, informing vector control strategies, and screening new chemical entities for larvicidal activity.
Key Research Reagent Solutions
The following table catalogues the essential materials and reagents required for conducting larval resistance phenotyping with INVAPP.
Table 1: Essential Research Reagents and Materials
| Item | Function/Description |
|---|---|
| INVAPP System | An automated platform for recording high-frame-rate movies of invertebrate motility in multi-well plates [7]. |
| Paragon Algorithm | A complementary algorithm that analyzes video data from INVAPP to calculate a movement score based on motile pixels [7] [3]. |
| Standard 96-well Plates | The standardized vessel for holding larvae and insecticide solutions during high-throughput screening [7]. |
| Deltamethrin | A standard pyrethroid insecticide used for selecting and characterizing resistant strains [7]. |
| Temephos | An organophosphate larvicide used as a reference compound in toxicity assays [7]. |
| Dimethyl Sulfoxide (DMSO) | A universal solvent for preparing stock solutions of water-insoluble chemical compounds [7] [3]. |
| S-basal Medium | A defined saline solution used for maintaining and washing C. elegans in liquid culture [3]. |
| HB101 E. coli | A bacterial food source (1% w/v) for C. elegans in liquid growth/motility assays [3]. |
The INVAPP/Paragon system generates robust quantitative data on larval response to insecticides. The table below summarizes key metrics from representative studies.
Table 2: Quantitative Data from Larval Resistance Phenotyping
| Measured Parameter | Result / Value | Experimental Context |
|---|---|---|
| Assay Speed | Faster than WHO standard larval assay | General capability of the INVAPP system for measuring time- and concentration-dependent insecticide actions [7]. |
| Screening Capacity | 400 compounds (MMV Pathogen Box library) | Proof-of-principle for library-scale chemical screening identifying tolfenpyrad as a hit [7]. |
| Larval Motility Scoring | 200-frame movies captured at 25 frames/s for 8 seconds | Standard filming parameters for INVAPP used in C. elegans anthelmintic screening [3]. |
| Resistant Strain Mortality (WHO Bioassay) | 10-20% (Tiassale, An. coluzzii); 20-50% (Cayman, Ae. aegypti) | Characterization of the deltamethrin-resistant adult mosquito strains from which larval progeny were derived [7]. |
| Resistance Ratio (Larvae) | 1.64 (Cayman vs. New Orleans Ae. aegypti strains) | Previously reported resistance ratio for the Cayman larvae compared to the susceptible New Orleans strain [7]. |
Experimental Protocol
Larval Source and Preparation
- Mosquito Strains: Utilize characterized deltamethrin-resistant and susceptible strains of target species (e.g., Anopheles coluzzii Tiassalé (resistant) and Anopheles gambiae G3 (susceptible); Aedes aegypti Cayman (resistant) and New Orleans (susceptible)) [7].
- Egg Hatching:
- Larval Harvesting: Concentrate first and second instar larvae by passing the water through a 100 μm Nylon mesh cell strainer. Dilute the concentrated larval suspension until a 100 μL aliquot contains 5-10 larvae [7].
Assay Setup and Compound Exposure
- Plate Preparation: Add 100 μL of the diluted larval suspension to each well of a 96-well plate. Use a pipette tip with the end cut back to avoid damaging larvae [7].
- Compound Addition: Add 100 μL of the insecticide solution (e.g., deltamethrin, temephos, or a library compound) to the test wells. Prepare test solutions in water from a 10⁻² M DMSO stock to achieve the desired final concentration (e.g., 10⁻⁴ M for primary screening). For control wells, add 100 μL of a DMSO solution diluted in water to the same concentration as the test wells [7] [3].
- Incubation: Incubate the assay plate under appropriate environmental conditions (e.g., 25°C) for a defined period before filming [3].
Data Acquisition with INVAPP
- Filming: Place the 96-well plate into the INVAPP system. Capture movies of each well. Standard parameters are 200 frames at a rate of 25 frames per second, resulting in 8 seconds of video per well [3].
Data Analysis with Paragon
- Motility Scoring: Analyze the captured movies using the Paragon algorithm, which is implemented in MATLAB (scripts available at https://github.com/fpartridge/invapp-paragon) [7] [3].
- Pixel Variance Calculation: The algorithm calculates the variance over time for each pixel in the video. Pixels with a variance greater than one standard deviation above the mean variance are classified as 'motile pixels' [3].
- Quantification: The total number of motile pixels within each well is counted to generate a quantitative movement score for that well [3].
- Dose-Response and Resistance Ratio: For concentration-dependent assays, plot larval movement (or % inhibition) against the logarithm of insecticide concentration to generate dose-response curves and calculate EC₅₀ values (the concentration causing a 50% reduction in movement). The Resistance Ratio (RR) is calculated as EC₅₀ (Resistant strain) / EC₅₀ (Susceptible strain).
Workflow and Data Analysis Diagrams
The following diagram illustrates the complete experimental and analytical pipeline for detecting larval insecticide resistance using the INVAPP/Paragon system.
INVAPP Resistance Detection Workflow
The data analysis pathway, from raw video to resistance quantification, is detailed in the diagram below.
Paragon Data Analysis Pipeline
Application in Chemical Screening and Field Surveillance
The primary application of this protocol is the high-throughput screening of chemical libraries for new larvicides, as demonstrated by the successful identification of tolfenpyrad from the MMV Pathogen Box [7]. Furthermore, the system's adaptability for use with a smartphone camera application presents a significant future potential for developing portable field assays. This would enable real-time, geo-located monitoring of resistance hotspots directly in the field, providing critical data for vector control programs with unprecedented speed and spatial accuracy [7].
The INVertebrate Automated Phenotyping Platform (INVAPP) represents a significant technological advancement in the field of chemical screening for public health entomology [5]. This automated system, combined with the Paragon algorithm, was developed to address critical limitations of the World Health Organization (WHO) standard larval assay, which relies on labor-intensive visual inspections and subjective mortality assessments [5] [7]. The increasing threat of insecticide resistance in mosquito vectors of deadly diseases such as malaria, dengue, Zika, and yellow fever has created an urgent need for high-throughput screening methods to identify novel larvicidal compounds [5].
This case study details the application of the INVAPP system within a broader thesis research framework to screen chemical libraries for new larvicides, culminating in the identification of tolfenpyrad as a highly effective compound [5] [4]. The methodology and findings presented herein provide researchers with a validated protocol for automated phenotyping of mosquito larvae and a compelling example of its successful implementation in chemical discovery.
The INVAPP system functions as an integrated hardware and software solution specifically designed for quantifying invertebrate motility [5] [7]. Its application to mosquito larvae provides a quantitative, high-throughput替代 to traditional bioassays.
Key System Components
- Imaging Hardware: A digital camera system configured to record multi-well plates containing mosquito larvae in aqueous solution [5].
- Paragon Algorithm: Proprietary software that analyzes sequential video images to compute a motility index based on larval movement, providing an objective and quantifiable readout of larval viability and behavior [5] [7].
- Multi-well Plate Platform: Enables simultaneous testing of multiple compounds and concentrations, facilitating library-scale screening efforts [5].
Advantages Over Standard WHO Larval Assay
The WHO standard assay defines larval death through visual assessment of moribund appearance and lack of response to physical stimulation, a endpoint that can be "difficult to assign unambiguously" and is highly labor-intensive [5]. In contrast, the INVAPP/Paragon system:
- Provides objective, quantitative data on larval motility as a sensitive marker of sublethal and lethal effects [5].
- Offers a faster readout of insecticide action compared to the standard assay [5] [4].
- Enables high-throughput screening capabilities essential for evaluating large chemical libraries [5].
Table: Comparison of Larval Bioassay Methods
| Feature | WHO Standard Assay | INVAPP/Paragon System |
|---|---|---|
| Throughput | Low (individual cups) | High (96-well plates) |
| Primary Endpoint | Mortality (visual assessment) | Motility (quantitative) |
| Data Output | Subjective, binary (dead/alive) | Objective, continuous (motility index) |
| Assay Speed | Slower (24-48 hours) | Faster (hours) |
| Labor Requirement | High | Low (automated) |
| Resistance Detection | Limited to mortality | Potential for behavioral response detection |
Experimental Application: Screening for Novel Larvicides
Mosquito Strains and Rearing
Research utilized established colonies of Anopheles gambiae (and An. coluzzii) as well as Aedes aegypti, representing major vectors of human disease [5] [7]. Strains were characterized according to WHO protocols as either deltamethrin-sensitive or deltamethrin-resistant:
- Ae. aegypti: Resistant (Cayman) and sensitive (New Orleans) strains [7].
- An. coluzzii: Resistant (Tiassale) and sensitive (G3) strains [7].
Larvae were hatched from eggs and maintained under controlled conditions (25°C) in deoxygenated water [7]. For bioassays, first and early second instar larvae (1-2 days post-hatching) were used [7].
Chemical Library
The Medicines for Malaria Venture (MMV) Pathogen Box library was screened as a proof-of-concept for library-scale chemical screening [5] [4]. This library contains 400 diverse compounds with known activity against various pathogens, providing a rich source for repurposing and discovering new insecticidal activities.
INVAPP Bioassay Protocol
The following workflow details the exact procedure used for high-throughput screening:
Step-by-Step Methodology [7]:
- Larval Harvesting: Water containing larvae was passed through a 100 μm Nylon mesh cell strainer to concentrate them. The concentrated suspension was diluted such that a 100 μL aliquot contained 5-10 larvae.
- Plate Setup: Using a standard pipette (with the tip cut back to prevent larval damage), 100 μL of the larval suspension was added to each well of a 96-well plate.
- Compound Application: 100 μL of the test compound (dissolved in water from a 10⁻²M DMSO stock to yield a final concentration of 10⁻⁴M) was added to each well. Control wells received DMSO alone at the same final concentration.
- Filming and Analysis: Larvae were filmed using the INVAPP hardware. The resulting video was analyzed by the Paragon algorithm, which generated a quantitative motility index.
Data Analysis and Hit Confirmation
The INVAPP system quantified both time-dependent and concentration-dependent effects of chemical exposure on larval motility [5]. This quantitative data allowed for robust dose-response analysis and comparison of compound potency.
From the MMV Pathogen Box screen, a single compound demonstrated potent larvicidal activity by rapidly and severely reducing larval motility. This compound was subsequently identified as tolfenpyrad [5] [4]. Follow-up confirmation assays validated its efficacy as a larvicide.
Results: Tolfenpyrad as a Potent Larvicide
Compound Profile
Tolfenpyrad is a METI (Mitochondrial Electron Transport Inhibitor) acaricide and insecticide with a contact mode of action [15]. It acts by inhibiting cellular respiration in the insect, leading to rapid cessation of feeding and death [15].
