Fasciola hepatica: A Comprehensive Analysis of Life Cycle, Egg Morphology, and Implications for Diagnostics and Drug Development
This article provides a detailed synthesis of the complex life cycle and critical egg morphology of the parasitic trematode Fasciola hepatica, tailored for researchers and drug development professionals.
Fasciola hepatica: A Comprehensive Analysis of Life Cycle, Egg Morphology, and Implications for Diagnostics and Drug Development
Abstract
This article provides a detailed synthesis of the complex life cycle and critical egg morphology of the parasitic trematode Fasciola hepatica, tailored for researchers and drug development professionals. It covers the foundational biology of its heteroxenous life cycle, spanning mammalian and snail hosts, and the defining characteristics of its eggs, which remain the diagnostic gold standard. The content delves into advanced methodologies for laboratory maintenance and egg analysis, addresses pressing challenges such as anthelmintic resistance, and validates findings through comparative morphometrics and molecular techniques. By integrating recent discoveries, such as novel egg appendages and the role of the glycocalyx in host-parasite interactions, this review aims to inform the development of novel diagnostic, therapeutic, and vaccine strategies against a significant zoonotic pathogen.
The Biological Blueprint: Deconstructing the Fasciola hepatica Life Cycle and Egg Architecture
Fascioliasis, caused by the parasitic trematode Fasciola hepatica (the common liver fluke), represents a significant global zoonotic disease with substantial economic impacts on livestock production and emerging importance in human public health [1] [2]. This digenean fluke employs a complex heteroxenous life cycle that alternates between an invertebrate intermediate host and a mammalian definitive host, with several free-living stages in the environment [3]. The intricate developmental pathway of F. hepatica involves five principal phases encompassing both hosts and the external environment, requiring precise biological and environmental conditions for successful transmission [3]. Within the context of broader research on the life cycle and egg morphology of F. hepatica, understanding these developmental transitions provides critical insights for diagnostic improvements, drug development, and transmission control strategies. This technical guide provides an in-depth analysis of each life cycle stage, supported by experimental data and methodologies relevant to researchers, scientists, and drug development professionals working in parasitology and tropical medicine.
The Heteroxenous Life Cycle: Stage-by-Stage Analysis
Eggs and Miracidial Development
The life cycle initiation begins when adult flukes residing in the bile ducts of the mammalian host release eggs that are passed into the environment through the host's feces [1]. These eggs are broadly ellipsoidal, operculated, and measure 130-150 µm long by 60-90 µm wide when passed unembryonated in stool [1]. Recent morphological studies have identified that some F. hepatica eggs possess a previously unreported abopercular appendage, a finding that may contribute to improved diagnostic differentiation [4] [5].
Table 1: Developmental Characteristics of Fasciola hepatica Eggs
| Parameter | Specification | Experimental Conditions |
|---|---|---|
| Size | 130-150 µm by 60-90 µm [1] | Microscopic measurement of fixed samples |
| Embryonation Time | Approximately 2 weeks [1] | Freshwater incubation at 20-30°C |
| Hatching Temperature Threshold | Inhibited below 10°C, optimal between 20-30°C [6] | Controlled laboratory incubation |
| Distinctive Morphological Features | Abopercular appendage, knob, egg shell thickening [4] [5] | Light and electron microscopy |
Under favorable environmental conditions in freshwater, including appropriate temperature parameters, the eggs undergo embryonation over approximately two weeks, eventually hatching to release ciliated miracidia [1]. These miracidia are motile, free-swimming larvae that must rapidly locate and invade a suitable snail intermediate host, as they can survive only for a few hours outside the snail [7].
Intramolluscan Stages
Upon successfully penetrating a susceptible lymnaeid snail intermediate host (particularly species in the genera Galba, Fossaria, and Pseudosuccinea), the miracidium undergoes a complex developmental multiplication process within the snail [1]. This intramolluscan phase involves several transformational stages:
- Sporocysts: The initial developmental stage following miracidial penetration
- Rediae: Daughter stages that develop within sporocysts
- Cercariae: The final larval stage that emerges from the snail [1]
Table 2: Intramolluscan Development Parameters of F. hepatica
| Developmental Stage | Duration | Temperature Dependence |
|---|---|---|
| Sporocyst Development | Variable, dependent on temperature | Snail growth rates peak at 25°C [6] |
| Rediae Multiplication | Multiple generations | Cercarial shedding accelerates around 27°C [6] |
| Total Intramolluscan Phase | Several weeks | Snail susceptibility to infection depends on temperature [6] |
The entire intramolluscan development is highly temperature-dependent, with snail growth rates peaking at approximately 25°C and cercarial shedding accelerating around 27°C [6]. The specific snail intermediate hosts for F. hepatica belong to the family Lymnaeidae, with at least 20 snail species identified as competent intermediate hosts [1]. In experimental settings, Galba pervia snails have been successfully used to maintain laboratory life cycles and obtain metacercariae [8].
Metacercarial Formation and Mammalian Infection
The cercariae released from infected snails encyst as metacercariae on aquatic vegetation or other substrates [1]. These metacercariae represent the infective stage for the mammalian host and are remarkably resilient in the environment [7]. Metacercarial viability is prolonged by higher humidity, though it declines at elevated temperatures [6].
Mammalian hosts, including humans, become infected by ingesting metacercariae-contaminated vegetation such as watercress [1]. Following ingestion, the metacercariae excyst in the duodenum in response to the action of gastric juices, releasing the newly excysted juveniles (NEJ) [8] [2]. These juvenile flukes penetrate through the intestinal wall into the peritoneal cavity, then migrate through the liver parenchyma to reach the biliary ducts [1] [2]. This migratory phase through the liver parenchyma typically lasts several weeks and causes significant tissue damage [2].
Development into Adult Flukes
Upon reaching the bile ducts, the immature flukes complete their maturation into adult flukes and commence egg production [1]. The maturation process from metacercariae to egg-producing adults generally requires approximately 3-4 months in the definitive host [1]. Adult F. hepatica are large, broadly-flattened parasites measuring up to 30 mm long and 15 mm wide, with a distinctive cone-shaped anterior end [1]. These adults reside in the bile ducts of the liver, where they can survive for extended periods, continuously producing eggs that are excreted into the environment to complete the life cycle [1].
Environmental Influence on the Life Cycle
The successful completion of the F. hepatica life cycle is profoundly influenced by environmental conditions, particularly temperature and humidity [6]. Each developmental stage exhibits specific temperature thresholds and optima, creating a complex relationship between climatic fluctuations and parasite transmission potential.
Table 3: Temperature Dependence Across F. hepatica Life Cycle Stages
| Life Cycle Stage | Temperature Inhibition | Optimal Temperature | Key Climate Influences |
|---|---|---|---|
| Egg Hatching | Below 10°C [6] | 20-30°C [6] | Requires freshwater; inhibited by desiccation |
| Miracidial Survival | Limited to hours [7] | Not specified | Requires thin films of moisture for migration |
| Snail Development | Variable with species | 25°C (snail growth peak) [6] | Prefers muddy, slightly acidic conditions with poor drainage [7] |
| Cercarial Shedding | Reduced at lower temperatures | Accelerates around 27°C [6] | Requires suitable temperature and moisture |
| Metacercarial Survival | Declines at higher temperatures | Prolonged by higher humidity [6] | Highly resilient stage; survives on wet herbage |
The transmission dynamics are further complicated by the requirement for specific snail intermediate hosts that prefer muddy, slightly acidic conditions associated with poor drainage [7]. This ecological specificity means the incidence of fasciolosis is considerably greater in wetter geographical regions and during years with above-average summer rainfall [7]. Climate change has consequently emerged as a significant factor influencing the distribution and prevalence of fasciolosis, with warmer and wetter conditions potentially expanding endemic regions [6].
Laboratory Models for Life Cycle Maintenance
Animal Model Systems
Experimental maintenance of the complete F. hepatica life cycle requires suitable laboratory animal models. Recent comparative studies have evaluated three small animal models to determine their suitability for laboratory investigations:
- Rabbits: Exhibit acute suppurative hepatitis with mortality occurring 60-69 days post-infection; eggs appear in feces around day 63 post-infection [8]
- SD Rats: Demonstrate punctate liver lesions by day 3 post-infection with subsequent pathological changes, but show liver repair by week 9; survive over one year post-infection and successfully maintain the life cycle [8]
- Kunming Mice: Develop severe liver pathology similar to but more pronounced than SD rats, with mortality observed by day 31 post-infection [8]
These comparative studies indicate that while all three species can serve as experimental hosts, SD rats represent more suitable models due to their superior tolerance to infection and defined pathological response [8].
Infection Methodologies
Standardized laboratory infection protocols have been developed for life cycle maintenance:
Diagram 1: Laboratory Life Cycle Workflow. The workflow illustrates the standardized procedure for maintaining F. hepatica in laboratory conditions, highlighting the critical transition between intermediate (yellow) and definitive (green) hosts.
For definitive host infection, metacercariae are administered orally to experimental animals. Typical infection doses in research settings include:
- Rabbits: 15-30 metacercariae per animal [8]
- SD Rats: 20-30 metacercariae per animal [8]
- Kunming Mice: 2-10 metacercariae per animal, with dose-dependent survival observed [8]
Experimental Approaches and Research Tools
Proteomic Analysis of Intra-Mammalian Stages
Advanced proteomic studies have identified 689 F. hepatica proteins across intra-mammalian stages, representing the most comprehensive protein catalog for this species to date [2]. Critical findings include:
- Proteinases dominate the excretory/secretory (E/S) products of both NEJ and adult flukes, representing 83% and 73% of total proteins respectively [2]
- Cathepsin L and B families constitute the majority of these proteinases [2]
- Stage-specific expression patterns with 52 proteins shared between NEJ and adult E/S products, while 90 proteins are unique to NEJ and 202 unique to adults [2]
These proteomic profiles provide valuable insights into the host-parasite interface and identify potential targets for diagnostic and therapeutic development.
Metabolic Studies and Drug Target Identification
Recent investigations into the metabolic adaptations of F. hepatica have revealed a sophisticated hybrid respiration system that enables the parasite to thrive in varying oxygen concentrations throughout its migration in the mammalian host [9].
Diagram 2: Dual Respiration Pathways in F. hepatica. The parasite employs different respiratory chains depending on oxygen availability, utilizing distinct electron mediators for aerobic (red) and anaerobic (blue) conditions.
This metabolic plasticity is particularly crucial for NEJs, which transition through various oxygen concentrations during migration from the duodenum to the bile ducts [9]. Adult flukes predominantly utilize fumarate respiration with rhodoquinone-10 (RQ10) as the primary electron mediator, while NEJs demonstrate metabolic flexibility, employing both aerobic and anaerobic pathways appropriate to their microenvironment [9]. This essential fumarate respiration pathway in both developmental stages represents a promising drug target, as it is absent in mammalian hosts [9].