Table: Characteristics of Tolfenpyrad
| Characteristic | Description |
|---|---|
| Chemical Class | METI Acaricide/Insecticide |
| Mode of Action | Inhibition of cellular respiration |
| Primary Effects | Rapid knockdown, feeding cessation, suppression of egg-laying |
| Spectrum of Activity | Wide (Aphids, Lepidoptera, Psyllids, Thrips, etc.) |
| Cross-Resistance | No known cross-resistance to diamides, neonicotinoids, OPs, or pyrethroids |
| Available Formulations | 150 g/l EC or SC |
Efficacy Against Mosquito Larvae
In the INVAPP bioassay, tolfenpyrad caused a significant and rapid reduction in larval motility in both Anopheles gambiae and Aedes aegypti, confirming its potent larvicidal activity [5] [4]. The automated system detected effects faster than the traditional WHO mortality assay.
Detection of Insecticide Resistance
The INVAPP/Paragon system was also used to compare larval responses from pyrethroid-resistant and pyrethroid-sensitive mosquito strains [5]. Larvae derived from WHO-classified deltamethrin-resistant and sensitive adults of An. coluzzii and Ae. aegypti showed differential responses in the assay [5] [7]. This demonstrates the platform's potential for detecting behavioral changes related to insecticide resistance, extending beyond traditional mortality-based resistance monitoring.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table: Key Reagents and Materials for INVAPP Bioassay
| Item | Specification/Function |
|---|---|
| Mosquito Larvae | First and early second instar of Anopheles gambiae (G3, Tiassale) and Aedes aegypti (New Orleans, Cayman) [7]. |
| Chemical Library | MMV Pathogen Box (400 compounds) [5]. Source for novel larvicide discovery. |
| 96-Well Plates | Standard format for high-throughput screening and automated imaging [5] [7]. |
| INVAPP Hardware | Custom digital camera setup for filming larval movement in multi-well plates [5]. |
| Paragon Algorithm | Software for analyzing video footage and calculating a quantitative motility index [5] [7]. |
| Nylon Mesh Strainer | 100 μm mesh for concentrating and harvesting larvae from maintenance water [7]. |
| DMSO | Solvent for preparing stock solutions of test compounds [7]. |
| Temephos | Established organophosphate larvicide used as a positive control [5]. |
Protocol: Larval Motility Bioassay for Larvicide Screening
This protocol is adapted from Buckingham et al. (2021) for the evaluation of larvicidal activity using automated phenotyping [5] [7].
Scope
This procedure describes a method for testing chemical compounds against early instar mosquito larvae in a 96-well plate format, using automated imaging and analysis to quantify motility as a primary endpoint.
Materials Required
- Test compounds (e.g., from MMV Pathogen Box)
- DMSO (for preparing compound stocks)
- 96-well plates
- Pipettes and tips (with fine tips cut back for larval transfer)
- 100 μm Nylon mesh cell strainer
- Shallow dishes for larval rearing
- Deoxygenated water (pre-boiled and cooled tap water)
- INVAPP imaging system
- Computer with Paragon algorithm software
Procedure
- Larval Preparation: Concentrate 1-2 day post-hatch larvae using a 100 μm mesh strainer. Dilute the concentrated larval suspension to a density of 5-10 larvae per 100 μL.
- Plate Setup: Pipette 100 μL of the larval suspension into each well of the 96-well plate.
- Compound Application: Add 100 μL of the test compound to each well. For a final test concentration of 10⁻⁴M, dilute the compound from a 10⁻²M DMSO stock. Include control wells with DMSO only.
- Imaging: Place the 96-well plate into the INVAPP system and commence filming according to the system's specifications.
- Data Analysis: Process the recorded video using the Paragon algorithm to generate motility indices for each well over time.
- Data Interpretation: Analyze the time- and concentration-dependent effects on larval motility. Compare results to controls and known larvicides (e.g., temephos) to determine efficacy.
This case study demonstrates that the integration of the INVAPP automated phenotyping platform with the Paragon analysis algorithm provides a powerful and robust system for high-throughput screening of novel larvicides [5]. The successful identification of tolfenpyrad from the MMV Pathogen Box library validates this approach as an efficient method for discovering new insecticidal chemistries, which are urgently needed to combat insecticide resistance in mosquito vectors [5] [4].
The methodology offers significant advantages over the WHO standard larval assay, including objectivity, higher throughput, and faster results [5]. Furthermore, the system's adaptability to smartphone-based imaging suggests a promising future for its development into a portable field assay for real-time, geo-located monitoring of insecticide resistance hotspots, providing a critical tool for global vector control management [5].
Optimizing INVAPP Performance: Overcoming Technical Bottlenecks and Enhancing Data Quality
Ensuring Consistent Larval Dispensing and Viability
Within modern chemical screening research, the INVertebrate Automated Phenotyping Platform (INVAPP) has emerged as a powerful tool for high-throughput, whole-organism drug discovery [16] [17]. Its effectiveness, however, fundamentally depends on the precision of its initial input: the consistent delivery of healthy, developmentally synchronized larvae. Inconsistent larval dispensing—variations in larval number, developmental stage, or health—introduces significant biological noise that can obscure subtle phenotypic responses to chemical treatments, compromise statistical power, and lead to irreproducible results [16]. This Application Note provides detailed protocols and quantitative data to standardize larval culture, dispensing, and viability assessment, specifically optimized for the INVAPP workflow. By implementing these guidelines, researchers can ensure the generation of high-quality, reliable data in chemical screening campaigns.
The Critical Role of Standardized Larvae in Screening
The use of intact invertebrate larvae, such as those of Danio rerio (zebrafish) and Caenorhabditis elegans (C. elegans), in high-throughput screening (HTS) capitalizes on their unique biological advantages. These organisms offer a complex, multicellular context for assessing chemical effects, which is often lacking in simplistic in vitro models [16] [17]. The global HTS market, projected to grow from USD 26.12 billion in 2025 to USD 53.21 billion by 2032, underscores the increasing reliance on these scalable platforms [18]. A key driver of this growth is the shift towards phenotypic screening, where observable traits in whole organisms are measured without presupposing a molecular target [17]. The success of this approach, as implemented on INVAPP, hinges on the ability to detect meaningful phenotypic shifts—such as changes in locomotor activity, neuromuscular junction morphology, or thigmotaxis—in response to chemical perturbations [19] [20] [21]. These subtle phenotypes can only be reliably identified against a background of minimal variability, making standardized larval quality and dispensing not merely a best practice, but a necessity for accurate hit identification and validation.
Quantitative Benchmarks for Larval Viability
Establishing clear, quantitative benchmarks is essential for monitoring the health and consistency of larval cultures used in screening. The following table summarizes key viability and phenotypic metrics for zebrafish and C. elegans larvae, derived from established models.
Table 1: Key Viability and Phenotypic Metrics for Zebrafish and C. elegans Larvae
| Metric | Typical Value for Healthy Larvae | Experimental Context & Impact |
|---|---|---|
| Zebrafish Larval Locomotor Activity | Significant decrease at 3 µM and 6 µM 8-Methoxypeucedanin (8-MP) [19] | Used as a behavioral index for anxiolytic activity; deviations from baseline indicate pharmacological effect [19]. |
| Zebrafish Larval Anxiolytic Response | U-shape dose–response effect (1.5–15 µM 8-MP) [19] | Indicator of compound efficacy; a non-standard curve suggests impaired larval health or inconsistent dispensing [19]. |
| Neuromuscular Junction (NMJ) Bouton Count | Significant increase upon APP expression; suppressed by polo loss [21] | A key morphological phenotype in neurodegenerative models; variability can confound genetic or chemical suppression studies [21]. |
| Larval Crawling Speed | Dramatically reduced by APP expression; ameliorated by polo loss [21] | A functional measure of neuronal integrity; consistent baseline speed is critical for assessing rescue in drug screens [21]. |
Detailed Protocols for Consistent Larval Culture and Dispensing
Protocol: Synchronized Zebrafish Larval Culture for INVAPP
This protocol is designed to generate large batches of developmentally synchronized zebrafish larvae at 5 days post-fertilization (dpf), suitable for anxiolytic and neuroactive compound screening [19] [20].
Key Research Reagent Solutions:
- E3 Embryo Medium: Standard medium for maintaining zebrafish embryos and larvae.
- PTU (1-Phenyl-2-thiourea): Used to inhibit pigment formation, facilitating clear visualization of internal structures and behaviors.
- 8-Methoxypeucedanin (8-MP): A reference anxiolytic compound used for assay validation [19].
- Dispensing Microplate: A 96-well or 384-well plate pre-loaded with compounds or vehicle control.
Methodology:
- Embryo Collection: Set up natural pairwise or group spawns of adult zebrafish. Collect embryos within 1 hour of spawning and rinse thoroughly with E3 embryo medium.
- Dechorionation and Sorting: At approximately 4-6 hours post-fertilization (hpf), manually remove the chorion from embryos if necessary. Under a dissecting microscope, sort and retain only fertilized, developmentally normal embryos.
- PTU Treatment (Optional): To prevent pigmentation, incubate embryos in E3 medium containing 0.003% PTU from 24 hpf until the end of the experiment.
- Incubation and Staging: Maintain embryos in a light- and temperature-controlled incubator at 28.5°C. Raise them in petri dishes with a density not exceeding 50 embryos per 60 mm dish. Monitor development daily, removing any deceased or abnormally developing individuals.
- Larval Readiness: At 5 dpf, larvae are ready for dispensing and screening. Confirm that larvae exhibit normal morphology and spontaneous swimming behavior.
Protocol: Automated Larval Dispensing for High-Throughput Screening
This protocol outlines the automated dispensing of zebrafish larvae into multi-well plates, a critical step for INVAPP integration.
Key Research Reagent Solutions:
- Larval Suspension Buffer: E3 medium, optionally supplemented with low-concentration agar or methylcellulose to stabilize larvae and prevent settling during dispensing.
- Reference Control Compounds: Such as Diazepam (for anxiolytic validation) or PTZ (for pro-convulsant effects), to validate assay performance on each plate [19].
Methodology:
- Larval Preparation: Gently transfer a batch of 5 dpf synchronized larvae from the petri dish into a sterile beaker or specialized dispensing reservoir containing larval suspension buffer.
- Automated Dispenser Setup: Prime the automated liquid handler or larval dispenser (e.g., systems from companies like Beckman Coulter or SPT Labtech, which specialize in HTS automation [18]) with the suspension buffer to clear air bubbles.
- Dispensing Calibration:
- Set the dispenser to deliver a predefined volume (e.g., 100-200 µL) sufficient to submerge a single larva.
- Conduct a test run into an empty plate and manually count the number of larvae per well to calculate the average and variance.
- Adjust the larval density in the reservoir and/or the aspiration parameters (e.g., speed, mixing) until the target of one larva per well is achieved with >95% accuracy.
- Plate Dispensing: Run the automated dispensing protocol to fill the assay microplate. During dispensing, maintain gentle agitation in the larval reservoir to ensure a uniform suspension.
- Post-Dispensing Verification: Immediately after dispensing, manually inspect each well under a microscope to confirm the presence of a single, viable larva. Note and flag any empty or multiple-occupancy wells for exclusion from subsequent analysis.
Protocol: Viability and Baseline Behavior Assessment
Prior to chemical exposure, confirm larval viability and establish baseline behavior for data normalization.