Pharmacological Research and Drug Entry Mechanisms
Investigations into anthelmintic pharmacology have elucidated critical mechanisms of drug entry and action in F. hepatica. Studies with closantel (CLS), a salicylanilide derivative effective against adult flukes, have demonstrated:
- Oral ingestion represents the primary route of drug entry into adult F. hepatica, rather than trans-tegumental diffusion [10]
- Drug accumulation in flukes correlates positively with plasma concentrations in the host [10]
- The presence of bile markedly diminishes CLS diffusion into parasites under ex vivo conditions [10]
- CLS lacks significant ovicidal activity at therapeutically relevant concentrations [10]
These findings highlight the importance of understanding drug pharmacokinetics and entry mechanisms for optimizing chemotherapeutic efficacy against fascioliasis.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for F. hepatica Life Cycle Studies
| Reagent/Model | Specification | Research Application |
|---|---|---|
| Intermediate Host Snails | Galba pervia, G. truncatula, G. humilis [1] [8] | Maintenance of intramolluscan stages; metacercariae production |
| Definitive Host Models | SD Rats, Kunming Mice, Rabbits [8] | Study of parasite migration, pathogenesis, and drug efficacy |
| Proteomic Analysis | LC-MS/MS protein identification [2] | Characterization of somatic and E/S products; vaccine target identification |
| Metabolic Inhibitors | Rotenone (Complex I), Atpenin A5 (Complex II), Ascochlorin (Complex III) [9] | Investigation of respiratory pathways; validation of drug targets |
| Immunodiagnostic Antigens | Recombinant FhSAP2 (~38 kDa) [1] | Serological detection of pre-patent infections; disease surveillance |
| Culture Media | RPMI 1640 for ex vivo incubation [10] | Drug uptake studies; parasite maintenance outside host |
The heteroxenous life cycle of Fasciola hepatica represents a sophisticated biological adaptation that ensures parasite survival and transmission across disparate environments and hosts. From egg morphology through intramolluscan development to complex intra-mammalian migration, each stage presents unique vulnerabilities that may be targeted for disease control. Contemporary research has significantly advanced our understanding of the proteomic, metabolic, and pharmacological aspects of host-parasite interactions, providing novel insights for diagnostic improvement and therapeutic development. The ongoing challenges of triclabendazole resistance and climate-driven changes in transmission patterns underscore the imperative for continued investigation into the fundamental biology of this pervasive parasite. Future research directions should prioritize integrated approaches that combine traditional parasitological methods with emerging omics technologies to identify and validate novel intervention targets throughout the parasite's developmental cycle.
Fasciola hepatica, commonly known as the sheep liver fluke, is a parasitic trematode that infects the livers of various mammals, including humans, causing a disease known as fascioliasis [11]. As a digenetic parasite, F. hepatica requires a definitive mammalian host to reach sexual maturity and an intermediate molluscan host to complete its life cycle [11]. Within the definitive host, the parasite undertakes a complex and precise migratory pathway, moving from the digestive system through the liver parenchyma to ultimately reside in the biliary ducts, where it matures and reproduces [1]. This whitepaper examines the definitive host spectrum, the molecular and cellular mechanisms underlying hepatic migration, and the establishment of chronic infection in the biliary system, providing a technical foundation for researchers and drug development professionals working within the broader context of F. hepatica life cycle and egg morphology research.
Definitive Host Spectrum and Infection Dynamics
Fasciola hepatica demonstrates a broad host range, primarily infecting herbivorous mammals but capable of parasitizing a wide variety of definitive hosts. The parasite's mammalian host specificity is relatively low, enabling it to establish infections in numerous species across different geographic distributions [11] [12].
Quantitative Host Range and Prevalence
Table 1: Definitive Hosts and Natural Infection Prevalence of Fasciola hepatica
| Host Category | Specific Hosts | Reported Natural Infection Prevalence/Notes | Primary Research Context |
|---|---|---|---|
| Primary Domestic Hosts | Cattle, Sheep, Goats | Major economic losses; main reservoirs for human infection [11] | Global agricultural systems [11] |
| Wild Ruminants | African Buffalo, Water Buffalo | Act as definitive hosts in endemic areas [11] | Field studies [11] |
| Lagomorphs (Wild) | Lepus capensis (Hare) | 39.2% infection prevalence [12] | Natural watercress beds, Central France [12] |
| Oryctolagus cuniculus (Rabbit) | 42.0% infection prevalence [12] | Natural watercress beds, Central France [12] | |
| Sylvilagus floridanus | 25.0% infection prevalence [12] | Natural watercress beds, Central France [12] | |
| Newly Documented Hosts | Rangifer tarandus (Reindeer) | Confirmed as final host based on egg morphology and ITS rDNA analysis [4] [5] | Palearctic region survey [4] [5] |
| Human Infections | Humans | ~2.6 million people affected globally; ~90 million at risk [13] | Zoonotic transmission [13] |
| Other Mammals | Camelids, Cervids | Infections reported [1] | Regional epidemiological studies [1] |
The table illustrates that lagomorphs, particularly rabbits and hares, can serve as significant wildlife reservoirs, with studies in central France's watercress beds showing high natural infection rates exceeding 39% in some species [12]. Recent research has genetically confirmed reindeer (Rangifer tarandus) as a definitive host for F. hepatica, expanding the known host range and suggesting the Novaya Zemlya archipelago as potentially the northernmost site of fasciolosis [4] [5]. Humans act as accidental definitive hosts, with an estimated 2.6 million people infected globally and up to 90 million at risk, classifying fascioliasis as a neglected tropical disease [13] [11].
Hepatic Migration: Pathways and Molecular Mechanisms
The migratory journey of F. hepatica from the intestinal lumen to the biliary system represents a critical phase of infection, involving precisely coordinated parasite movements and molecular interactions with host tissues.
Migration Route and Timeline
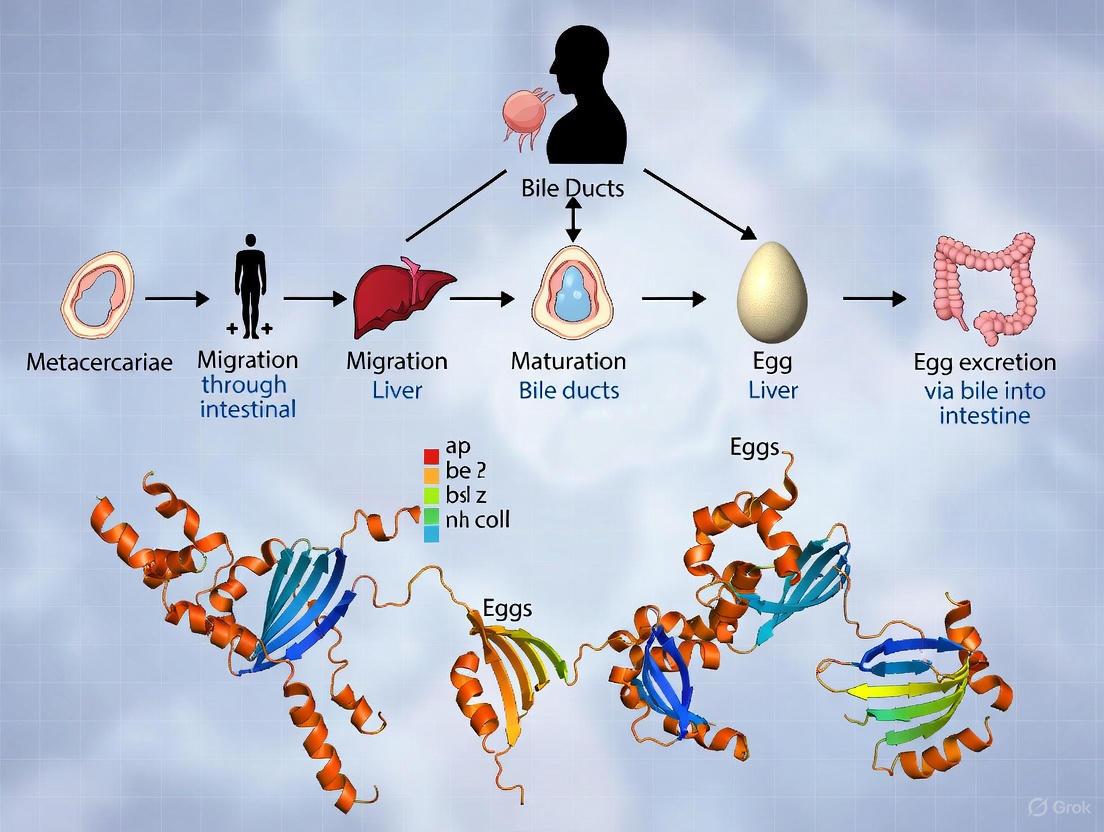
Following excystment in the duodenum, newly excysted juveniles (FhNEJ) burrow through the intestinal wall into the peritoneal cavity [11]. From there, they penetrate the liver capsule and migrate through the liver parenchyma for 6-8 weeks before entering the biliary ducts, where they mature into adults [1] [11]. The entire maturation process from metacercariae to adult fluke in the bile ducts takes approximately 3-4 months in humans [1] [11].
The diagram below illustrates the detailed migratory pathway of Fasciola hepatica juveniles from excystment through biliary establishment.
Proteomic Changes During Early Migration
Recent research using quantitative SWATH-MS proteomics has identified significant molecular changes in FhNEJ during intestinal wall crossing, revealing proteins critical for host-parasite interactions [13].
Table 2: Key Proteomic Changes in FhNEJ During Gut Passage
| Protein Category | Specific Proteins | Regulation Direction | Putative Function in Migration |
|---|---|---|---|
| Proteolytic Enzymes | Cathepsin L3, Cathepsin L4 | Upregulated | Tissue penetration, nutrient acquisition, immune evasion [13] |
| Protease Inhibitors | Fh serpin 2 | Upregulated | Regulation of proteolytic activity, immune modulation [13] |
| Nutrient Uptake & Metabolism | Low-density lipoprotein receptor, Fatty acid binding protein, Glutathione-S-transferase | Upregulated | Nutrient acquisition, detoxification, metabolism [13] |
| Structural & Regulatory | Histone H4, H2A, H2B, Tetraspanin | Upregulated | Gene regulation, tegument structure [13] |
| Stress Response | HSP90, Alpha crystallin | Downregulated | Reduced stress response during active migration [13] |
The upregulation of cathepsin L peptidases and other proteolytic enzymes facilitates tissue penetration through degradation of extracellular matrix components, while their secretion helps modulate host immune responses [13]. The simultaneous upregulation of nutrient acquisition molecules suggests a metabolic shift to support the energy demands of active migration. The downregulation of heat shock proteins like HSP90 may indicate a specific adaptation to the host environment, potentially reducing parasite visibility to the host immune system [13].
Biliary Residence and Chronic Infection
Upon reaching the biliary ducts, F. hepatica establishes a chronic infection that can persist for years, with adult flukes capable of surviving in the bile ducts for 20-30 years if untreated [14].
Anatomical Adaptations for Biliary Residence
Adult F. hepatica possesses specialized anatomical features that enable long-term survival in the biliary environment:
- Tegument: A syncytial, anucleate epithelium covered by a scleroprotein layer and glycocalyx that protects against host digestive juices and immune responses [11]. The tegument contains numerous folds to increase surface area for nutrient absorption and spines that aid in attachment [11].
- Attachment Organs: Powerful oral and ventral suckers provide firm anchorage within the bile ducts [11] [15].
- Digestive System: A blind gut without anus, adapted for extracellular digestion; waste materials are egested through the mouth [11].
- Reproductive System: Hermaphroditic adults with both male and female reproductive organs capable of producing up to 25,000 eggs per fluke per day [11].
Pathological Consequences and Disease Phases
Table 3: Clinical Manifestations of F. hepatica Infection in Definitive Hosts
| Disease Phase | Timeline | Primary Pathology | Clinical Manifestations |
|---|---|---|---|
| Acute Phase (Migratory/Invasive) | 2-3 days to 3-4 months post-infection | Larval migration through liver parenchyma causing tissue destruction, inflammation, and toxic/allergic reactions [1] | Fever, chills, abdominal pain (especially right upper quadrant), hepatomegaly, nausea, vomiting, urticaria, malaise, eosinophilia, elevated liver transaminases [14] [1] |
| Chronic Phase (Biliary/Adult) | Months to years post-infection | Inflammation, hyperplasia, and fibrosis of bile ducts; mechanical obstruction [14] [1] | Intermittent abdominal pain, jaundice, weight loss, cholangitis, cholecystitis, pancreatitis, biliary stones [14] [1] |
| Long-Term Complications | Years post-infection | Biliary fibrosis, liver cirrhosis, bile duct cancer (cholangiocarcinoma) [14] | Chronic hepatic insufficiency, portal hypertension, increased risk of cholangiocarcinoma [14] |
The chronic inflammatory response to adult flukes in the bile ducts can lead to hyperplasia and fibrosis of the biliary epithelium [14]. Heavy or long-standing infections may cause significant mechanical obstruction of the biliary tract [14]. The World Health Organization has classified F. hepatica as a Group 1 biological carcinogen due to its association with cholangiocarcinoma, particularly in endemic areas [14].
Experimental Models and Methodological Approaches
Ex Vivo Intestinal Migration Model
An advanced ex vivo model system has been developed to study the early host-parasite interactions during intestinal wall crossing, a critical stage in infection establishment [13].
Experimental Protocol:
- FhNEJ Preparation: Metacercariae are excysted in vitro using a CO₂-sodium dithionite system followed by incubation in excystment medium (HEPES, rabbit bile, Hank's balanced salt solution) at 37°C for 4 hours [13].
- Intestinal Preparation: Mouse duodenum and jejunum sections (15cm) are collected, lumens flushed with PBS, and ends ligated [13].
- Inoculation: Approximately 2,500 FhNEJ in 200μL excystment medium are introduced into the intestinal lumen [13].
- Incubation: Injected intestines are placed in RPMI medium and incubated for 2.5 hours at 39°C with 5% CO₂ [13].
- Sample Analysis: FhNEJ that successfully cross the intestinal wall are collected for comparative proteomic analysis using SWATH-MS [13].
Proteomic Workflow for Migration Studies
The diagram below outlines the integrated experimental and analytical workflow for identifying proteomic changes during gut migration.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for Studying F. hepatica-Host Interactions
| Reagent/Category | Specific Examples | Research Application | Function in Experimental Design |
|---|---|---|---|
| Parasite Material | F. hepatica metacercariae (Italian strain), Adult flukes from slaughterhouses | Infection studies, life cycle maintenance | Source of biological material for excystment, infection models, and antigen preparation [13] [16] |
| Excystment Reagents | Sodium dithionite, CO₂, HEPES, Rabbit bile, Hank's balanced salt solution | In vitro excystment protocol | Mimics host intestinal conditions to trigger metacercariae to release infective FhNEJ [13] |
| Proteomic Reagents | SWATH-MS, iTRAQ, MALDI-TOF MS/MS | Protein identification and quantification | High-throughput identification and quantification of differentially expressed proteins in host-parasite interactions [13] [16] |
| Immunoassay Reagents | Recombinant FhSAP2 antigen, ES antigens, HRP-conjugated antibodies | Serodiagnosis, antibody detection | Detection of host immune response to infection; diagnosis of acute and chronic fascioliasis [1] |
| Cell Culture Media | RPMI medium, PBS, Hanks' solution | Maintenance of host cells and tissues | Supports viability of ex vivo intestinal models and cell cultures during infection studies [13] |
The intricate relationship between Fasciola hepatica and its definitive hosts involves a precisely orchestrated sequence of migration, establishment, and long-term survival within the biliary system. The expanding known host range, including recent confirmation of reindeer as competent hosts, demonstrates the parasite's adaptability [4] [5]. The molecular mechanisms underlying hepatic migration, particularly the proteomic changes in FhNEJ during gut passage, reveal potential targets for therapeutic intervention [13]. The chronic phase of infection, characterized by the fluke's sophisticated adaptations to the biliary environment, results in significant pathology including potential carcinogenesis [14]. Future research directions should focus on exploiting the identified migration-related proteins for vaccine development, understanding the mechanisms of immune modulation in the biliary niche, and developing novel chemotherapeutic approaches that target both juvenile and adult stages, particularly in light of emerging triclabendazole resistance [13]. Integrating this molecular understanding with the parasite's complete life cycle, including recently updated egg morphology details [4] [5], provides a comprehensive foundation for developing next-generation control strategies against this globally significant parasite.
Within the comprehensive research on the life cycle and egg morphology of Fasciola hepatica, the phase of development within the intermediate host represents a critical amplification and transmission bottleneck. The sheep liver fluke, Fasciola hepatica, is a parasitic trematode of significant medical and veterinary importance, causing the disease fascioliasis in ruminants and humans worldwide [1]. This trematode possesses a complex, indirect life cycle that is obligately dependent on a freshwater snail intermediate host to progress from a single miracidium to the production of numerous infective cercariae [17]. Understanding the molecular and cellular mechanisms governing snail penetration and the subsequent intramolluscan larval development is not only fundamental to parasitology but also provides a foundation for identifying novel targets for drug and intervention strategies aimed at disrupting the parasite's life cycle. This whitepaper provides an in-depth technical guide to the processes of host finding, penetration, and the intricate sequence of larval stages within the snail, synthesizing classical morphology with contemporary experimental data.
Snail Host Finding and Miracidial Penetration
The first contact between the parasite and its intermediate host is mediated by the miracidium, a free-swimming, ciliated larval stage that hatches from the egg in water. The success of this phase is governed by a narrow temporal window and a precise series of host-recognition behaviors.
Miracidium Biology and Host Finding
The miracidium of F. hepatica is a pyriform, motile larva measuring 150-200 µm in length and is covered with ciliated epidermal plates [17]. It does not feed and is entirely reliant on its endogenous energy reserves, which limit its lifespan in water to less than 24 hours [18] [17]. Within this short period, it must locate and invade a suitable snail host. The miracidium exhibits a positive chemotactic response to chemical cues released by specific lymnaeid snails, guiding it through the aquatic environment [17]. The primary intermediate hosts for F. hepatica are snails of the family Lymnaeidae, with the species varying geographically; Galba truncatula is the major host in Europe, parts of Asia, Africa, and South America, while other species like Lymnaea neotropica and Pseudosuccinea columella are important in the Americas [1] [11].
Mechanism of Snail Penetration
Upon contact with a compatible snail, the miracidium adheres to the host's tissues, typically the foot, antenna, or gill, using its apical papilla [19]. The penetration process is both mechanical and enzymatic:
- Mechanical Action: The miracidium uses its apical papilla to actively probe and create an initial entry point into the snail's soft tissues [19].
- Enzymatic Degradation: Glandular sacs associated with the papilla release proteolytic enzymes that break down the snail's extracellular matrix and facilitate tissue penetration [19].
Following successful entry, the miracidium migrates to specific sites within the snail, most commonly the digestive gland and gonads, which provide a nutrient-rich environment for subsequent development [17]. Immediately upon establishing itself in the snail tissue, the miracidium undergoes a profound morphological transformation: it casts off its ciliated epidermis, loses its sensory organs, and swells to form the next larval stage, the sporocyst [19].
Table 1: Key Characteristics of the Fasciola hepatica Miracidium
| Characteristic | Specification |
|---|---|
| Origin | Hatches from an operculated egg passed in the feces of the definitive host [17] |
| Size | 150-200 µm in length [17] |
| Lifespan | < 24 hours (must find a host within 8-30 hours for optimal success) [18] [19] |
| Locomotion | Free-swimming via ciliated epidermal plates [17] |
| Host Finding | Positive chemotaxis to chemical cues from lymnaeid snails [17] |
| Penetration Apparatus | Apical papilla, penetration glands [19] |
| Target Snail Tissues | Pulmonary sac, foot, antennae; subsequent migration to digestive gland [17] [19] |
Intramolluscan Larval Development and Multiplication
The development within the snail is characterized by a sequence of larval stages, each with distinct morphology and function, and involves extensive asexual multiplication, which is a key amplification step in the life cycle.
Sporocyst Stage
The sporocyst is the first developmental stage within the snail. It appears as a simple, sac-like germinal body, lacking a mouth, gut, or other complex organs [19]. Its body wall is covered by a thin cuticle, beneath which are mesenchymal cells and muscle fibers [19]. The internal cavity of the sporocyst is filled with germinal cells, which are descended directly from the original germ cells of the miracidium [19]. The primary function of the sporocyst is asexual reproduction. The germinal cells develop into the next larval stage, the redia. A single sporocyst can produce 5 to 8 rediae [19]. The sporocyst stage is transient and motile, moving within the host tissues to facilitate the distribution of the next generation.
Redia Stage
Rediae are more morphologically complex than sporocysts and represent a motile, feeding stage. They are elongated, measuring 1.3 mm to 1.6 mm in length, and possess several specialized structures [19]:
- Muscular Pharynx and Sac-like Intestine: Allows the redia to actively feed on snail host tissues, including hepatopancreatic cells and gonads, which provides energy for further development and can cause significant damage to the snail [19].
- Birth Pore: Located near the anterior end, this structure allows mature daughter larvae to exit.
- Ventral Processes (Lappets): Paired, locomotor structures near the posterior end that aid in movement within the host [19].
Rediae can give rise to two types of offspring. During warmer months, a single primary (mother) redia can produce a second generation of daughter rediae, further amplifying the parasite population within the snail. Later, or during winter conditions, the germ balls within the rediae (whether mother or daughter) develop into the next larval stage, the cercaria. Each redia is capable of producing 14 to 20 cercariae [19].
Cercaria Stage
The cercaria is the final larval stage produced within the snail and is the precursor to the infective stage for the definitive host. Cercariae have an oval body (approx. 0.25-0.35 mm long) and a long, simple tail that enables swimming upon emergence [19]. Their anatomy includes rudiments of adult organs, such as an oral sucker, ventral sucker (acetabulum), and a bifurcated intestine [19]. The body contains cystogenous glands that are critical for the next stage of the life cycle. After a developmental period within the snail of approximately 5 to 7 weeks post-miracidial penetration, mature cercariae exit the rediae via the birth pore, migrate through the snail's tissues, and emerge from the snail into the surrounding water [17]. The emergence is often rhythmic and can be influenced by environmental factors such as light and temperature.
Table 2: Quantitative Summary of Intramolluscan Larval Development of Fasciola hepatica
| Larval Stage | Size | Key Morphological Features | Reproductive Output & Function | Approximate Time Post-Infection |
|---|---|---|---|---|
| Sporocyst | ~0.7 mm [19] | Sac-like, no gut, thin cuticle [19] | Asexual multiplication; produces 5-8 rediae [19] | Days 3-10 [20] [19] |
| Redia (Mother) | 1.3-1.6 mm [19] | Muscular pharynx, gut, birth pore, lappets [19] | Feeds on snail tissue; produces 14-20 daughter rediae or cercariae [19] | From day 10-14 onwards [20] [19] |
| Cercaria | Body: 0.25-0.35 mm [19] | Oral/ventral suckers, bifid gut, cystogenous glands, tail for swimming [19] | The final intramolluscan stage; emerges from snail to encyst as metacercaria. Non-feeding. | Emerges from ~day 39 onwards [20] |
The following diagram illustrates the sequence and relationships of these developmental stages within the snail host.
Experimental Protocols for Larval Development Studies
Robust experimental methodologies are essential for investigating the complex biology of F. hepatica's intramolluscan stages. The following protocols, adapted from key studies, provide a framework for such research.
Protocol 1: Snail Infection and Larval Stage Monitoring
This protocol outlines the procedure for experimentally infecting snails and tracking the development of larval stages, based on the methodology used for F. gigantica [20].
Preparation of Miracidia:
- Obtain fresh F. hepatica eggs from the gallbladders of infected ruminants at an abattoir or from laboratory-maintained adult flukes.
- Wash eggs repeatedly with dechlorinated tap water and collect via sedimentation or under a stereomicroscope.
- Incubate approximately 1,000 eggs per well in multi-well plates filled with dechlorinated tap water.