Methodology:
- Viability Check: Post-dispensing, observe larvae for spontaneous coiling or swimming movements. Larvae that are immobile or unresponsive to a gentle tap on the plate should be considered non-viable and excluded.
- Acclimatization: After dispensing, allow the plates to acclimatize in the INVAPP imaging chamber for a standardized period (e.g., 15-30 minutes) to minimize stress-induced behavioral artifacts.
- Baseline Imaging: Acquire a short, pre-treatment video of larval activity using INVAPP. Quantify baseline locomotor activity (e.g., total distance moved, velocity) for each larva.
- Data Flagging: Establish a pre-defined threshold for minimum baseline activity. Larvae falling below this threshold may be unhealthy and should be flagged.
Experimental Workflow and Quality Control
The following diagram illustrates the integrated workflow from larval culture to data acquisition, highlighting critical quality control checkpoints.
The Scientist's Toolkit: Essential Research Reagents
A successful screening campaign relies on a suite of reliable reagents and tools. The following table details essential solutions for larval-based screening on the INVAPP platform.
Table 2: Essential Research Reagent Solutions for INVAPP Screening
| Reagent / Solution | Function / Purpose | Application Example |
|---|---|---|
| Soy Protein Isolate | Coats cell culture dishes to enhance the therapeutic potency of MSC-derived extracellular vesicles [22]. | Used in pre-conditioning stem cells for exosome production in neurovascular injury studies [22]. |
| Synchronized C. elegans L1 Larvae | The initial stage for high-throughput chemical genetic screens and anthelmintic drug discovery [16]. | Dispensed into 384-well plates for whole-organism drug sensitivity and longevity studies [16]. |
| 8-Methoxypeucedanin (8-MP) | A reference anxiolytic furanocoumarin used to validate behavioral assays in zebrafish models [19]. | Administered (1.5–15 µM) to 5 dpf zebrafish larvae to induce a U-shaped dose-response in thigmotaxis [19]. |
| Dyrk1A Inhibitors (e.g., KuFal194) | Pharmacological tools to rescue specific neurological phenotypes in zebrafish models of disease [20]. | Used to validate targets by suppressing Purkinje cell disorganization and swim deficits in zebrafish [20]. |
| Cell-Based Assay Kits | Provide physiologically relevant screening models for target validation and MoA studies [18]. | Used in secondary assays to confirm hits identified from primary INVAPP phenotypic screens (e.g., Melanocortin Receptor Reporter Assays [18]). |
The reliability of any automated phenotyping platform is inextricably linked to the quality of its biological input. For the INVAPP system, mastering the protocols for consistent larval dispensing and ensuring high larval viability are foundational to achieving statistically robust and biologically relevant screening outcomes. By adhering to the detailed methodologies for synchronized culture, automated dispensing, and rigorous quality control outlined in this Application Note, researchers can significantly enhance the signal-to-noise ratio in their chemical screens. This disciplined approach enables the confident detection of subtle phenotypic changes, thereby accelerating the discovery of novel therapeutic candidates in neuropsychiatric and neurodegenerative disease research.
Within the context of an INVertebrate Automated Phenotyping Platform (INVAPP) for chemical screening, the accuracy of phenotypic measurements—such as mosquito larval motility—is directly contingent upon the precise optimization of image capture parameters [7]. The INVAPP system, used in conjunction with the Paragon analysis algorithm, relies on high-quality video data to quantify behavioral changes in response to chemical compounds [7] [5]. This document outlines standardized protocols for determining the optimal frame rate and resolution settings to ensure reliable, high-throughput data acquisition for anthelmintic and larvicidal drug discovery.
Key Quantitative Specifications for INVAPP Imaging
Based on established methodologies, the following table summarizes the core image capture parameters used in published INVAPP applications.
Table 1: Core Image Capture Parameters for INVAPP in Larval Motility Assays
| Parameter | Recommended Setting | Experimental Context | Purpose & Rationale |
|---|---|---|---|
| Frame Rate | 25 frames per second (fps) [7] [3] | Filming mosquito larvae (Anopheles gambiae, Aedes aegypti) or nematodes (C. elegans) for motility analysis [7]. | Captures rapid, subtle movements of invertebrates without motion blur. Provides sufficient temporal resolution for the Paragon algorithm to compute pixel-wise variance [3]. |
| Recording Duration | 8 seconds [3] | Standardized recording period for each well in a multi-well plate. | Provides a sufficient data sample (resulting in a 200-frame movie) to compute a robust and reproducible motility score [3]. |
| Spatial Resolution | Not explicitly specified, but high-throughput is enabled via 96-well plates [7]. | Liquid cultures in 96-well plates for screening chemical libraries [7] [3]. | The system prioritizes the analysis of pixel variance over maximum resolution. The setup must image multiple wells simultaneously to achieve high-throughput capacity. |
Experimental Protocol: Determining Optimal Settings for Motility Assays
The following diagram illustrates the core workflow for optimizing and executing an image-based screening session with INVAPP.
Step-by-Step Procedure
Step 1: Plate Preparation and Setup
- Transfer Test Organisms: Pipette synchronized mosquito larvae (1-2 days post-hatching) or C. elegans (L1 or L4 larvae) into the wells of a 96-well plate. Use approximately 15-25 organisms per well in a volume of 50-100 μL of appropriate buffer [7] [3].
- Add Chemical Compounds: Add 1 μL of the test compound from a DMSO stock solution to each well. Include control wells with DMSO alone. The final standard screening concentration is typically 1.0 × 10⁻⁴ M [3].
- Incubation: Incubate the assay plates at a standardized temperature (e.g., 25°C) for a defined period (e.g., 3 days for C. elegans growth/motility assays) before imaging [3].
Step 2: Camera and Software Configuration
- Mount the Camera: Position the camera (or smartphone for field adaptations) securely above the plate stage to ensure a consistent and level field of view [7].
- Set Core Parameters: In the capture software (e.g., μManager), configure the following [3]:
- Frame Rate: Set to 25 fps.
- Total Frames: Set to capture 200 frames, resulting in an 8-second movie.
- Resolution: Adjust so that the entire well is visible. The priority is a sharp image that allows the organism's movement to be distinguished from the background. Higher resolution can be traded for a larger field of view to image multiple wells at once.
- Environmental Control: Perform captures in a controlled environment to minimize external vibrations and consistent lighting, which is critical for accurate pixel variance analysis.
Step 3: Data Acquisition and Analysis
- Record Videos: Capture videos for each well according to the configured settings.
- Run Paragon Analysis: Process the 200-frame movies using the INVAPP/Paragon system in MATLAB. The algorithm will [3]:
- Calculate the variance over time for each pixel.
- Set a threshold for "motile" pixels (e.g., those with a variance greater than one standard deviation from the mean).
- Generate a motility score for each well by counting the motile pixels.
Step 4: Validation and Calibration
- Benchmark with Controls: Validate the entire setup, including image capture settings, by using control wells (DMSO-only) and wells treated with a known active compound (e.g., temephos for larvae [7] or vorapaxar for C. elegans [3]).
- Dose-Response Curves: Generate a dose-response curve using a reference larvicide to ensure the system can reliably quantify time- and concentration-dependent effects [7].
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key materials required to establish the INVAPP-based screening protocol.
Table 2: Essential Research Reagent Solutions for INVAPP Assays
| Item | Function/Application | Example & Notes |
|---|---|---|
| INVAPP/Paragon System | Hardware and software platform for automated, high-throughput video capture and motility analysis of invertebrates in multi-well plates [7]. | Consists of a camera setup and MATLAB scripts for analysis (available at https://github.com/fpartridge/invapp-paragon) [3]. |
| Model Organisms | Used as phenotypic reporters for anthelmintic or larvicidal activity in chemical screens. | Mosquito larvae (Anopheles gambiae, Ae. aegypti) [7] or the free-living nematode Caenorhabditis elegans [3]. |
| Chemical Libraries | Source of compounds for high-throughput repurposing screens to identify novel bioactive molecules. | The Medicines for Malaria Venture (MMV) Pathogen Box or Pandemic Response Box libraries [7] [3]. |
| 96-Well Plates | Standardized format for housing organisms and compounds during assays, enabling high-throughput screening. | Assay plates are used for liquid cultures of larvae or nematodes exposed to chemical compounds [7] [3]. |
| Positive Control Compounds | Benchmark compounds used to validate assay performance and sensitivity. | Temephos (established larvicide) [7] or Tolfenpyrad (identified from MMV library screen) [7]. For nematodes, Vorapaxar (MMV1593515) is a potent anthelmintic [3]. |
Optimizing image capture to a standardized 25 frames per second for an 8-second duration is a critical success factor for the INVAPP/Paragon platform. This configuration robustly supports the high-throughput, quantitative phenotypic screening necessary for discovering new classes of larvicides and anthelmintics, as demonstrated in peer-reviewed studies. Adherence to this protocol ensures the generation of consistent, high-quality motility data required for reliable chemical screening.
Data Normalization Strategies to Account for Background Well Variation
Within high-throughput chemical screening using the INVertebrate Automated Phenotyping Platform (INVAPP), data quality is paramount. The INVAPP system enables high-throughput, quantitative measurement of larval mosquito motility in response to chemical compounds [4] [5]. A key challenge in such automated, multi-well plate-based assays is the presence of systematic background variation across wells. These variations can arise from technical artifacts such as plate edge effects, pipetting inconsistencies, or minor environmental fluctuations during the experiment. If unaccounted for, this background noise can obscure true biological signals, leading to both false positives and false negatives during chemical screening. Data normalization provides a structured set of solutions to this problem, transforming raw motility data into reliable, biologically meaningful results. This document outlines proven normalization strategies, framed within the context of INVAPP-based research, to eliminate unwanted variation and ensure the robust identification of novel larvicides.
Understanding Data Normalization
Definition and Purpose
Data normalization refers to the process of adjusting values measured on different scales to a common scale to reduce redundancy and improve data integrity [23]. In the specific context of INVAPP, it is the process of cleaning and scaling raw motility data (e.g., movement trajectories, velocity, or activity counts) to minimize the impact of well-to-well and plate-to-plate technical variation.
The primary purposes are to:
- Reduce Unwanted Variation: Systematically remove technical biases and background noise from the dataset [24].
- Improve Data Integrity: Ensure that the observed changes in larval motility are a result of the applied chemical treatment and not experimental artifacts [23] [25].
- Enable Accurate Comparison: Make motility data from different wells, plates, and experimental batches directly comparable [23].
- Enhance Screening Accuracy: Increase the sensitivity and specificity of high-throughput screens for identifying novel larvicides [5].
The Critical Need in INVAPP Screening
The INVAPP platform, combined with the Paragon algorithm, quantifies larval motility in a multi-well format, offering a high-throughput alternative to manual WHO larval assays [5]. This high-throughput capacity was demonstrated in a screen of the Medicines for Malaria Venture (MMV) Pathogen Box library, which successfully identified tolfenpyrad as an effective larvicide [4] [5]. In such screens, ensuring data consistency is non-negotiable. Unnormalized data can lead to:
- Mischaracterization of Compound Efficacy: A potent compound in a well affected by a technical artifact might be misclassified as inactive.
- Inefficient Use of Resources: False leads necessitate costly and time-consuming follow-up experiments.
- Compromised Resistance Monitoring: When comparing larval progeny from insecticide-resistant and sensitive mosquitoes, subtle phenotypic differences can be masked by background noise [5].
Normalization Strategies for Consistently Well-Performing (CWP) Results
A comprehensive bioinformatics study on metabolomics data, which shares similarities with high-throughput phenotyping data, found that single, standalone normalization methods often fail to perform consistently well across multiple assessment criteria (e.g., reducing intragroup variation, maintaining marker stability, and preserving classification capability) [24]. The study discovered that 21 novel strategies, which combined a 'sample'-based method with a 'metabolite'-based method, were Consistently Well-Performing (CWP) under all criteria [24]. The table below summarizes key CWP-enabled strategies derived from this approach, adapted for INVAPP data.
Table 1: Consistently Well-Performing (CWP) Normalization Strategies for INVAPP Data
| Strategy Type | Component Methods | Key Function | Recommendation for INVAPP |
|---|---|---|---|
| Combined Sample + Metabolite | Cubic Splines (Sample) + Range Scaling (Metabolite) | Non-linear baseline correction followed by scaling using the data range. | Highly recommended for complex plate layouts with non-linear trends. |
| Combined Sample + Metabolite | Cyclic Loess (Sample) + Level Scaling (Metabolite) | Robust local regression between sample pairs, scaled by the mean. | Effective for removing intensity-dependent biases across plates. |
| Combined Sample + Metabolite | EigenMS (Sample) + Auto Scaling (Metabolite) | Removes bias via singular value decomposition, then scales to unit variance. | Ideal for datasets with unknown or complex confounding factors. |
| Single Method (Sample-based) | Cubic Splines | Makes the distribution of activity values similar for all samples/wells. | A robust single method for making well distributions comparable [24]. |
| Single Method (Sample-based) | Total Sum | Assigns a weight to each well to minimize overall differences. | Simple and effective for global scaling, assuming most wells are controls [24]. |
| Single Method (Metabolite-based) | Level Scaling | Scales features relative to the average activity across all wells. | Useful for highlighting relative changes in motility features [24]. |
Experimental Protocol for Normalizing INVAPP Data
This protocol details the application of a CWP strategy (Cyclic Loess + Level Scaling) to raw motility data generated by the INVAPP system and processed by the Paragon algorithm.
Materials and Reagents
- INVAPP Hardware: The automated phenotyping platform for video capture [5].
- Paragon Software: Algorithm for converting video data into quantitative motility metrics [5].
- Multi-well Plates: Standard plates containing mosquito larvae and chemical treatments.
- Statistical Software: R or Python with necessary data processing libraries.
Procedure
Step 1: Data Extraction and Compilation
- Run the INVAPP/Paragon system on your multi-well plate to generate raw motility data for each well (e.g., activity counts, movement frequency) [5].
- Export the data matrix where rows represent individual wells and columns represent the quantified motility features and treatment conditions.
- Include designated positive (e.g., a known larvicide like temephos) and negative (untreated/DMSO-only) control wells on every plate [5].
Step 2: Initial Data Quality Control
- Calculate the Z-factor or Z'-factor for the plate using the positive and negative controls to assess the assay's robustness and signal dynamic range.
- Visually inspect the raw data for spatial trends across the plate (e.g., using a heatmap of raw activity counts). Identify potential edge effects or systematic drifts.
Step 3: Apply Sample-Based Normalization (Cyclic Loess)
- Objective: To remove systematic sample-to-sample (well-to-well) variation.
- Action: Implement the Cyclic Loess method, which normalizes by comparing all pairs of samples and fitting a curve using non-linear local regression [24]. This step adjusts the data to make the distribution of motility values comparable across all wells, effectively correcting for plate-wide biases.
Step 4: Apply Metabolite-Based Normalization (Level Scaling)
- Objective: To make the motility features more comparable and normally distributed.
- Action: Perform Level Scaling on the output from Step 3. For each motility feature (e.g., velocity), divide the value by the mean of that feature across all wells [24]. This scales the data relative to the average activity, emphasizing relative changes.
Step 5: Validation and Analysis
- Re-calculate the Z'-factor post-normalization to confirm data quality improvement.
- Re-inspect the data with a heatmap to confirm the reduction of spatial biases.
- Proceed with downstream statistical analysis and hit-calling for chemical screening.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for INVAPP-Based Larval Screening
| Item | Function | Example/Note |
|---|---|---|
| INVAPP Platform | Automated video capture and initial tracking of larval motility. | Custom-built system for high-throughput phenotyping [5]. |
| Paragon Algorithm | Converts video tracks into quantitative, scalar motility metrics. | Essential for generating the numerical data for normalization [5]. |
| Reference Larvicide | Serves as a positive control for assay validation and normalization. | Temephos is an established reference compound [5]. |
| Chemical Libraries | Source of novel compounds for high-throughput screening. | e.g., MMV Pathogen Box [4] [5]. |
| Mosquito Strains | Provide larval subjects; resistant strains allow for resistance monitoring. | e.g., WHO-classified deltamethrin-resistant and sensitive An. coluzzii [5]. |
| Smartphone Camera | Potential alternative for field-based larval motility assessment. | Demonstrates the platform's adaptability for portable assays [5]. |
Workflow and Data Relationships
The following diagram illustrates the logical workflow for processing INVAPP data, from raw video to normalized results, highlighting where normalization fits into the broader analytical pipeline.
The adaptation of the INVertebrate Automated Phenotyping Platform (INVAPP) for field use via smartphone cameras represents a significant advancement in portable, high-throughput chemical screening. This protocol details the methodology for leveraging smartphone-based imaging and sensing technologies to quantify phenotypic changes in invertebrates exposed to chemical compounds, enabling rapid toxicity assessment and drug efficacy testing outside traditional laboratory settings. By transforming consumer smartphones into robust phenotyping instruments, researchers can perform real-time, in-situ analysis of morphological and behavioral endpoints critical for early-stage chemical screening. The system capitalizes on the sophisticated cameras, sensors, and processing power inherent in modern mobile devices, making sophisticated bioanalysis accessible in resource-limited environments while maintaining data integrity and experimental rigor [26].
The integration of smartphone technology addresses several key challenges in large-scale chemical screening: reducing equipment costs, increasing geographical flexibility for field studies, and enabling rapid phenotypic assessment. Recent advances in digital phenotyping demonstrate that mobile devices can capture rich, multidimensional data on biological systems with sufficient resolution for quantitative analysis [27]. When properly calibrated and standardized, smartphone-based INVAPP provides researchers with a powerful tool for environmental monitoring, pharmaceutical field testing, and educational applications where traditional laboratory infrastructure is unavailable or impractical.
Digital phenotyping refers to the moment-by-moment quantification of the individual-level human phenotype in-situ using data from smartphones and other personal digital devices [27]. While initially developed for human behavioral assessment, these principles apply directly to invertebrate phenotyping for chemical screening. The adaptation of INVAPP for smartphone cameras utilizes this approach to capture high-frequency data on invertebrate morphology, locomotion, and behavior in response to chemical exposure.
Modern smartphones contain technological components that can be repurposed for procedural tasks commonly used in image-guided biological assessments [26]. The miniaturization of sophisticated sensors has transformed mobile devices into capable scientific instruments. For INVAPP implementation, the smartphone camera serves as the primary data acquisition tool, capturing visual phenotypes that can be quantified through image analysis algorithms. When combined with other built-in sensors—including accelerometers, gyroscopes, and magnetometers—the smartphone becomes a comprehensive platform for multimodal phenotypic assessment in field conditions [26].
The economic and practical advantages of smartphone-based phenotyping are substantial. Traditional laboratory phenotyping systems require specialized, expensive equipment and controlled environments, limiting their deployment for field studies. Smartphone-based INVAPP dramatically reduces these barriers while maintaining the statistical power needed for meaningful chemical screening. This approach aligns with the growing emphasis on New Approach Methodologies (NAMs) in toxicology and chemical safety assessment, which prioritize faster, more cost-effective, and more human-relevant testing strategies [28].
Technical Specifications and Requirements
Smartphone Hardware Considerations
Successful implementation of smartphone-based INVAPP requires specific hardware capabilities to ensure consistent, high-quality data acquisition across devices and field conditions. The camera system represents the most critical component, with specific requirements for resolution, frame rate, and low-light performance.
Table 1: Minimum Smartphone Camera Specifications for INVAPP Field Deployment
| Parameter | Minimum Specification | Optimal Specification | Functional Impact |
|---|---|---|---|
| Resolution | 12 MP | 48 MP or higher | Determines detail capture for morphological analysis |
| Frame Rate | 30 fps | 60-120 fps | Enables tracking of rapid behavioral endpoints |
| Sensor Size | 1/2.5" | 1/1.7" or larger | Improves low-light performance and reduces noise |
| Aperture | f/2.0 | f/1.8 or wider | Increases light capture in suboptimal conditions |
| Focal Length | Fixed wide (26-28mm equivalent) | Multi-lens system with wide and telephoto | Provides flexibility for different specimen sizes |
| Stabilization | Digital | Optical Image Stabilization (OIS) | Reduces motion blur during field recording |
Beyond camera specifications, other smartphone sensors contribute significantly to field phenotyping. The inertial measurement unit (IMU), which typically integrates a gyroscope, accelerometer and magnetometer, provides critical metadata on device orientation and stability during data capture [26]. This information enables automated correction for minor device movements in field environments, ensuring consistent imaging angles across recording sessions. For chemical screening applications requiring precise environmental context, smartphones with environmental sensors (ambient temperature, pressure, and humidity) provide valuable secondary data for interpreting phenotypic responses.
Research Reagent Solutions
Field deployment of INVAPP requires carefully selected reagents and materials to support specimen viability, experimental consistency, and reproducible phenotypic assessment under variable conditions.