- Incubate at room temperature (27-31°C) under natural light conditions and monitor daily for hatching. Hatching of miracidia typically occurs after 11-12 days under these conditions [20].
Snail Infection:
- Use one-month-old, laboratory-reared susceptible lymnaeid snails (e.g., Galba truncatula).
- Place groups of 20 snails into clay pots or aquaria containing 2 liters of dechlorinated tap water.
- Add approximately 200 actively swimming miracidia to each pot.
- Provide fresh lettuce leaves ad libitum as food for the snails.
Monitoring Larval Development:
- Beginning 24 hours post-exposure (PE), sacrificially crush one exposed snail daily under a stereomicroscope.
- Carefully dissect and examine snail tissues (especially digestive gland and gonads) for the presence and morphology of larval stages (sporocysts, rediae, cercariae).
- Document the first appearance and maturation of each stage. Under optimal conditions, sporocysts can be observed by day 3 PE, rediae by day 10-14 PE, and cercarial shedding can begin around 39 days PE [20].
Protocol 2: Metacercariae Production and Viability Assessment
This protocol describes the production of metacercariae from shed cercariae and the assessment of their viability and infectivity.
Cercariae Collection and Encystment:
- Upon the initiation of cercarial shedding from infected snails (from ~5-7 weeks PE), place mature cercariae in containers with dechlorinated water.
- Introduce suitable substrates for encystment into the water. These can include aquatic vegetation (e.g., watercress, rice plants Oryza sativa), glass slides, or plastic strips [20].
- Allow cercariae to settle and encyst on the substrates for 24-48 hours. The cysts, now metacercariae, will be visible as small, white, spherical structures.
Metacercariae Harvesting and Quantification:
- Carefully remove the substrates from the water.
- For vegetation, use a stereomicroscope to count metacercariae per unit area or weight to standardize the infection dose.
- For artificial substrates, metacercariae can be gently scraped off and suspended in a known volume of water for counting.
Infectivity Assay:
- Use a susceptible experimental definitive host, such as a rodent (e.g., mice, rats) or a lamb.
- Administer a known number of metacercariae (e.g., 30 per mouse) orally via gavage [20].
- Sacrifice animals at predetermined time points post-infection (e.g., every 3-7 days).
- Examine the intestines, peritoneal cavity, liver, and bile ducts for the presence of migrating juvenile or adult flukes.
- Calculate the worm recovery rate as a percentage of the administered metacercariae to determine infectivity. An average recovery rate of 35.8% has been reported in experimental mouse models for F. gigantica [20].
The workflow for these integrated protocols is summarized below.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful research into the intramolluscan stages of F. hepatica requires specific biological materials and reagents. The following table details key components of the experimental toolkit.
Table 3: Key Research Reagent Solutions for Investigating Fasciola-Snail Interactions
| Reagent/Material | Specification / Example | Primary Function in Research |
|---|---|---|
| Fresh Fasciola Eggs | Isolated from infected livestock bile or laboratory-maintained adult flukes. | Source of miracidia for experimental snail infections; fundamental for initiating the life cycle in a lab setting [20]. |
| Susceptible Snail Colony | Galba truncatula, Lymnaea spp., maintained in laboratory aquaria. | Essential intermediate host; required for supporting parasite development from miracidium to cercaria [1] [20]. |
| Dechlorinated Tap Water | Water treated to remove chlorine and chloramines, often by aging or chemical neutralization. | Provides the necessary aquatic environment for egg embryonation, miracidial hatching, snail survival, and cercarial encystment [20]. |
| Recombinant Antigens (e.g., FhSAP2) | Recombinant F. hepatica antigen used in immunoblot assays [1]. | Serological detection of Fasciola infection in definitive hosts; confirms success of experimental infections and assesses vaccine/drug efficacy [1]. |
| Culture Substrates | Aquatic plants (e.g., watercress, rice plants - Oryza sativa), glass slides, plastic strips [20]. | Provides a surface for cercariae to encyst and form metacercariae, enabling the production of the infective stage for definitive host studies [20]. |
| Experimental Definitive Hosts | Mice (Mus musculus), rats, hamsters, or susceptible lambs. | Used in infectivity assays to determine the viability of metacercariae and to study the pathogenicity and immunology of early infection [20]. |
The journey of Fasciola hepatica within its snail intermediate host—from the host-finding miracidium through the asexually amplifying sporocyst and redia stages to the cercaria—is a remarkable example of complex parasitism. This phase is characterized by precise host-parasite recognition, radical morphological transformations, and massive biological amplification, making it an attractive target for intervention. Disrupting any single step in this process, be it miracidial penetration, the metabolic pathways of the redia, or cercarial maturation, has the potential to break the transmission cycle entirely. Future research, leveraging advanced molecular techniques like genomics, transcriptomics, and RNA interference, should focus on identifying and characterizing key parasite molecules involved in snail invasion, immune evasion, and developmental regulation. Such efforts will undoubtedly yield new candidates for vaccines, novel anthelmintic drugs, or snail-specific control agents, contributing to the integrated management of a globally significant neglected tropical disease.
Within the complex life cycle of the trematode Fasciola hepatica, the environmental stages comprising metacercariae encystment and the subsequent mechanisms of infectivity represent a critical nexus for disease transmission and a potential point for therapeutic intervention. This whitepaper dissects the fundamental biological processes that enable the parasite's environmental persistence and successful host invasion. Framed within broader research on life cycle and egg morphology, this analysis integrates the initial stages of egg embryonation with the terminal infective stage of the metacercariae. A deep understanding of these mechanisms is paramount for researchers and drug development professionals aiming to disrupt the parasite's transmission, particularly in the face of growing anthelmintic resistance that threatens current control strategies [21] [22].
The Life Cycle ofFasciola hepatica: Contextualizing Metacercariae
The metacercarial stage is the product of a multi-stage, heteroxenous life cycle involving a definitive mammalian host and an intermediate snail host. The cycle begins when adult flukes in the bile ducts produce eggs that are expelled into the environment via the host's feces [1]. These eggs embryonate in freshwater, typically over approximately two weeks, and release miracidia [1]. The ciliated miracidia must rapidly locate and penetrate a suitable lymnaeid snail intermediate host, such as Galba truncatula [11]. Within the snail, the parasite undergoes several asexual developmental stages: sporocysts, rediae, and finally, cercariae [1] [20]. The free-swimming cercariae are the immediate precursors to the metacercariae. They are released from the snail and, within a short timeframe, encyst on aquatic vegetation or other substrates to become metacercariae, the infective stage for the definitive host [1] [11]. The following diagram illustrates this complete lifecycle, highlighting the central role of the metacercaria.
Environmental Dynamics of Encystment and Survival
The transition from cercaria to metacercaria and the subsequent survival of the cyst are governed by precise environmental conditions. Understanding these parameters is crucial for modeling disease risk and forecasting the impact of climate change on fasciolosis epidemiology.
The Encystment Process and Viability Factors
Upon emergence from the snail, cercariae swim freely and then encyst as metacercariae on vegetation or other solid surfaces [1]. The viability and longevity of these metacercariae are highly dependent on ambient temperature and humidity. Metacercariae are susceptible to desiccation, and their viability is prolonged by higher humidity levels [6]. The following table synthesizes key quantitative data on the effects of temperature on different stages of Fasciola hepatica, based on experimental observations.
Table 1: Temperature Dependence of Fasciola hepatica Development and Transmission Dynamics
| Life Cycle Stage | Temperature Effect | Optimal/Threshold Values | Experimental Context |
|---|---|---|---|
| Egg Hatching | Inhibited below a minimum threshold; hatching time decreases with higher temperatures. | Below 10°C inhibited; optimal between 20–30°C [6]. | Laboratory incubation of eggs in water [6]. |
| Cercarial Shedding | Accelerates with increasing temperature. | Accelerated around 27°C [6]. | Observation of infected snails under controlled temperatures [6]. |
| Metacercarial Viability | Declines at higher temperatures; longevity is prolonged by higher humidity. | Viability declines above optimal range; prolonged by high humidity [6]. | Stability studies of metacercariae under varying climatic conditions [6]. |
| Snail Host Growth | Increases with temperature to a peak. | Peaks at 25°C [6]. | Rearing of snail intermediate hosts in laboratory settings [6]. |
Host Range and Environmental Persistence
The definitive host range for F. hepatica is broad, primarily including domestic and wild ruminants such as sheep, cattle, and goats [1]. Humans can become aberrant hosts, typically through the ingestion of contaminated aquatic vegetation like watercress [1] [11]. Recent research has confirmed that reindeer (Rangifer tarandus) can also act as a final host, suggesting the parasite's potential to spread in arctic and subarctic ecosystems [23]. The resilience of the metacercariae in the environment, which can remain infective for extended periods under suitable moist conditions, is a key factor in the successful transmission and maintenance of the parasite's life cycle across diverse geographic regions.
Molecular and Cellular Mechanisms of Infection
The infectivity of F. hepatica is not a passive process but is driven by a sophisticated arsenal of molecular tools deployed by the parasite. Upon ingestion by the definitive host, the metacercariae excyst in the duodenum, releasing the invasive newly excysted juvenile (NEJ) flukes [1] [11]. These juveniles execute a precise migratory pathway: they penetrate the intestinal wall, enter the peritoneal cavity, migrate through the liver parenchyma, and ultimately reach the bile ducts, where they mature into adults [1] [24].
This entire journey is facilitated by a suite of parasite-derived molecules, predominantly proteases and antioxidants, which are developmentally regulated and tailored to specific host environments and macromolecules [24] [25]. For instance, proteases such as cathepsin B and FhCL3 are crucial for larval activation and intestinal wall penetration, while FhCL1, FhCL2, and FhCL5 are involved in tissue migration and feeding within the liver and bile ducts [24]. The parasite's digestive and secretory machinery is concentrated in the gastrodermal cells lining the gut. These cells are the principal source of secreted proteins, releasing cathepsin proteases via a novel atypical secretory mechanism that involves the rupture of the apical plasma membrane to void secretory vesicles, including extracellular vesicles (EVs), into the gut lumen [25]. This process is essential for nutrient acquisition, primarily the digestion of host hemoglobin, and for immunomodulation.
Table 2: Key Proteolytic Enzymes in Fasciola hepatica Infectivity and Pathogenesis
| Protease / Enzyme | Primary Function in Infection | Expression Site | Significance |
|---|---|---|---|
| Cathepsin L (e.g., FhCL1, FhCL2, FhCL5) | Extracellular digestion of host proteins (e.g., haemoglobin, tissue components); immunomodulation [24] [25]. | Gastrodermal cells; secreted into gut lumen and host tissues [25]. | Major components of the secretome; critical for nutrient acquisition and liver penetration. |
| Cathepsin B & FhCL3 | Larval activation and penetration of the intestinal wall [24]. | Secreted by early juvenile stages (NEJs) [24]. | Essential for establishing initial infection post-excystment. |
| Asparaginyl Endopeptidase | Protein processing and degradation within the parasite's migratory pathway [24]. | Secreted | Contributes to the degradation of host tissues. |
| Aminopeptidases | Final degradation of host-derived peptides into absorbable amino acids [25]. | Gastrodermal cells [25]. | Complete the digestive process initiated by cathepsins. |
The molecular interplay during infection is complex. The diagram below outlines the key secretory and infective processes driven by the gastrodermal cells, from synthesis to host interaction.
Experimental Models and Methodologies for Analysis
Robust experimental protocols are essential for investigating metacercarial biology and screening potential interventions. The following sections detail key methodologies relevant to this field.
Protocol: In Vitro Egg Hatch Test (EHT) for Anthelminthic Efficacy
The Egg Hatch Test is a critical in vitro tool for diagnosing anthelmintic resistance, particularly to drugs like triclabendazole and albendazole [22] [26].