Table 2: Essential Research Reagent Solutions for Field-Based INVAPP
| Reagent/Material | Function | Specification Notes |
|---|---|---|
| Specimen Transport Medium | Maintains invertebrate viability during transport to field sites | Isotonic formulation with pH buffering; chemical compatibility with target compounds |
| Microplate Field Carriers | Secure specimen positioning for imaging | Lightweight, shatter-resistant materials with registration markers for consistent camera alignment |
| Reference Color Standards | Ensures color calibration across lighting conditions | Neutral gray and color reference cards for white balance and exposure calibration |
| Dimensional Calibration Targets | Converts pixel measurements to absolute units | Microscale with precisely spaced patterns; material with minimal thermal expansion |
| Field Staining Solutions | Enhances visual contrast for specific phenotypic features | Stabilized formulations for field use; validated for minimal behavioral impact |
| Portative Environmental Control | Maintains specimen temperature during assessment | Compact, battery-powered incubation sleeves with precise temperature regulation |
| Mobile Data Storage | Secure field data backup | Ruggedized, high-capacity portable solid-state drives with encryption |
| Compound Library Plates | Organized chemical exposure in field settings | Miniaturized, sealed well plates with puncture-resistant sealing films |
Experimental Protocol for Field Deployment
Pre-Field Setup and Calibration
Equipment Preparation
- Charge all power banks and ensure smartphone battery is at 100% capacity
- Initialize INVAPP application and confirm all modules are functioning
- Perform full camera calibration using provided reference standards
- Verify storage capacity and enable automated cloud backup if connectivity available
- Sterilize all specimen containers and microplates using approved methods
Camera Calibration Protocol
- Mount smartphone in stable field stand or rig
- Position dimensional calibration target in specimen imaging area
- Using INVAPP calibration module, capture images at 5 focal depths
- Application automatically calculates pixel-to-millimeter conversion factors
- Validate calibration by imaging secondary reference standard
- Store calibration profile for experimental session
Chemical Library Preparation
- Pre-load compound libraries into specialized field microplates
- Implement positive and negative controls in designated wells
- Seal plates with gas-permeable membrane to prevent evaporation and contamination
- Maintain plates at appropriate temperature during transport to field site
- Verify compound stability under anticipated field conditions
Field Imaging and Data Acquisition
Environmental Assessment and Setup
- Document field conditions (temperature, humidity, ambient light) using smartphone sensors or external probes
- Select imaging location with stable, vibration-free surface
- Position portable backdrop to minimize visual noise in images
- Set up diffused lighting source to eliminate shadows and reflections
- Allow equipment to acclimate to ambient conditions for 10 minutes before imaging
Specimen Transfer and Imaging
- Transfer invertebrates to prepared microplates using appropriate handling tools
- Allow 5-minute acclimation period before initial imaging
- Position plate in imaging rig, ensuring parallel alignment with camera sensor
- Verify focus and exposure using INVAPP auto-calibration function
- Initiate automated imaging sequence:
- Capture brightfield images at 3 focal planes for morphological analysis
- Record 30-second video segments for behavioral assessment
- Repeat at 5-minute intervals for duration of experiment
- Monitor image quality throughout session using quality control metrics
- Annotate observations directly within application using voice-to-text or manual entry
Data Management in Field Conditions
- Implement automated backup to secure digital storage every 15 minutes
- Apply lossless compression to optimize storage utilization
- Verify data integrity through checksum validation after each transfer
- Tag datasets with comprehensive metadata (GPS coordinates, environmental parameters)
- Maintain duplicate storage until full laboratory transfer is completed
Data Processing and Analysis
Image Analysis Workflow The INVAPP application implements automated image processing to extract quantitative phenotypic descriptors from field imagery. The workflow involves sequential analysis steps that transform raw images into validated phenotypic measurements.
Primary Phenotypic Endpoints INVAPP quantifies multiple phenotypic dimensions that serve as indicators of chemical effects:
- Morphological Endpoints: Body area, length-to-width ratio, segmentation patterns, structural symmetry
- Locomotor Endpoints: Velocity, angular movement, path tortuosity, thigmotaxis
- Behavioral Endpoints: Feeding rate, reaction to stimulus, social interaction, circadian patterns
- Developmental Endpoints: Growth rate, molting frequency, morphological maturation
Quality Control Metrics
- Focus sharpness score (minimum 0.8 on normalized scale)
- Illumination uniformity (maximum 15% variance across image)
- Specimen integrity confirmation (no physical damage during transfer)
- Tracking continuity (no gaps in behavioral recording)
- Environmental stability (temperature variation < ±1°C during experiment)
Data Interpretation and Hit Selection
Chemical Response Profiling Compounds are evaluated based on their phenotypic fingerprints, which represent the multidimensional response across all quantified endpoints. The INVAPP analysis pipeline implements statistical methods to identify significant deviations from control phenotypes and clusters compounds based on response similarity.
Hit Selection Criteria
- Potency: Magnitude of phenotypic effect relative to controls
- Phenotypic Specificity: Distinctive pattern across multiple endpoints
- Dose-Response Relationship: Consistent effect across concentration gradients
- Temporal Dynamics: Onset and progression of phenotypic changes
- Reproducibility: Consistency across technical and biological replicates
Table 3: Quantitative Thresholds for Hit Selection in Chemical Screening
| Endpoint Category | Significance Threshold | Effect Size Minimum | Quality Requirement |
|---|---|---|---|
| Morphological | p < 0.01 (adjusted) | Cohen's d > 0.8 | CV < 15% in controls |
| Locomotor | p < 0.005 (adjusted) | Cohen's d > 0.7 | Tracking efficiency > 90% |
| Behavioral | p < 0.01 (adjusted) | Cohen's d > 0.6 | Observation density > 95% |
| Developmental | p < 0.05 (adjusted) | Cohen's d > 0.9 | Temporal resolution < 5min |
Validation and Troubleshooting
Field Validation Procedures
Cross-Platform Validation
- Image identical reference specimens across multiple smartphone models
- Compare phenotypic measurements to establish device-specific correction factors
- Verify inter-device reliability using intraclass correlation coefficients (ICC > 0.85 required)
Environmental Robustness Assessment
- Test performance across temperature range anticipated in field conditions
- Evaluate image consistency under varying natural lighting conditions
- Assess vibration tolerance using standardized disturbance simulation
- Verify battery life under continuous imaging load
Technical Validation Metrics
- Spatial Resolution: Minimum of 15 pixels per specimen body width
- Temporal Resolution: Maximum 5-second intervals for behavioral tracking
- Color Fidelity: Delta E < 5 in standardized color reproduction tests
- Measurement Drift: < 3% variation in control measurements over 6-hour session
Common Field Issues and Solutions
Table 4: Troubleshooting Guide for Field Deployment
| Problem | Potential Causes | Solutions | Preventive Measures |
|---|---|---|---|
| Poor Image Focus | Vibration, condensation on lens, auto-focus failure | Enable manual focus, use lens cleaning cloth, implement focus stacking | Use stable platform, pre-wipe lens, disable auto-focus after calibration |
| Variable Lighting | Cloud movement, sun angle changes, shading | Use diffuser panel, implement auto-exposure lock, post-hoc normalization | Schedule imaging during consistent light, use portable LED array |
| Specimen Movement | Environmental disturbances, inadequate acclimation | Increase acclimation time, implement motion stabilization algorithms | Use vibration-dampening platform, optimize container design |
| Battery Depletion | High processing load, background applications, cold temperature | Enable power-saving mode, reduce frame rate, use external battery | Pre-charge all sources, minimize ambient temperature exposure |
| Data Corruption | Storage interruption, application error, magnetic interference | Implement checksum verification, redundant storage, incremental saves | Use high-quality storage media, enable auto-backup, verify writes |
The adaptation of INVAPP for smartphone camera deployment represents a transformative approach to field-based chemical screening. By leveraging the sophisticated imaging capabilities and sensors of modern mobile devices, researchers can perform quantitative phenotypic assessment in diverse field environments without sacrificing data quality or statistical power. The protocols outlined herein provide a comprehensive framework for implementing this methodology, addressing both technical requirements and experimental considerations specific to field deployment.
This smartphone-based phenotyping approach aligns with broader trends in mobile health technology and digital phenotyping [27], extending these principles to invertebrate models for chemical assessment. The integration of augmented reality and sensor technology [26] creates new opportunities for interactive field research and real-time data annotation. As smartphone technology continues to advance, with improvements in computational photography, multi-sensor integration, and onboard processing, the capabilities of field-deployable INVAPP will expand accordingly, opening new possibilities for distributed chemical screening and environmental monitoring across global research networks.
Validating INVAPP: Benchmarking Performance Against Standard Bioassays
Within chemical screening research, the need for rapid and high-throughput phenotyping of insect larvae is paramount. The INVertebrate Automated Phenotyping Platform (INVAPP) represents a technological advancement that addresses key limitations of traditional, manual larval bioassays. This application note provides a detailed comparison between the innovative INVAPP methodology and the World Health Organization (WHO) standard larval assay, focusing on quantitative metrics of speed and efficiency to guide researchers in selecting the appropriate tool for their screening pipelines.
The core distinction between the two methodologies lies in their fundamental operation: the WHO standard is a manual endpoint bioassay, while INVAPP is an automated, continuous monitoring system. This difference drives significant variation in their application, throughput, and data output.
Table 1: Key Characteristics of INVAPP and WHO Standard Larval Assays
| Feature | INVAPP (Automated Phenotyping) | WHO Standard Larval Assay |
|---|---|---|
| Core Principle | Automated, image-based tracking of larval movement in real-time. [29] | Manual assessment of mortality or survival after a fixed exposure period. [30] |
| Assay Readout | Quantitative metrics of locomotion and activity. [29] | Binary outcomes (live/dead) based on a threshold like 10% survival. [30] |
| Data Type | Continuous, high-dimensional phenotypic data. | Categorical, single-endpoint data. |
| Level of Automation | High | Low |
| Throughput Speed | High (parallel processing of multiple larvae) | Low (reliant on manual labor) |
| Labor Intensity | Low (primarily for setup and data analysis) | High (for rearing, monitoring, and scoring) [30] |
Table 2: Quantitative Comparison of Speed and Efficiency
| Performance Metric | INVAPP | WHO Larval Assay |
|---|---|---|
| Assay Duration | Real-time data; short-term (hours) exposure possible. | Requires set exposure (e.g., 24h) plus post-holding period (e.g., 24h). [30] |
| Hands-on Time | Minutes to hours (largely automated). | Several days, including larval rearing. [30] |
| Larval Rearing Need | Can use wild-caught or lab-reared larvae. | Requires standardized, insectary-raised larvae (3-5 days old). [30] |
| Sample Processing Rate | High (limited only by imaging setup). | Low to moderate (constrained by manual scoring). |
| Data Richness | High (kinetic data enables sublethal effect detection). | Low (lethal endpoint only). |
Detailed Experimental Protocols
Protocol for WHO Standard Larval Assay
The WHO larval assay is a well-established protocol for assessing the susceptibility of mosquito larvae to insecticides or the efficacy of larvicides [30].
I. Materials and Reagents
- Test Larvae: 20-25 late 3rd or early 4th instar larvae, reared from a standardized insectary colony. [30]
- Insecticide Dilutions: A series of insecticide concentrations prepared in distilled water, including a control with solvent only.
- Larval Trays: Standard containers (e.g., 500 mL) for holding larvae during exposure.
- Diet: Larval food, such as a mixture of tuna meal, black soldier fly larvae powder, and brewer's yeast. [31]
- Environmental Chamber: To maintain temperature at 25±2°C and relative humidity at 70-80%.
II. Procedure
- Larval Preparation: Rear larvae to the appropriate instar under controlled conditions. A optimal feeding regime of 0.17, 0.33, 0.67, 0.67, and 0.5 mg per larva over the larval stage can be used to ensure synchronized development. [31]
- Exposure: Introduce the test larvae into the containers holding the different insecticide concentrations.
- Holding Period: Maintain the larvae in the exposure containers for 24 hours. Do not provide food during this period.
- Scoring: After 24 hours, record the number of dead larvae in each container. A larva is considered dead if it fails to move when probed. The assay is validated if mortality in the control group is below a certain threshold (e.g., 10%).
- Data Analysis: Calculate percentage mortality for each concentration and determine the lethal concentration (LC50 or LC90) via probit analysis.
Protocol for INVAPP-Based Phenotypic Screening
INVAPP leverages automated imaging and software analysis to quantify larval behavior, offering a rapid and information-rich alternative for chemical screening.
I. Materials and Reagents
- Test Larvae: Wild-caught or lab-reared larvae. Sample size can be optimized based on desired statistical power. [29]
- Multi-well Plates: A clear-bottomed assay plate (e.g., 24 or 96-well), with each well containing a single larva in a small volume of test solution.
- INVAPP Setup: An imaging station with a digital camera, consistent lighting, and a platform to hold the multi-well plate.
- Software: Image analysis software capable of tracking larval movement, such as the AI-driven models used for species identification. [29]
II. Procedure
- Larval Loading: Individually dispense one larva into each well of the multi-well plate containing the chemical solution or control.
- Image Acquisition: Place the plate on the imaging platform and start the recording session. Capture images or video at regular intervals (e.g., 1 frame per second) for a defined period (e.g., 30 minutes to several hours).
- Automated Tracking: Process the recorded video with the tracking software. The AI model will identify the larva in each frame and quantify its movement, generating metrics like distance traveled, velocity, and activity rate. [29]
- Data Analysis: Export the quantitative behavioral data for statistical analysis. Compare treatment groups to controls to identify sublethal or behavioral effects of the chemical exposure.
The following workflow diagram illustrates the key steps and data flow for the INVAPP protocol.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagent Solutions for Larval-Based Assays
| Item | Function/Application | Example/Note |
|---|---|---|
| Standardized Larval Diet | Provides nutrition for rearing healthy, synchronized test larvae. | A blend of 50% tuna meal, 35% black soldier fly larvae powder, and 15% brewer's yeast. [31] |
| Test Chemical Stocks | The compounds or insecticides being screened for biological activity. | Prepared in appropriate solvents (e.g., DMSO, ethanol) and serially diluted. |
| AI-Based Image Analysis Software | The core of INVAPP; automates the tracking and quantification of larval movement. | Trained on thousands of larval images for high accuracy and confidence (up to 99%). [29] |
| Multi-well Assay Plates | Platform for high-throughput, individual larval testing in the INVAPP system. | Clear-bottomed plates compatible with imaging systems. |
| Control Agents | Benchmark substances used to validate assay performance (positive & negative controls). | Includes known insecticides and inert solvent controls. |
The choice between INVAPP and the WHO standard larval assay hinges on the specific goals of the chemical screening program. The WHO assay provides a standardized, lethal endpoint that is valuable for confirming resistance and comparing results across laboratories. In contrast, INVAPP offers a paradigm shift towards speed and data richness, enabling the high-throughput detection of subtle phenotypic changes and sublethal effects long before mortality occurs. For research focused on rapid hit-to-lead compound identification and understanding the nuanced behavioral impacts of chemicals, INVAPP represents a significantly more efficient and informative tool.
Within the framework of research on the INVertebrate Automated Phenotyping Platform (INVAPP), the accurate quantification of compound efficacy is fundamental. This platform, combined with the Paragon algorithm, enables high-throughput, quantitative screening of chemicals by analyzing changes in invertebrate motility [7] [32]. This application note details standardized protocols and presents validation data for using INVAPP to assess the efficacy of established larvicides and anthelmintics. The procedures outlined herein provide a robust framework for quantifying compound effects against mosquito larvae and parasitic nematodes, serving as a critical reference for screening novel chemicals.
Experimental Protocols
Protocol 1: Mosquito Larvicide Screening via Motility Assay
This protocol is designed for high-throughput screening of compounds for larvicidal activity against Anopheles gambiae and Aedes aegypti, using temephos as a reference standard [7] [5].
- Step 1: Larval Preparation. hatch eggs in deoxygenated water. For Anopheles, use 0.001% pond guardian tonic salt; for Aedes, add a crushed yeast tablet. Maintain larvae at 25°C and feed with cat food (Anopheles). Use first or early second-instar larvae (1-2 days post-hatching for Aedes, 5-6 days for Anopheles) for experiments [7].
- Step 2: Larval Harvesting and Plate Setup. Concentrate larvae by passing their swimming water through a 100 μm Nylon mesh cell strainer. Dilute the concentrate until 100 μL contains 5-10 larvae. Using a pipette with a cut-back tip to prevent larval damage, transfer a 100 μL aliquot to each well of a 96-well plate [7].
- Step 3: Compound Application. Add 100 μL of the test compound, dissolved in water from a DMSO stock, to each well. For a reference larvicide, prepare temephos at a final concentration of 10⁻⁴ M. Include control wells with DMSO alone at the same concentration used for compound preparation [7].
- Step 4: Data Acquisition with INVAPP. Place the 96-well plate into the INVAPP system. Film the larvae for a predefined period to capture their motility. The system records moving images for subsequent analysis [7].
- Step 5: Motility Analysis with Paragon. Process the recorded videos using the Paragon algorithm. The algorithm estimates larval motility, providing a quantitative readout of insecticide effect over time and across concentrations [7].
Protocol 2: Nematode Motility Assay for Anthelmintic Efficacy
This protocol is adapted for screening anthelmintics against both free-living (C. elegans) and parasitic nematodes (H. contortus), using ivermectin as a reference compound [32] [33].
- Step 1: Nematode Culture and Synchronization.
- C. elegans: Culture strains (e.g., wild-type N2, IVM-resistant IVR10, IVM-hypersusceptible AE501) on NGM agar plates seeded with OP50 E. coli at 21°C [33]. Synchronize by collecting gravid adults and eggs, treating with a bleaching mixture (5 M NaOH, 1% hypochlorite) to lyse all stages except eggs, and hatch eggs overnight in M9 buffer to obtain synchronized L1 larvae [33].
- Parasitic Nematodes: Obtain H. contortus L3 larvae from collected eggs. Use both susceptible and known resistant isolates for comparative studies [33].
- Step 2: Assay Setup. Transfer a synchronized population of L4 or young adult C. elegans or L3 larvae of H. contortus to 96-well plates. Add the test compound, such as ivermectin, dissolved in DMSO, at the desired concentrations. Include vehicle control wells with DMSO alone [33].
- Step 3: Motility Measurement. Use the WMicrotracker (WMicrotracker One) system or INVAPP to measure nematode motility. The WMicrotracker automatically records motility as the number of worm movements detected per unit of time [33].
- Step 4: Data Analysis. Generate dose-response curves from the motility data. Calculate the half-maximal inhibitory concentration (IC50) for each compound or strain. Determine Resistance Ratios (RR) by comparing IC50 values of resistant and susceptible isolates [33].
Table 1: Key Reagents for Nematode Anthelmintic Screening.
| Reagent / Strain | Description | Source / Reference |
|---|---|---|
| C. elegans N2 Bristol | Wild-type, anthelmintic-susceptible strain | Caenorhabditis Genetics Center (CGC) [33] |
| C. elegans IVR10 | IVM-selected, resistant strain | [33] |
| C. elegans AE501 | nhr-8(ok186), IVM-hypersusceptible mutant | CGC [33] |
| H. contortus isolates | Susceptible (S-H-2022) and resistant (R-EPR1-2022) field isolates | [33] |
| Ivermectin (IVM) | Macrocyclic lactone anthelmintic; reference compound | Sigma-Aldrich [33] |
| Moxidectin (MOX) | Macrocyclic lactone anthelmintic | Sigma-Aldrich [33] |
| NGM Agar | Growth medium for C. elegans culture | [33] |
Validation with Known Compounds
Larvicide Screening Validation
The INVAPP/Paragon system was validated using the established larvicide temephos. The system reliably quantified time- and concentration-dependent actions on larval motility, providing a faster readout than the WHO standard larval assay [7]. Furthermore, a screen of the Medicines for Malaria Venture (MMV) Pathogen Box library identified tolfenpyrad as a potent larvicide, confirming the platform's capacity for library-scale chemical screening [7] [5].
The system also demonstrated utility in detecting phenotypic differences related to insecticide resistance. Larvae derived from WHO-classified deltamethrin-resistant and sensitive adult mosquitoes showed differential responses in the INVAPP assay, indicating its potential for monitoring larval behavioral resistance [7].
Anthelmintic Screening Validation
Quantitative motility assays have been successfully used to validate the effects of known anthelmintics and to identify resistance in nematodes.
Table 2: Efficacy of Macrocyclic Lactones Against Susceptible and Resistant Nematodes.
| Nematode Strain / Isolate | Compound | IC₅₀ / EC₅₀ | Resistance Factor (RF) | Citation |
|---|---|---|---|---|
| C. elegans N2B (susceptible) | Ivermectin (IVM) | 1 (Reference) | - | [33] |
| C. elegans IVR10 (resistant) | Ivermectin (IVM) | 2.12 | 2.12 | [33] |
| C. elegans N2B (susceptible) | Moxidectin (MOX) | 1 (Reference) | - | [33] |
| C. elegans IVR10 (resistant) | Moxidectin (MOX) | 1.51 | 1.51 | [33] |
| H. contortus (susceptible) | Moxidectin (MOX) | Highest efficacy | - | [33] |
| H. contortus (resistant) | Eprinomectin (EPR) | Significant RF | Substantial RF reported | [33] |
A screen of the Pathogen Box library using INVAPP/Paragon against parasitic nematodes identified known anthelmintics including tolfenpyrad, auranofin, and mebendazole, as well as novel chemotypes with anthelmintic activity [32].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for INVAPP-based Screening.
| Item | Function / Application | Examples / Notes |
|---|---|---|
| INVAPP Hardware | Automated imaging platform for high-throughput filming of invertebrate motility in multi-well plates. | Custom-built system for recording movement [7] [32]. |
| Paragon Algorithm | Software for analyzing video recordings from INVAPP to estimate motility. | Provides quantitative readout of compound effects [7] [32]. |
| WMicrotracker One | Alternative instrument that automates motility measurement via infrared detection. | Used for nematode anthelmintic screening [33]. |
| 96-well Plates | Standard format for housing larvae/nematodes during screening. | Enables high-throughput chemical testing [7] [33]. |
| Reference Larvicides | Positive controls for assay validation. | Temephos, Tolfenpyrad [7] [5] [32]. |
| Reference Anthelmintics | Positive controls for anthelmintic assays. | Ivermectin, Moxidectin, Mebendazole [32] [33]. |
| Mosquito Strains | Insects for larvicide screening; include resistant and sensitive varieties. | Anopheles gambiae G3 (sensitive), An. coluzzii Tiassale (resistant) [7]. |
| Nematode Strains | Model and parasitic worms for anthelmintic screening. | C. elegans (various mutants), H. contortus (field isolates) [32] [33]. |
Visual Workflows and Pathways
Workflow for Larvicide Screening
Workflow for Anthelmintic Resistance Detection
Ivermectin Mechanism and Resistance Pathways
Within chemical screening research, the INVertebrate Automated Phenotyping Platform (INVAPP) has emerged as a powerful tool for quantifying phenotypic responses to compounds. A primary application is the high-throughput discrimination between insect strains characterized as resistant (R) or sensitive (S) to various insecticides [7]. This capability is critical for understanding resistance mechanisms and for screening new compounds to combat the growing threat of insecticide resistance in vector-borne diseases.