- Egg Collection and Recovery: F. hepatica eggs are recovered from the bile or feces of naturally infected cattle. Bile samples are washed, and eggs are recovered and quantified. A minimum of 1,000 eggs per experimental vial is recommended [22]. For fecal samples, sequential washing and sedimentation techniques are used to purify eggs from debris [23] [26].
- Drug Exposure: Eggs are exposed to a range of concentrations of the anthelmintic compound under investigation. Commercial drugs are diluted according to manufacturer instructions. Tests typically include negative controls (e.g., DMSO and distilled water) [26].
- Incubation and Assessment: The vials are incubated for 28 days at a stable temperature (e.g., 27°C). During this period, eggs are regularly examined under a stereomicroscope and classified based on their degree of development and morphological integrity [22] [26]. The key metric is the percentage of eggs that successfully hatch compared to controls, which allows for the calculation of inhibitory concentrations (e.g., IC₅₀) and the establishment of discriminatory doses (DD) for resistance monitoring [26].
Protocol: Experimental Life Cycle Maintenance in the Laboratory
Maintaining the entire life cycle of F. gigantica (closely related to F. hepatica) in a laboratory setting has been demonstrated and provides a model for similar work with F. hepatica [20].
- Egg Collection and Miracidia Hatching: Eggs are collected from the gall bladders of infected water buffaloes and incubated in dechlorinated tap water at ~29°C. Eggs develop into embryonated stages and hatch as miracidia after 11-12 days [20].
- Snail Infection: Free-swimming miracidia are exposed to susceptible snails (Lymnaea auricularia rubiginosa). Exposed snails are maintained in clay pots and provided with fresh lettuce. One exposed snail can be crushed daily to observe the internal development of larval stages (sporocysts, rediae, cercariae) [20].
- Metacercaria Production: Cercariae shed from snails (beginning around day 39 post-infection) are allowed to adhere to aquatic vegetation, such as rice plants (Oryza sativa), where they encyst to form metacercariae [20].
- Definitive Host Infection: Laboratory animals (e.g., mice) are experimentally infected by oral administration of a known number of metacercariae. Juvenile and adult flukes can be recovered from the intestines and livers of these animals at various time points post-infection to study development and maturation [20].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents and Materials for Fasciola Research
| Item | Function in Research | Specific Application Example |
|---|---|---|
| Lymnaeid Snails | Acting as the intermediate host to support parasite development from miracidium to cercaria. | Maintenance of the parasite life cycle; study of host-parasite interactions [20]. |
| Albendazole Sulfoxide / Nitroxynil | Anthelminthic compounds used for in vitro efficacy testing and resistance monitoring. | Active ingredients in Egg Hatch Tests (EHT) to determine discriminatory doses and IC₅₀ values [26]. |
| Laser Microdissection (LMD) | Precision isolation of specific parasite tissues (e.g., gastrodermal cells, tegument) for omics studies. | Proteomic analysis of tissue-specific protein expression and secretory pathways [25]. |
| Cathepsin L Substrates/Inhibitors | Tools to study the function and activity of key proteolytic enzymes. | Functional characterization of digestive and immunomodulatory mechanisms in invasion [24] [25]. |
| Dimethyl Sulfoxide (DMSO) | Universal solvent for reconstituting water-insoluble anthelmintic compounds for in vitro assays. | Negative control and drug diluent in Egg Hatch Tests [26]. |
The processes of metacercariae encystment and the execution of infectivity are orchestrated by highly specialized biological mechanisms finely tuned to environmental cues and host interactions. The resilience of the environmental stages, coupled with the molecular sophistication of the infective juveniles, underpins the successful global distribution of Fasciola hepatica. Future research must continue to elucidate the precise molecular signals governing encystment and excystment, and the full role of the secretome, particularly extracellular vesicles, in modulating the host environment. This knowledge, integrated with a refined understanding of egg morphology and development, is fundamental for the strategic development of next-generation interventions, such as vaccines and novel anthelmintics, aimed at breaking the chain of environmental transmission and controlling this pervasive parasite.
Within the complex life cycle of the liver fluke Fasciola hepatica, the egg represents a critical stage for dissemination, environmental persistence, and diagnosis. A precise understanding of its core morphology—encompassing standard dimensions, opercular structures, and the embryonation process—is fundamental to research aimed at controlling fascioliasis, a neglected tropical disease affecting millions globally and causing substantial livestock losses [27] [28]. This technical guide synthesizes current research to provide an in-depth analysis of F. hepatica egg morphology, framing these characteristics within the context of the parasite's life cycle and highlighting their importance for diagnostic accuracy, life cycle disruption, and drug development.
Standard Dimensions and Key Morphological Features
The egg of Fasciola hepatica is broadly ellipsoidal and operculated, characteristics it shares with closely related species, which often complicates differential diagnosis [1] [29].
Table 1: Standard Morphometric Data for Fasciola hepatica Eggs
| Characteristic | Measurement Range | Notes |
|---|---|---|
| Length | 130 - 150 µm [1] | Size can show variability depending on the definitive host species [23] [29]. |
| Width | 60 - 90 µm [1] | |
| Operculum | Present [1] | Shape can differ from that of F. gigantica; described as short and straight/flat in F. hepatica [29]. |
| Abopercular End | Often has a roughened or irregular area [1]. | A recently described appendage has been observed on some eggs, a feature previously thought to distinguish other species [23]. |
| Egg Shell | Yellowish in color [1] [29]. | Contains an umbilicus-like invagination at the posterior end [29]. |
| Content at Deposition | Unembryonated, containing a one-cell stage embryo and vitelline cells [1] [29]. |
A significant morphological update is the recent description of an abopercular appendage on some F. hepatica eggs, a feature previously considered a key diagnostic criterion to distinguish them from the eggs of Fascioloides magna [23]. This finding, confirmed in eggs derived from both adult fluke uteri and host feces, indicates that the presence of an appendage is not a reliable distinguishing feature and necessitates a reevaluation of coprological diagnostic keys [23].
The Embryonation Process
Following release from the definitive host, the unembryonated egg must mature in the environment before it can hatch and infect the intermediate snail host. This process, known as embryonation, is highly dependent on external conditions.
Table 2: Embryonation and Hatching Parameters for F. hepatica Eggs
| Parameter | Details | Influencing Factors |
|---|---|---|
| Development to Maturation | 12 - 16 days at ~26°C [29]. | Temperature: Egg hatching and development are inhibited below 10°C and optimal between 20°C and 30°C [6]. |
| Hatching Event | Miracidia hatch within 4 days after maturation is complete [29]. | Light: Acts as a key trigger for hatching. Light stimulates the miracidium, initiating the hatching mechanism [30] [29]. |
| Miracidium Size (Fixed) | 136 µm x 74 µm (average) [29]. | |
| Miracidium Lifespan | Does not exceed 10 hours [29]. |
The entire developmental sequence, from an unembryonated egg to a hatched miracidium, can be visualized as a controlled process dependent on specific environmental cues.
Detailed Experimental Protocols for Egg Analysis
To ensure reproducible research in morphology, viability, and molecular biology, standardized protocols are essential. Below are detailed methodologies for key experimental procedures.
Coprological Egg Recovery and Examination
This protocol is adapted from established helminthological diagnostic standards and used for isolating eggs from host feces [23].
- Homogenization: Homogenize a 3 g fecal sample using a mortar and pestle.
- Suspension: Add 50 mL of water and mix carefully.
- Filtration: Filter the mixture through two layers of synthetic fiber with a 1 mm mesh diameter.
- Sedimentation: Allow the filtered mixture to settle for 5 minutes until a sediment forms.
- Supernatant Removal: Decant the supernatant.
- Washing: Resuspend the sediment in 50 mL of fresh water, settle for 5 minutes, and decant the supernatant.
- Repetition: Repeat the washing step until the supernatant is clear.
- Microscopy: Transfer the final sediment to a microscope slide, cover with a cover slip (24x24 mm), and examine under a light microscope using 40x and 100x objective lenses [23].
Egg Hatching Assay (EHA)
The EHA is a critical technique for assessing egg viability, useful in vaccine trials, fasciolicide efficacy studies, and resistance monitoring [31].
- Egg Collection: Collect eggs from fecal samples or from the uteri of adult flukes dissected from the bile ducts.
- Incubation: Incubate the eggs in water or a suitable buffer in the dark. Temperature and duration are experiment-dependent but often follow established guidelines (e.g., 20-25°C for 14-20 days) to allow for miracidial development [31] [29].
- Light Stimulation: Expose the incubated eggs to light to trigger hatching.
- Enumeration: Count the number of eggs that have successfully hatched, releasing a miracidium. The viability percentage is calculated as (hatched eggs / total eggs) * 100 [31]. Studies show that eggs of fecal origin can demonstrate higher viability (>90%) than those collected directly from the gallbladder [31].
Molecular Analysis of Single Eggs
Genetic identification allows for precise species confirmation and population studies [23].
- Isolation: Individually collect eggs from a sediment sample using a handmade eyelash manipulator under a microscope.
- Lysis: Place a sufficient number of eggs (e.g., ≥65) into a 1.5 mL microcentrifuge tube. Disrupt the egg shells through freezing (e.g., at -70°C).
- DNA Extraction: Use a commercial DNA extraction kit (e.g., QIAamp DNA Micro kit) to purify genomic DNA.
- Amplification: Amplify target genetic regions, such as the internal transcribed spacer (ITS) of ribosomal DNA, using specific primers (e.g., BD1 and BD2) via polymerase chain reaction (PCR) [23].
- Sequencing and Analysis: Sequence the amplified PCR products and compare the resulting sequences to known databases for species identification.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for F. hepatica Egg Studies
| Reagent/Material | Function/Application | Experimental Context |
|---|---|---|
| Handmade Eyelash Manipulator | Precise manual isolation of individual eggs from fecal sediment or culture. | Single-egg DNA analysis [23]. |
| Commercial DNA Extraction Kit | Purification of high-quality genomic DNA from egg pools or single eggs. | Genetic identification (e.g., ITS sequencing), population genetics [23] [32]. |
| ITS Primers (e.g., BD1/BD2) | Amplification of the Internal Transcribed Spacer region for genetic characterization. | Molecular differentiation of Fasciola species [23]. |
| Fluorogenic Peptide Substrates & Class-Selective Inhibitors | Detection and classification of proteolytic activities in egg lysates. | Functional proteomics to study enzyme activity during embryonation [28]. |
| RNA-Seq (e.g., QuantSeq 3' mRNA-Seq) | Transcriptomic profiling to identify genes expressed during egg development. | Investigation of molecular changes at host body temperature [28]. |
| Liquid Chromatography-Tandem Mass Spectrometry | Identification and quantification of proteins translated during egg development. | Proteomic analysis of egg lysates [28]. |
Discussion and Research Implications
The precise morphology and biology of the F. hepatica egg are not merely academic concerns; they have direct implications for disease control. The recent discovery of an abopercular appendage challenges long-held diagnostic paradigms and underscores the need for molecular confirmation in conjunction with morphological identification [23] [27]. Furthermore, the strong temperature dependence of embryonation provides a critical variable for predictive modeling of disease spread in the context of climate change [6].
From a therapeutic development perspective, the egg stage represents a vulnerable target. Research into the transcriptomic and proteomic changes during embryonation, particularly the shift in peptidase expression from cysteine peptidases to aspartic peptidases and metallopeptidases, reveals potential molecular targets for interventions aimed at disrupting development and preventing environmental transmission [28]. While vaccine development has faced challenges, the evaluation of novel antigens, especially those involved in early host-parasite interactions, remains a promising strategy for reducing parasite establishment and egg output [33]. A comprehensive understanding of the core egg morphology and embryonation process is, therefore, a cornerstone of efforts to mitigate the global impact of fascioliasis.
Within the broader context of Fasciola hepatica life cycle and egg morphology research, the eggshell represents a critical interface between the developing miracidium and its external environment. Its structure is not merely a static container but a dynamic, complex architecture essential for both protecting the embryo from external hazards and facilitating the precisely timed release of the miracidium. This whitepaper provides an in-depth technical analysis of the eggshell's ultrastructure, its composition, and the functional mechanisms governing its protective and hatching roles. Understanding these processes at a high resolution is fundamental for researchers and drug development professionals aiming to identify novel targets for interrupting the parasite's transmission, a crucial strategy in controlling a disease that affects millions globally and causes significant livestock losses [27].
Structural and Chemical Composition of the Eggshell
The eggshell of Fasciola hepatica is a specialized structure whose composition and architecture are optimized for durability and selective permeability.
Ultrastructural Architecture
Early electron microscopy studies revealed that the shell consists of a fine, cross-linked fibrillar network, contradicting previous hypotheses of a lamellated structure [34]. This fibrous matrix provides mechanical resilience. The shell is completed by an operculum, a specialized cap that opens during hatching. Recent research using light microscopy has shown that the opercular shape can be a diagnostic feature for differentiating between Fasciola species [35]. Furthermore, some eggs display a previously unreported appendage on the abopercular pole (the pole opposite the operculum), a morphological trait observed in eggs derived from both adult flukes and host feces [4] [23].
Chemical Makeup and Permeability
Chemically, the shell is composed of a hardened, sclerotin-like protein, a result of the quinone tanning process within the Mehlis' gland [34]. Chromatographic analyses indicate the base protein is a simple fibrous type. The shell is freely permeable to small molecules and ions, allowing for the exchange of gases and nutrients [34]. The main barrier to permeability is not the shell itself, but the underlying vitelline membrane complex, which consists of a layer of peri-vitelline material beneath two closely apposed unit membranes [34]. This complex regulates the internal microenvironment of the developing embryo.
Table 1: Key Structural Components of the Fasciola hepatica Egg
| Component | Description | Function |
|---|---|---|
| Shell | Fine reticulum of sclerotin-tanned protein fibrils [34]. | Mechanical protection; permeable to small molecules [34]. |
| Operculum | Specialized cap with species-specific shape variations [35]. | Provides a pre-formed opening for miracidial escape during hatching. |
| Abopercular Appendage | An appendage on the pole opposite the operculum, not always present [4] [23]. | Unknown; potential diagnostic marker. |
| Vitelline Membrane Complex | Peri-vitelline layer with two underlying unit membranes [34]. | Main regulatory barrier for internal egg environment [34]. |
The Shell's Role in Hatching
The hatching of the Fasciola hepatica egg is a temperature-dependent process that involves a complex interaction between the mature miracidium and the eggshell structures.
Hatching Mechanism
The process is initiated by environmental cues, primarily a rise in temperature and exposure to light [6]. The mature miracidium inside the egg becomes active and secretes enzymes, possibly proteases, that act on the opercular seal. Scanning electron microscopy (SEM) studies of the hatching process have captured the operculum partially opening, allowing the miracidium to escape [35]. The miracidium is covered in ciliated epidermal plates arranged in specific tiers (e.g., 20 plates in four tiers for F. gigantica), which provide motility immediately upon release [35].
Developmental Timing and Viability
The time from egg excretion to miracidial hatching is influenced by temperature. Under optimal laboratory conditions (26±1 °C), miracidia of F. hepatica develop within 13-15 days [35]. Once hatched, the miracidium has a short life span, not exceeding 10 hours for F. hepatica, during which it must locate and infect a suitable snail host [35]. The viability of eggs is notably high; studies in sheep have shown that over 90% of eggs of fecal origin can successfully hatch under suitable conditions [31].
Table 2: Hatching and Developmental Parameters of Fasciola hepatica Eggs
| Parameter | Value / Description | Conditions / Notes |
|---|---|---|
| Miracidial Development Period | 13-15 days [35]. | At 26±1 °C. |
| Hatching Trigger | Increase in temperature and light exposure [6]. | |
| Hatching Observation | Partial opening of the operculum [35]. | Observed via Scanning Electron Microscopy. |
| Miracidial Size | 136 × 74 μm [35]. | For F. hepatica. |
| Miracidial Lifespan | Does not exceed 10 hours [35]. | For F. hepatica. |
| Egg Viability (Fecal Origin) | >90% [31]. | In experimentally infected sheep. |
Experimental Methodologies for Ultrastructural and Functional Analysis
A multi-faceted approach is required to fully dissect the structure and function of the Fasciola eggshell. The following are key protocols used in the field.
Protocol 1: Egg Hatching Assay (EHA) for Viability Studies
The EHA is a standard method to determine the viability of eggs in fasciolicide or vaccine trials [31].
- Egg Collection: Eggs can be collected from the gallbladder of infected animals or from fecal samples via sedimentation techniques. Studies indicate fecal-derived eggs can show higher viability (>90%) than those from the gallbladder [31].
- Incubation: A known number of eggs (e.g., 90-110) are placed in well plates with water or test solutions [36].
- Hatching Conditions: Plates are incubated for a set period (e.g., 16 days) at a suitable temperature (e.g., 28°C) and high humidity (e.g., 80%) to allow for embryonic development [36].
- Stimulation and Counting: After incubation, eggs are exposed to artificial light for 2 hours to stimulate hatching. The number of hatched miracidia is counted, and viability is calculated as a percentage [36].
Protocol 2: Scanning Electron Microscopy (SEM) for Structural Analysis
SEM is used for high-resolution imaging of the eggshell surface and hatching mechanisms [35] [36].
- Fixation: Eggs are washed and immersed in a fixative, such as 4% formalin, for 24 hours [36].
- Dehydration: Samples are dehydrated through a graded series of ethanol (e.g., 50%, 70%, 90%, 100%) to remove all water.
- Critical Point Drying: The eggs are dried to their critical point using extra-dry carbon dioxide (CO₂) to prevent structural collapse [36].
- Mounting and Metallization: Dried eggs are mounted on aluminum stubs using conductive adhesive and coated with a thin film (e.g., 20 mA gold film for 2 minutes) to make them electrically conductive [36].
- Imaging: Samples are observed under a scanning electron microscope (e.g., Hitachi S450) at an appropriate accelerating voltage (e.g., 10–15 kW) [36].
Protocol 3: Molecular Workflow for Egg Detection and Speciation
This workflow enables highly sensitive detection and species differentiation directly from fecal samples [37].
- Sample Sedimentation: Feces are homogenized and subjected to a sedimentation protocol to concentrate the eggs.
- Pellet Formation and Disruption: The sedimented pellet is processed, and a portion is used for DNA isolation. A key step involves vigorous disruption using a benchtop homogenizer and bead-beating to break down the tough eggshell and release genomic DNA [37].
- DNA Isolation and Amplification: DNA is isolated using a commercial kit. Species-specific real-time PCR (e.g., TaqMan assay) is then performed to detect and differentiate F. hepatica from F. gigantica [37].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for Fasciola Egg Studies
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| Ethyl Acetate Extract of Artemisia ludoviciana | Plant-derived anthelmintic; causes damage to eggshell integrity, compromising stability [36]. | Ovicidal assays to evaluate efficacy of novel compounds. |
| Triclabendazole | Reference anthelmintic drug; standard for comparing resistance and efficacy in EHA [27]. | Drug resistance studies and fasciolicide evaluation. |
| Formalin (4%) | Fixative for preserving egg morphology prior to electron microscopy [36]. | Sample preparation for SEM structural analysis. |
| Critical Point Dryer | Instrument for dehydrating biological samples without structural collapse using CO₂ [36]. | Essential preparation step for high-quality SEM imaging. |
| FLOTAC/Mini-FLOTAC System | Concentration technique for parasite eggs from feces; used with digital microscopy [38]. | Standardized fecal egg count and diagnostic quantification. |
| Anti-Fasciola Monoclonal Antibodies | Specific antibodies for immunodiagnostic assays like coprological ELISA (coproELISA) [37]. | Detection of coproantigens for early-stage infection diagnosis. |
| BD1 and BD2 Primers | Primers for amplifying the Internal Transcribed Spacer (ITS) rDNA region [23]. | Genetic identification and differentiation of Fasciola species. |
Implications for Drug and Diagnostic Development
The structural and functional insights into the eggshell present unique opportunities for therapeutic and diagnostic innovation. The shell's permeability to small molecules suggests that targeting critical internal developmental processes with specific compounds is feasible [34]. Furthermore, the vulnerability of the shell to certain chemical extracts, such as those from Artemisia ludoviciana, which cause visible damage and compromise stability, validates the eggshell itself as a potential drug target [36]. Disrupting the opercular opening mechanism or the enzymatic processes of hatching could effectively block transmission. For diagnostics, the discovery of morphological variations, like the abopercular appendage, enhances the toolkit for microscopic identification [4] [23]. Meanwhile, molecular workflows that break open the shell to access internal DNA enable highly sensitive and species-specific detection from fecal samples, surpassing the limitations of traditional microscopy and serology [37]. Advanced AI-powered digital microscopy systems are now being trained to automatically detect and differentiate Fasciola eggs from morphologically similar species, increasing diagnostic throughput and accuracy [38].
From Lab to Diagnosis: Protocols for Life Cycle Maintenance and Egg Analysis
Laboratory Protocols for Maintaining Life Cycle Stages In Vitro
Within the broader research on the life cycle and egg morphology of Fasciola hepatica, the development of reliable in vitro culture systems is a cornerstone for advancing our understanding of parasite biology and host-parasite interactions [4]. These protocols enable researchers to bypass the complexities of in vivo models, providing controlled environments for studying parasite development, screening potential anthelmintic compounds, and investigating fundamental physiological processes. This technical guide synthesizes current methodologies for maintaining key life cycle stages of F. hepatica in vitro, with a focus on standardized protocols, viability assessments, and practical applications for researchers and drug development professionals.
Protocol for Newly Excysted Juveniles (NEJ) Using 3D HepG2 Spheroids
Background and Principle
The migratory juvenile stage of F. hepatica causes significant tissue damage during the acute phase of infection but is notoriously difficult to access and study in in vivo models [39]. Traditional two-dimensional (2D) monolayer culture systems provide limited support for NEJ growth and development. The 3D co-culture system using HepG2-derived spheroids, often termed "mini-livers," creates a microenvironment that more closely mimics the physiology of in vivo liver tissue, promoting parasite survival, growth, and morphological development [39] [40]. This system supports the development of the gut and body musculature and stimulates the tegument to elaborate spines and sensory structures, particularly around the oral and ventral suckers [40].
Materials and Equipment
Research Reagent Solutions:
- HepG2 Cells: Human hepatoma cell line (e.g., 85011430, Sigma-Aldrich).
- Basal Medium: Dulbecco's Modified Eagle Medium (DMEM), high glucose, with GlutaMAX supplement.
- Supplements: Foetal Bovine Serum (FBS, 10% v/v) and Antibiotic-Antimycotic (1% v/v).
- Cell Dissociation Reagent: Trypsin-EDTA (2× concentration).
- Spheroid Formation Plates: Ultra-low attachment (ULA) U-bottom 96-well cell culture microplates (e.g., 174925, Thermo Scientific).
- F. hepatica Metacercariae: Isolated from infected snails (e.g., Italian isolate from Ridgeway Research, UK).
- Excystment Medium: Contains 1.2% (w/v) sodium bicarbonate, 0.9% (w/v) sodium chloride, 0.2% (w/v) sodium tauroglycocholate, 0.07% (v/v) concentrated hydrochloric acid, and 0.006% (w/v) L-cysteine.