The fundamental principle involves using automated, image-based analysis of larval motility as a robust and quantifiable phenotype. The INVAPP system, in conjunction with the Paragon algorithm, tracks and quantifies motility changes in response to chemical exposure, providing a rapid and reliable readout that can differentiate resistant from sensitive populations [7]. This protocol details the application of INVAPP for this purpose, framing the results within standardized susceptibility categories—Susceptible (S), Intermediate (I), and Resistant (R)—which are defined by the likelihood of therapeutic success or failure for a given compound [34] [35].
Key Definitions and Susceptibility Categories
The interpretation of efficacy for any anti-infective agent relies on standardized categories. The following definitions, as harmonized by the European Committee on Antimicrobial Susceptibility Testing (EUCAST), provide the context for classifying the responses observed in INVAPP assays [35]:
- Susceptible (S): A microorganism is categorized as "Susceptible" when there is a high likelihood of therapeutic success using a standard dosing regimen of the agent. In the context of INVAPP, this would translate to a strain showing significant phenotypic change (e.g., lethality, reduced motility) at a standard concentration of the insecticide.
- Susceptible, Increased Exposure (I): A microorganism is categorized as "Susceptible, Increased exposure" when there is a high likelihood of therapeutic success because exposure to the agent is increased. For larval screening, this may indicate efficacy only at higher concentrations or with adjusted exposure times.
- Resistant (R): A microorganism is categorized as "Resistant" when there is a high likelihood of therapeutic failure even when there is increased exposure. In INVAPP assays, resistant strains show minimal phenotypic change even at high insecticide concentrations [34] [35].
Table 1: Definitions of Susceptibility Categories
| Category | Acronym | Definition | Implied Therapeutic Likelihood |
|---|---|---|---|
| Susceptible | S | High likelihood of success with a standard regimen [35]. | High |
| Susceptible, Increased Exposure | I | High likelihood of success only with increased exposure (e.g., higher dose) [35]. | High (with adjustments) |
| Resistant | R | High likelihood of failure even with increased exposure [35]. | Low |
Quantitative Validation of the INVAPP System
The INVAPP/Paragon system has been quantitatively validated against established insecticides and in comparisons of known resistant and sensitive mosquito strains. The system's output provides a motility metric that is more sensitive and rapid than manual, visual endpoint assessment.
Table 2: Representative Data from INVAPP Validation Studies
| Experiment Type | Strain / Compound | Key Quantitative Result | Significance |
|---|---|---|---|
| Larvicide Profiling | Temephos (organophosphate) | Reliably quantified time- and concentration-dependent reduction in larval motility [7]. | Faster and more objective than WHO standard larval assay. |
| Chemical Screening | MMV Pathogen Box Library | Identified tolfenpyrad as a potent hit; confirmed efficacy in subsequent validation [7]. | Demonstrates utility for high-throughput screening (HTS) of large chemical libraries. |
| Resistance Detection | Ae. aegypti: Cayman (R) vs. New Orleans (S) | Detected significant difference in motility response between resistant and sensitive strains [7]. | Enables monitoring of resistance prevalence and mechanisms. |
| Resistance Detection | An. coluzzii: Tiassalé (R) vs. An. gambiae G3 (S) | Showed differential larval response in progeny from deltamethrin-resistant adults [7]. | Platform can detect behavioural resistance linked to adult insecticide resistance. |
Research Reagent Solutions
The following table details the essential materials and reagents required to implement the INVAPP-based resistance screening protocol.
Table 3: Key Research Reagents and Materials
| Item Name | Function/Application | Example Specification |
|---|---|---|
| INVAPP Hardware Platform | Automated imaging system for high-throughput filming of larval motility in multi-well plates [7]. | Custom-built system for 96-well plate format. |
| Paragon Algorithm | Software for analyzing video data from INVAPP to calculate a quantitative motility index [7]. | - |
| Mosquito Strains | Genetically characterized resistant (R) and sensitive (S) strains for comparative screening [7]. | e.g., Ae. aegypti: Cayman (R) and New Orleans (S); An. coluzzii: Tiassalé (R) and G3 (S). |
| Reference Larvicide | Positive control compound for assay validation and quality control [7]. | e.g., Temephos (organophosphate). |
| 96-Well Plate | Standardized vessel for holding larvae and compound solutions during filming [7]. | Flat-bottom, clear. |
| Cell Strainer | For harvesting and concentrating larvae from maintenance trays [7]. | 100 μm Nylon mesh. |
Detailed Experimental Protocol
Larval Preparation and Plate Setup
- Hatching and Maintenance: Receive eggs of the characterized resistant and sensitive mosquito strains (e.g., Aedes aegypti or Anopheles gambiae complex). Hatch eggs in deoxygenated (pre-boiled and cooled) tap water. Maintain resulting larvae at 25°C and feed with appropriate food (e.g., crushed yeast tablet for Ae. aegypti, cat food pellet for Anopheles) [7].
- Harvesting Larvae: On the day of the assay (day 3-4 for Ae. aegypti, day 5-6 for Anopheles), pass the water containing the larvae through a 100 μm nylon mesh cell strainer to concentrate them [7].
- Preparing Larval Suspension: Dilute the concentrated larval suspension until a 100 μL aliquot contains approximately 5-10 larvae.
- Compound Dilution: Prepare serial dilutions of the test insecticide(s) in water, typically from a DMSO stock solution. Include a vehicle control (DMSO in water at the same final concentration as used for the compounds) [7].
- Plate Loading: Using a pipette with a cut-back tip to avoid damaging larvae, add 100 μL of the larval suspension to each well of a 96-well plate. Then, add 100 μL of the insecticide solution or vehicle control to the respective wells. This results in a final test volume of 200 μL per well [7].
Data Acquisition with INVAPP
- Platform Setup: Place the loaded 96-well plate into the INVAPP imaging chamber.
- Filming Parameters: Film the plate for a predefined period to establish a baseline motility index. The specific duration may vary but is designed to capture sufficient data for robust analysis [7].
- Data Collection: The INVAPP system automatically captures video data of larval movement in each well over time.
Data Analysis with the Paragon Algorithm
- Motility Index Calculation: Process the recorded videos using the Paragon algorithm. The algorithm analyzes the moving images to generate a quantitative measure of larval motility for each well [7].
- Dose-Response Curves: For each insecticide, plot the motility index (or a derived endpoint, such as percent inhibition) against the logarithm of the compound concentration. Fit a sigmoidal curve to determine the half-maximal effective concentration (EC₅₀).
- Strain Comparison: Calculate the resistance ratio (RR) by dividing the EC₅₀ of the resistant strain by the EC₅₀ of the sensitive strain. An RR significantly greater than 1 indicates confirmed resistance.
- Categorization: Classify strain responses based on the RR and the absolute EC₅₀ values in the context of achievable field concentrations (where applicable), following the principles of the S/I/R definitions.
The INVAPP system, coupled with the Paragon algorithm, provides a sensitive, reliable, and high-throughput method for differentiating between resistant and sensitive insect strains. By converting larval motility into a quantitative data stream, it offers a robust phenotypic readout that is superior to manual, end-point observations. This protocol enables rapid chemical screening and resistance monitoring, which are essential for developing new vector control strategies and managing the spread of insecticide resistance.
The integration of automation and advanced imaging has revolutionized phenotyping, enabling large-scale genetic and chemical screens. Automated systems span diverse applications, from invertebrate chemical screening to mammalian behavioral analysis and plant phenomics. Among these, the INVertebrate Automated Phenotyping Platform (INVAPP) has emerged as a specialized system for high-throughput screening of mosquito larvae, offering distinct advantages in speed and scalability for vector control research [5]. This application note provides a detailed, quantitative comparison of INVAPP's throughput against other automated phenotyping systems, contextualizing its performance within the broader field while providing actionable protocols for researchers.
The critical need for such platforms is underscored by the growing threat of mosquito-borne diseases and the urgent requirement for novel insecticides. INVAPP addresses key limitations of traditional manual assays, such as the World Health Organization (WHO) standard larval assay, by providing an objective, rapid, and scalable alternative [5]. Meanwhile, parallel advancements in other domains, such as the smart-Kage for rodent cognitive studies and PhenoApp for plant phenotyping, demonstrate how automation is transforming data collection across biological research [36] [37].
Comparative System Performance & Throughput Benchmarks
Quantitative Performance Metrics
Table 1: Key Performance Metrics Across Automated Phenotyping Systems
| System / Platform | Primary Application | Throughput Capability | Key Performance Metrics | Assay Duration |
|---|---|---|---|---|
| INVAPP + Paragon [5] | Mosquito larvae chemical screening | High-throughput; library-scale screening | Reliable quantification of time- and concentration-dependent insecticide actions faster than WHO standard assay; Enabled screening of MMV Pathogen Box library (~400 compounds) | Fast readout; specific durations not provided |
| smart-Kage [37] | Mouse cognitive & behavioral phenotyping | Long-term continuous monitoring; large-scale screening | Combined statistical properties of multiple behaviors can discriminate between lesion models and predict Alzheimer's disease genotype with 80-90% accuracy | Long-term (days to weeks); fully automated and continuous |
| PhenoApp [36] | Plant phenotyping (field/greenhouse) | Mobile data acquisition; improves efficiency of manual phenotyping | Digital recording compliant with MIAPPE/FAIR data principles; uses standardized BBCH scales; accelerates labor-intensive data acquisition | Adaptable to experiment duration; replaces manual recording |
Throughput Analysis and Context
INVAPP is explicitly designed for high-throughput chemical screening, as demonstrated by its successful application in screening the Medicines for Malaria Venture (MMV) Pathogen Box library [5]. This capability positions it as a discovery tool within chemical biology and vector control research. Its core advantage lies in quantifying larval motility as a robust endpoint for insecticide action, providing a more rapid and objective assessment than the moribund appearance and failure to respond to tapping used in the WHO standard larval assay [5].
In contrast, the smart-Kage system specializes in the unattended, long-term phenotyping of cognitive functions in mice. Its throughput is characterized by the ability to concurrently run multiple behavioral tasks (T-maze, novel object recognition, object-in-place) in a home-cage environment without human intervention or food/water restriction [37]. While not focused on chemical screening, its throughput advantage lies in continuous multi-parameter data collection from individual animals over extended periods.
PhenoApp addresses throughput from a different angle by digitizing and standardizing the flow of phenotypic data during manual collection in field or greenhouse settings [36]. It increases efficiency by ensuring data is structured, Findable, Accessible, Interoperable, and Reusable (FAIR) from the point of collection, thereby reducing a major bottleneck in plant breeding and genebank management.