Step-by-Step Procedure
Production of HepG2 Spheroids:
- Harvest HepG2 cells cultured in complete DMEM and seed 200 cells/well in a ULA U-bottom 96-well plate containing 200 µL of complete DMEM per well [39].
- Centrifuge the plate at 400 × g for 10 minutes to encourage cell aggregation.
- Maintain the spheroids for 13 days at 37°C in 5% CO₂, replacing the medium 1:1 three times per week with centrifugation of plates at 260 × g during medium changes.
- On day 12, plate spheroids in triplets (3 spheroids/well) and incubate overnight to allow them to merge into a single larger tissue mass [39].
Excystment of F. hepatica Metacercariae:
- Agitate metacercariae in 2% sodium hypochlorite solution for a maximum of 8 minutes to remove the outer cyst walls [39].
- Wash the metacercariae three times with deionized water.
- Incubate in pre-warmed excystment medium for 2 hours at 37°C in 5% CO₂ to trigger excystment [39].
- Wash the newly excysted juveniles (NEJs) three times in filter-sterilized DMEM containing 10% FBS and 1% Antibiotic-Antimycotic.
Co-culture Establishment:
- Transfer five NEJs per well into the ULA plates containing the pre-formed 13-day-old HepG2 spheroid triplets [39].
- Maintain the co-culture for up to 21 days at 37°C in 5% CO₂.
- Perform a 1:1 medium change three times per week, carefully avoiding disturbance of the spheroids and parasites.
The following workflow diagram summarizes the key stages of this 3D co-culture protocol:
Assessment and Outcomes
Viability and Development: Assess parasite motility, growth, and development daily using phase-contrast microscopy. Successful culture is indicated by active movement, increase in parasite size, and development of the gut and musculature [39] [40]. Morphological Analysis: Use advanced microscopy to examine the development of tegumental spines and sensory papillae (S1, S2, S3), particularly around the oral and ventral suckers [40]. Biochemical Analysis: Monitor the release of developmentally regulated cysteine proteases (e.g., FhCL3 and FhCL1/2) in the culture supernatant, which mirrors the pattern observed in in vivo parasites [39] [2].
Protocol for Adult Fluke Maintenance
Background and Principle
Adult F. hepatica flukes reside in the bile ducts of the definitive host and are responsible for the pathology associated with the chronic phase of infection. Maintaining adults in vitro is essential for long-term studies on parasite physiology, reproduction, and anthelmintic drug screening [41]. Recent studies have focused on optimizing transport and culture conditions to maximize worm viability and facilitate experimental consistency.
Materials and Equipment
Research Reagent Solutions:
- Collection Source: Adult flukes collected from the bile ducts of naturally infected cattle at slaughterhouses.
- Culture Media: RPMI 1640 or DMEM, supplemented with antibiotics (e.g., penicillin at 1000 IU/mL and gentamicin at 0.1 mg/mL).
- Transport Containers: Sterile 50 mL Falcon tubes.
- Culture Vessels: 6-well or 24-well cell culture plates.
- Solvent Control: DMSO (for drug studies, use at concentrations < 0.5% v/v).
Step-by-Step Procedure
Collection and Transport:
- Collect adult flukes directly from the bile ducts immediately after slaughter.
- Place flukes in sterile 50 mL Falcon tubes containing 40 mL of pre-warmed transport medium (RPMI 1640 or DMEM, supplemented with antibiotics) [41].
- Maintain the transport temperature at 37±2°C using a thermal box. Avoid temperatures >39°C or environmental temperatures (~18°C), which significantly reduce viability [41].
Washing and Acclimation:
- Upon arrival at the laboratory, wash all flukes twice with Hedon-Fleig medium supplemented with antibiotics.
- Perform a final wash with the culture medium to be used for incubation (RPMI 1640 or DMEM) [41].
- Keep all media at 37°C during the washing process.
Cultivation:
- Transfer flukes to culture plates containing the selected culture medium.
- Provide a minimum of 3 mL of medium per fluke to ensure adequate gas exchange and nutrient availability [41].
- Incubate at 37°C in a microaerophilic atmosphere of 5% CO₂ with 95% humidity [41].
- For drug testing involving DMSO, maintain the solvent concentration below 0.5% v/v to avoid toxicity to the flukes [41].
The workflow for the optimized maintenance of adult flukes is outlined below:
Assessment and Outcomes
Viability Assessment: Evaluate fluke motility using a standardized scoring system [41]:
- 3 (Good motility): Fast movements with good intensity.
- 2 (Reduced motility): Slow movements with low intensity, reduced by ~50%.
- 1 (Very reduced motility): Moves only parts of the body.
- 0 (Immobile and dead): Total paralysis and pale coloration.
Quantitative Comparison of Culture Media for Adult Fluke Maintenance:
Table 1: Comparison of transport and culture media for adult F. hepatica viability.
| Media | Transport Viability at 37°C (Score) | Culture Viability (Score) | Key Characteristics |
|---|---|---|---|
| RPMI 1640 | 3 (Good) [41] | High [41] | Supports sustained motility, suitable for long-term maintenance. |
| DMEM | 3 (Good) [41] | High [41] | Comparable performance to RPMI 1640. |
| Hedon-Fleig | 2 (Reduced) [41] | Lower [41] | Traditional medium, lower efficacy compared to RPMI/DMEM. |
| PBS | 2 (Reduced) [41] | Not Recommended | Significant viability loss, for washing steps only. |
Supporting and Diagnostic Assays
Egg Development and Hatching Test (EDHT)
Principle: The EDHT is used to evaluate the ovicidal activity of anthelmintic compounds and diagnose resistance by monitoring the development and hatching of F. hepatica eggs exposed to drugs [22].
Procedure:
- Isolate eggs from the bile or uteri of adult flukes.
- Wash and quantify the eggs, distributing a minimum of 1,000 eggs per vial.
- Expose eggs to the anthelmintic compound of interest (e.g., diluted commercial formulations of triclabendazole, albendazole, nitroxynil).
- Incubate for 28 days at 27°C.
- Classify eggs under a stereomicroscope based on their degree of development (undeveloped, developed, hatched) [22].
Application: This test has been successfully used to identify isolates with resistance or undetermined susceptibility to triclabendazole and albendazole, providing a crucial tool for resistance management [22].
In Vitro Excystment Assay for Metacercariae Viability
Principle: This assay evaluates the viability and infectivity potential of metacercariae after exposure to disinfectants or environmental stressors, such as acetic acid (vinegar) [42].
Procedure:
- Expose metacercariae to the test solution (e.g., different concentrations of vinegar) for varying durations.
- Wash the metacercariae to remove the test substance.
- Induce excystment in vitro by simulating gastrointestinal conditions (using excystment medium with bile salts and reducing agents).
- Assess the rate of excystment, which serves as a direct indicator of metacercariae viability and infectivity [42].
Application: The assay demonstrated that common household practices like brief soaking in diluted vinegar are inadequate for eliminating F. hepatica metacercariae, informing public health recommendations [42].
Troubleshooting and Technical Considerations
- Parasite Source: The quality of the starting biological material is paramount. Use metacercariae from reputable suppliers or ensure rapid processing of adult flukes from slaughterhouses to maximize initial viability [39] [41].
- Contamination Control: Strict aseptic technique is critical. Supplement all media with appropriate antibiotics (e.g., penicillin and gentamicin) and perform procedures in a laminar flow cabinet [39] [41].
- Physiological Relevance: For NEJ studies, 3D spheroid co-cultures are superior to 2D monolayers as they promote more natural parasitic development and protease secretion profiles [39].
- Solvent Toxicity: In drug testing assays, carefully control the concentration of solvents like DMSO. Concentrations above 0.5% v/v can be toxic to adult flukes over 48-hour exposures [41].
The in vitro protocols detailed in this guide provide robust and reproducible methods for maintaining key life cycle stages of Fasciola hepatica. The 3D co-culture system for NEJs and the optimized conditions for adult fluke maintenance represent significant advancements over historical methods, enabling more accurate study of parasite development and host-parasite interactions in vitro [39] [40] [43]. When integrated with supportive assays like the EDHT and excystment test, these techniques form a comprehensive toolkit that directly supports research on the parasite's life cycle and egg morphology. These protocols are indispensable for accelerating the discovery of novel diagnostic markers and therapeutic interventions against fasciolosis.
Within the broader research on the life cycle and egg morphology of Fasciola hepatica, coprological techniques remain foundational for disease surveillance and control. Fasciolosis, caused by the trematode Fasciola hepatica, represents a significant global burden for livestock production and human health [1] [23]. The parasite's indirect life cycle involves an intermediate snail host and culminates in adult flukes residing in the biliary ducts of definitive mammalian hosts, where they produce eggs that are excreted in feces [1] [7]. The detection of these eggs is a critical endpoint for confirming active infection. Despite advancements in immunodiagnostic and molecular methods, microscopic detection of eggs in feces via sedimentation techniques maintains its status as the diagnostic gold standard in both human and veterinary medicine, largely due to its high specificity and direct proof of patent infection [23] [44] [45]. This whitepaper details the methodology, performance, and application of fecal sedimentation within the context of F. hepatica research and drug development.
The Role of Sedimentation in Fasciola hepatica Research
Integration with Parasite Life Cycle and Egg Morphology
The fecal sedimentation technique is intrinsically linked to a thorough understanding of the F. hepatica life cycle and egg morphology. Adult flukes in the bile ducts produce eggs that are passed into the environment via host feces [1] [46]. These eggs are broadly ellipsoidal, operculated, and measure 130–150 µm long by 60–90 µm wide [1] [44]. They are passed unembryonated and possess a characteristic golden-brown color [47]. Accurate identification requires knowledge of these morphological traits to distinguish F. hepatica eggs from those of other trematodes, such as Fasciolopsis buski or paramphistomes (rumen fluke) [1] [47]. Recent research has further described that eggs of F. hepatica may feature an appendage on the abopercular pole, a finding that challenges previous differential diagnostic criteria and underscores the need for ongoing morphological study [23].
The technique's applicability is timed with the prepatent period of the infection, which is the interval from metacercariae ingestion to the appearance of eggs in the feces. In humans and ruminants, this period typically spans 3 to 4 months [1] [45]. Consequently, sedimentation is a tool for detecting patent, chronic infections, while serological assays may be preferred for diagnosing acute, prepatent phases [1].
Diagnostic Performance and Quantitative Data
The natural sedimentation technique has been rigorously validated against the gold standard of post-mortem adult fluke recovery. The following table summarizes its diagnostic performance across different host species, demonstrating high specificity and variable sensitivity.
Table 1: Diagnostic Performance of the Natural Sedimentation Technique for Chronic Fasciolosis [44]
| Host Species | Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) | Negative Predictive Value (%) |
|---|---|---|---|---|
| Cattle | 93 ± 3.36 | 91 ± 5.58 | 96 ± 2.61 | 86 ± 6.57 |
| Sheep | 79 ± 5.13 | 83 ± 6.72 | 90 ± 4.04 | 66 ± 7.58 |
| Swine | 92 ± 6.06 | 100 ± 0.00 | 100 ± 0.00 | 96 ± 0.03 |
A key advantage of fecal sedimentation is its near-100% specificity when the animal's treatment history is known, as the presence of the characteristic egg is a definitive sign of active infection [47] [44]. The sensitivity, while lower, is sufficient for herd-level monitoring and is influenced by factors such as parasite burden and sample handling. Research has shown that using as little as 1 gram of feces yields satisfactory results without statistically significant differences in egg counts compared to larger sample sizes, simplifying the protocol for field conditions [44].
Experimental Protocols and Methodologies
Standard Natural Sedimentation Protocol
The following is a detailed methodology for the natural sedimentation technique, as applied in field and low-resource laboratory settings [23] [44].