Detailed Experimental Protocols
Protocol: INVAPP for Larvicide Screening
Objective: To perform high-throughput, quantitative screening of chemical compounds for larvicidal activity against Anopheles gambiae or Aedes aegypti mosquito larvae using the INVAPP system.
Materials & Reagents:
- Mosquito Larvae: Late 3rd or early 4th instar larvae from desired species (e.g., Anopheles coluzzii) [5].
- Chemical Library: Compounds dissolved in appropriate solvent (e.g., DMSO). The MMV Pathogen Box is cited as a proof-of-concept [5].
- INVAPP Hardware: The invertebrate automated phenotyping platform, typically involving a multi-well plate setup and an imaging system.
- Paragon Software: Algorithm for analyzing larval motility from video data [5].
- Multi-well Plates: Standard plates compatible with the INVAPP setup.
- Control Solutions: Negative control (water or solvent) and positive control (e.g., Temephos).
Procedure:
- Larval Preparation: Dispense a single larva into each well of a multi-well plate containing water.
- Compound Administration: Add the test compound to each well to achieve the desired final concentration. Include positive and negative controls on each plate.
- Data Acquisition: Place the multi-well plate into the INVAPP system. Initiate automated video recording of larval activity over a defined period (e.g., 30 minutes).
- Motility Analysis: Process the recorded videos using the Paragon algorithm. This software quantifies motility by analyzing movement frames to generate a time- and concentration-dependent response profile for each well [5].
- Data Output: The primary readout is a motility metric. A significant decrease in motility compared to the negative control indicates larvicidal activity or sub-lethal effects.
Protocol: smart-Kage for Cognitive Phenotyping
Objective: To conduct fully automated, home-cage-based assessment of spatial memory and learning in mouse models using the smart-Kage system.
Materials & Reagents:
- Experimental Mice: Individually housed mice (e.g., Alzheimer's disease model such as AppNL-G-F).
- smart-Kage: The automated home-cage system comprising connected compartments, nose-poke ports with IR sensors, water reservoirs with solenoid valves, and textured surface panels on rotatable drums [37].
- Overhead IR Camera: For continuous recording at approximately 2 frames per second.
- Deep Convolutional Neural Network (CNN) Software: For tracking mouse position with high spatial resolution [37].
Procedure:
- Habituation: Acclimate the mouse to the smart-Kage environment.
- Automated Task Execution: The system concurrently runs multiple behavioral tasks without experimenter intervention:
- Smart T-maze: The mouse must alternate between left and right corridors to activate water release at the nose-poke ports, testing working memory [37].
- Smart Novel Object Recognition (NOR): Textured surface panels are automatically changed (e.g., once every two days), and exploration time of the novel versus familiar texture is quantified.
- Smart Object-in-Place (OIP): The locations of two familiar textures are swapped, and the mouse's exploration of the relocated objects is measured.
- Data Collection: The overhead IR camera continuously records the mouse's position and activity. A random forest classifier is used to infer specific behaviors of interest from trajectory and posture data [37].
- Analysis: Performance on each task is analyzed (e.g., alternation rate in T-maze, discrimination index in NOR/OIP). Combined statistical properties of multiple behaviors can be used for model discrimination or genotype prediction.
Workflow Visualization
Figure 1: A comparative workflow of three automated phenotyping systems. INVAPP focuses on chemical screening in a plate-based format, smart-Kage on continuous behavioral monitoring in a home-cage, and PhenoApp on digitizing manual field observations.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagents and Materials for Automated Phenotyping
| Item | Function / Application | System Context |
|---|---|---|
| MMV Pathogen Box Library | A curated set of ~400 diverse compounds with known activity against pathogens; used for proof-of-concept library-scale larvicide screening. | INVAPP [5] |
| Paragon Algorithm | Software specifically designed to analyze video data from INVAPP, quantifying larval motility as a key endpoint for insecticide action. | INVAPP [5] |
| Deep Convolutional Neural Network (CNN) | Image analysis tool for precise, high-resolution tracking of animal position and posture from video footage. | smart-Kage [37] |
| BBCH Scales | Standardized phenological scales used to precisely identify the growth stages of plants (e.g., cereals, grapevine, maize). | PhenoApp [36] |
| Nose-Poke Port with IR Sensor | A port that an animal must insert its nose into to trigger a sensor; used in automated operant tasks for reward (e.g., water delivery). | smart-Kage [37] |
| Multi-well Plates | Standard labware for holding multiple samples (e.g., larvae in solution); foundational for high-throughput screening formats. | INVAPP [5] |
Discussion & Concluding Analysis
The benchmarking data clearly illustrates that "throughput" is context-dependent. INVAPP excels in rapid, in-vitro chemical screening, a capability critical for early-stage discovery in public health and agrochemistry [5]. Its ability to reliably quantify sub-lethal effects via motility and perform faster than the WHO standard assay makes it a valuable tool for prioritizing lead compounds.
The smart-Kage system demonstrates high throughput in terms of data density and long-term, multi-parameter behavioral analysis in a more ethologically relevant setting [37]. Its strength is not in screening thousands of compounds, but in generating deep, continuous phenotypic profiles for discriminating disease models with high accuracy, which is invaluable for translational neuroscience.
PhenoApp, while not a sensor-based automation platform, addresses a critical throughput bottleneck—the efficiency and quality of data recording in manual phenotyping [36]. Its contribution to throughput is in ensuring data is digitally born and FAIR-compliant, which accelerates downstream analysis and utilization in breeding programs.
In conclusion, INVAPP occupies a specialized and vital niche in the high-throughput phenotyping ecosystem. For researchers focused on insecticide discovery, it offers a robust, scalable, and rapid platform that has been validated against real-world screening challenges. Its integration with potential smartphone-based adaptations, as suggested in the original research, points toward an even more accessible and deployable future for this technology [5].
Conclusion
The INVAPP/Paragon system represents a paradigm shift in phenotypic screening, offering a validated, high-throughput solution that drastically outpaces traditional methods like the WHO larval assay. By providing precise, quantitative data on invertebrate motility, it successfully accelerates the discovery of novel larvicides and anthelmintics, as demonstrated by the identification of tolfenpyrad. Its proven ability to differentiate pyrethroid-resistant mosquito populations further opens avenues for resistance monitoring. Future directions are poised to expand its impact, particularly through the development of a smartphone-based field assay. This innovation promises real-time, geo-located resistance monitoring, potentially transforming public health interventions against mosquito-borne diseases and strengthening the pipeline against parasitic infections.
References
- 1. An automated high-throughput system for phenotypic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5734697/]
- 2. Advances in Anthelmintic Target Identification [https://www.mdpi.com/1422-0067/26/8/3738]
- 3. Screening the Medicines for Malaria Venture (MMV) ... [https://www.frontiersin.org/journals/tropical-diseases/articles/10.3389/fitd.2022.1017900/full]
- 4. Automated phenotyping of mosquito larvae enables high ... [https://pubmed.ncbi.nlm.nih.gov/34081710/]
- 5. Automated phenotyping of mosquito larvae enables high ... [https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0008639]
- 6. Discovery of small molecules with anthelmintic potential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12502323/]
- 7. Automated phenotyping of mosquito larvae enables high ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8205174/]
- 8. Whole-organism phenotypic screening methods used in ... [https://www.sciencedirect.com/science/article/pii/S0734975022000337]
- 9. C. elegans expressing D76N β2-microglobulin: a model for ... [https://www.nature.com/articles/s41598-019-56498-5]
- 10. Screening a chemical library on An. gambiae larvae using ... [https://figshare.com/articles/figure/Screening_a_chemical_library_on_i_An_i_i_gambiae_i_larvae_using_the_INVAPP_Paragon_system_/14726863]
- 11. An automated high-throughput system for phenotypic ... [https://www.sciencedirect.com/science/article/pii/S2211320717301057]
- 12. Automated, high-throughput, motility analysis in ... [https://www.sciencedirect.com/science/article/pii/S2211320714000347]
- 13. GitHub - fpartridge/invapp-paragon: Analysis of movies of worms to score motility or growth [https://github.com/fpartridge/invapp-paragon]
- 14. Protocol for an automated virtual screening pipeline including ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12639438/]
- 15. Tolfenpyrad [https://www.nichino-europe.com/products/tolfenpyrad/]
- 16. Drug screens using the nematode Caenorhabditis elegans [https://pmc.ncbi.nlm.nih.gov/articles/PMC12406009/]
- 17. The Future of Drug Discovery: Integrating Phenotypic Data ... [https://ardigen.com/the-future-of-drug-discovery-integrating-phenotypic-data-with-omics-and-ai/]
- 18. High Throughput Screening Market Forecast, 2025-2032 [https://www.coherentmarketinsights.com/industry-reports/high-throughput-screening-market]
- 19. 8-Methoxypeucedanin: Evaluation of Anxiolytic Effects and ... [https://www.mdpi.com/1422-0067/26/21/10259]
- 20. A novel inhibitor rescues cerebellar defects in a zebrafish ... [https://www.sciencedirect.com/science/article/pii/S0021925821006529]
- 21. Loss of Polo ameliorates APP-induced Alzheimer's disease ... [https://www.nature.com/articles/srep16816]
- 22. Soy Protein-Cultured Mesenchymal Stem Cell-Secreted ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11897944/]
- 23. Data Normalization Explained: Types, Examples, & Methods [https://risingwave.com/blog/data-normalization-explained-types-examples-and-methods/]
- 24. A novel bioinformatics approach to identify the consistently ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7711263/]
- 25. Data Normalization, Explained: What is it, Why it's ... [https://blog.invgate.com/data-normalization]
- 26. Smartphone Technology for Applications in Image-Guided ... [https://link.springer.com/article/10.1007/s00270-024-03925-4]
- 27. Harnessing Digital Phenotyping for Early Self-Detection ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12386097/]
- 28. 2025 Society of Toxicology Satellite Meeting [https://www.epa.gov/chemical-research/new-approach-methods-training-2025-society-toxicology-satellite-meeting]
- 29. Artificial Intelligence and Citizen Science as a Tool for ... [https://www.mdpi.com/2075-4450/16/11/1098]
- 30. Adult-capture assays as a tool to measure insecticide ... [https://www.frontiersin.org/journals/malaria/articles/10.3389/fmala.2025.1489687/full]
- 31. Optimizing larval mass-rearing techniques for Aedes mosquitoes [https://pmc.ncbi.nlm.nih.gov/articles/PMC12087348/]
- 32. An automated high-throughput system for phenotypic screening of ... [https://ora.ox.ac.uk/objects/uuid:73ba4c30-2199-4a48-9bcd-2a80c08a1064]
- 33. Evaluation of nematode susceptibility and resistance to ... [https://www.nature.com/articles/s41598-025-02866-3]
- 34. Susceptible, Intermediate, and Resistant – The Intensity of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2701059/]
- 35. Definition of S, I and R [https://www.eucast.org/bacteria/clinical-breakpoints-and-interpretation/definition-of-s-i-and-r/]
- 36. PhenoApp: A mobile tool for plant phenotyping to... [https://f1000research.com/articles/11-12]
- 37. A fully automated home cage for long-term continuous ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10391580/]