Materials Required:
- Microscope (with 10x to 40x magnification)
- Sedimentation cups or glass beakers (500 ml)
- Sieves or gauze (mesh sizes: 750-800 µm, 150-200 µm, and 38-55 µm recommended)
- Petri dishes
- Mortar and pestle (for homogenization)
- Measuring scale
- Disposable pipettes
- Methylene blue stain (1% w/v, optional, to aid visualization)
Procedure:
- Sample Collection and Preparation: Collect a defined mass of feces (e.g., 1g, 3g, or 10g, depending on protocol). Homogenize the fecal sample thoroughly in a mortar after adding a small amount of tap water to create a smooth suspension [23] [44].
- Filtration and Washing: Transfer the suspension to a stack of sieves, starting with the largest mesh and ending with the smallest. Wash the material with a copious volume of tap water until the water exiting the smallest sieve runs clear. This step removes fine debris and retains the parasite eggs on the small mesh sieve [47].
- Sedimentation: Transfer the sediment from the smallest sieve into a sedimentation cup or glass beaker. Fill the container with 500 ml of tap water and allow it to stand for a set period (e.g., 3-5 minutes) to let the eggs settle by gravity [23] [47].
- Decanting and Washing: Carefully decant the supernatant without disturbing the sediment. Resuspend the sediment in fresh water and repeat the sedimentation process until the supernatant is clear [47] [44].
- Microscopy: Transfer the final sediment to a Petri dish using a pipette. Systematically examine the entire dish under a stereo dissecting microscope at 10x to 40x magnification. The addition of a drop of methylene blue can stain background material, making the golden-brown, operculated F. hepatica eggs more conspicuous [47].
Composite Sampling for Herd-Level Diagnosis
For monitoring infection at the group level in livestock, a composite sampling strategy has been validated to reduce costs and labor. The optimal strategy for cattle involves collecting ten individual 10g samples to form a 100g composite sample. A 10g subsample is then taken from this composite for the sedimentation process. This method has demonstrated a diagnostic sensitivity of 0.69 (95% CI 0.5 to 0.85) for identifying an infected herd [47]. A negative composite test should be followed up with a second test due to this inherent sensitivity limitation.
Diagram 1: Fecal Sedimentation Workflow
The Scientist's Toolkit: Research Reagent Solutions
For researchers conducting fecal sedimentation, a set of essential materials and reagents is required. The following table details key items and their specific functions in the experimental workflow.
Table 2: Essential Research Reagents and Materials for Fecal Sedimentation
| Item | Function/Application | Technical Notes |
|---|---|---|
| Sieves (Nested Set) | Sequential filtration of fecal debris to retain parasite eggs. | Recommended mesh sizes: 750-800 µm (large), 150-200 µm (medium), 38-55 µm (small) [47]. |
| Methylene Blue Stain (1% w/v) | Selective staining of background organic material to improve egg contrast. | Aids in the visualization of golden-brown, operculated F. hepatica eggs under microscopy [47]. |
| Stereo Dissecting Microscope | Visualization and morphological identification of F. hepatica eggs. | Sufficient magnification range is 10x to 40x for initial detection and detailed examination [47] [44]. |
| Sedimentation Beakers (500ml) | Gravity-based sedimentation of eggs from filtered fecal suspension. | Standard glass beakers or conical cups allow for efficient decanting after settling [47] [44]. |
| Calibrated Scale | Accurate measurement of fecal sample mass for standardized protocols and quantitative EPG. | Ensures consistency, especially for composite sampling (e.g., 10g per individual sample) [47]. |
Applications in Drug Development and Resistance Monitoring
Fecal sedimentation is a cornerstone in the development and efficacy testing of novel fasciolicides. The Faecal Egg Count Reduction Test (FECRT), which relies on precise egg counting before and after treatment, is a primary method for evaluating drug efficacy and detecting anthelmintic resistance [45]. The high specificity of the egg count ensures that a reduction is truly indicative of parasite death.
Recent research highlights the emergence of resistance to frontline treatments like triclabendazole (TCBZ) [45]. In such studies, sedimentation and FECRT are used in conjunction with other tools like coproantigen ELISA (cELISA) and qPCR to provide a multi-modal assessment of resistance. For instance, a 2025 field investigation in Australia confirmed TCBZ resistance on one sheep property with an efficacy of 86–89% calculated via FECRT [45]. The sedimentation technique, with its ability to provide a direct, quantitative measure of patent infection, remains an indispensable part of this diagnostic arsenal for validating new therapeutic compounds and monitoring field efficacy.
Fecal sedimentation maintains its position as the diagnostic gold standard for patent Fasciola hepatica infection due to its unparalleled specificity, direct correlation with active infection, and practical utility in field and laboratory settings. Its methodology is fundamentally rooted in the parasite's biology, from the biliary production of morphologically distinct eggs to their release into the environment. For researchers and drug development professionals, mastery of this technique—including its standard and composite applications—is crucial for accurate disease surveillance, validation of diagnostic alternatives, and robust assessment of anthelmintic efficacy in the ongoing battle against fasciolosis.
Within the broader study of Fasciola hepatica life cycle and egg morphology, advanced imaging techniques serve as critical tools for research and diagnostics. The liver fluke Fasciola hepatica is a food-borne trematode causing significant zoonotic disease and economic losses in livestock production globally [1] [27]. A comprehensive understanding of its egg morphology is fundamental to multiple research domains, including taxonomy, drug development, and resistance monitoring. While traditional light microscopy remains the diagnostic gold standard in clinical and veterinary settings, it faces limitations in resolution and morphological detail [27]. The integration of Scanning Electron Microscopy (SEM) provides unprecedented ultrastructural detail, enabling researchers to identify novel morphological features, assess anthelmintic efficacy, and make accurate species differentiations [4] [36]. This technical guide outlines standardized protocols and applications of these imaging modalities to support research and drug development professionals in their investigation of Fasciola hepatica.
Standardized Egg Recovery and Preparation Protocols
Egg Recovery and Purification from Bile
The initial recovery and purification of eggs from host bile is a prerequisite for consistent imaging results. The following sedimentation protocol, adapted for high recovery rates, is recommended [36] [22].
- Bile Collection: Aseptically obtain bile from the gallbladders of infected sheep or cattle at slaughterhouses. Transport livers or bile samples maintained at 4-8°C [36].
- Sedimentation Process: Mix bile with 500 ml of distilled water and allow to stand for 20-30 minutes to facilitate egg settling. Decant two-thirds of the supernatant volume and replenish with fresh distilled water. Repeat this washing process until the supernatant is clear [36].
- Egg Storage: Recover the sediment containing eggs and store at 4°C for 24 hours prior to analysis to stabilize the samples [36].
Light Microscopy Preparation
For routine light microscopic examination, prepare unstained wet mounts of the purified eggs [1].
- Slide Preparation: Place a suspension of purified eggs on a standard microscope slide and apply a coverslip. No staining is typically required [1].
- Imaging Parameters: Examine using standard compound light microscopy at 400x magnification. Focus on the operculum and abopercular region for diagnostic characteristics [1].
SEM Sample Preparation
For ultrastructural analysis, proper fixation and processing are essential to preserve egg morphology [36].
- Primary Fixation: Transfer eggs onto Millipore filters and wash 2-3 times with distilled water. Immerse samples in 4% formalin for 24 hours [36].
- Dehydration: Subject fixed eggs to an ascending ethanol series (e.g., 50%, 70%, 80%, 90%, 100%) to gradually remove all water from the specimens [36].
- Critical Point Drying: Use extra-dry carbon dioxide (CO₂) in a critical point dryer to eliminate residual ethanol without causing structural collapse from surface tension [36].
- Metallization: Mount dried eggs on aluminum stubs with carbon adhesive strips. Apply a 20-mA gold film for 2 minutes using a sputter coater to create a conductive surface [36].
- Imaging: Observe samples under a scanning electron microscope (e.g., Hitachi S450) operating at 10-15 kW acceleration voltage [36].
Quantitative Morphological Analysis
Standard Egg Morphometrics
Computer Image Analysis Systems (CIAS) have standardized the morphometric characterization of Fasciola hepatica eggs, providing researchers with quantitative parameters for phenotypic studies [48]. The table below summarizes key dimensional data from established studies.
Table 1: Standard Morphometric Parameters of Fasciola hepatica Eggs
| Parameter | Measurement Range | Imaging Method | Significance |
|---|---|---|---|
| Length | 130-150 µm [1] | Light Microscopy | Baseline diagnostic characteristic |
| Width | 60-90 µm [1] | Light Microscopy | Baseline diagnostic characteristic |
| Abopercular Appendage | Present on anti-opercular pole [4] | SEM | Novel diagnostic feature; not valid for species differentiation |
| Shell Surface | Irregular/roughened at abopercular end [1] | SEM/Light Microscopy | Distinction from Fasciolopsis buski |
Experimental Data on Anthelmintic Effects
SEM imaging provides quantitative data on structural alterations induced by experimental anthelmintic compounds. The following table summarizes efficacy data for an ethyl acetate extract of Artemisia ludoviciana (EAEAL) against Fasciola hepatica eggs [36].
Table 2: Ovicidal Efficacy of Artemisia ludoviciana Extract (EAEAL) on Fasciola hepatica Eggs
| EAEAL Concentration (mg/L) | Ovicidal Effectiveness (%) | Observed Ultrastructural Damage (SEM) |
|---|---|---|
| 100 | 48% | Moderate shell compromise |
| 200 | 52% | Moderate shell compromise |
| 300 | 87% | Severe structural damage |
| 400 | 89% | Severe structural damage |
| 500 | 92% | Severe structural damage, stability critically compromised |
Research Applications and Workflows
Diagnostic Differentiation
Advanced imaging addresses significant challenges in differentiating Fasciola hepatica eggs from other trematode species, particularly given morphological overlaps that complicate parasitological diagnosis [1] [27].
Diagram 1: Diagnostic differentiation workflow for F. hepatica eggs using microscopy.
Anthelmintic Efficacy Assessment
The Egg Development and Hatching Test (EDHT) represents a sophisticated application of light microscopy for evaluating anthelmintic resistance and drug efficacy [22].
EDHT Protocol:
- Egg Isolation: Recover eggs from naturally infected cattle bile, wash, and quantify. Distribute a minimum of 1,000 eggs per vial [22].
- Drug Exposure: Dilute commercial anthelmintics (e.g., albendazole sulfoxide, closantel, nitroxynil, triclabendazole-fenbendazole) according to manufacturer specifications. Test each drug in triplicate alongside untreated controls [22].
- Incubation: Maintain vials at 27°C for 28 days to allow for embryonic development [22].
- Analysis: Classify eggs according to development stage under a stereomicroscope. Compare treatment groups to controls to determine ovicidal activity [22].
The Scientist's Toolkit
Table 3: Essential Research Reagents and Materials for Fasciola Hepatica Egg Imaging
| Reagent/Material | Application | Function |
|---|---|---|
| Ethyl Acetate | Plant extract preparation [36] | Solvent for extracting bioactive compounds with anthelmintic properties |
| Phosphate-Buffered Saline (PBS) | Sample washing [36] | Maintains physiological pH and osmolarity during processing |
| Formalin (4%) | SEM sample fixation [36] | Preserves structural integrity by cross-linking proteins |
| Ethanol Series | SEM sample dehydration [36] | Gradually replaces water to prevent morphological distortion |
| Critical Point Dryer | SEM sample preparation [36] | Removes solvent without surface tension damage |
| Gold Conductive Coating | SEM sample preparation [36] | Creates conductive surface to prevent charging under electron beam |
| RPMI-1640 Medium | Adult fluke culture [36] | Provides nutrient medium for maintaining flukes in vitro |
| Carmine Stain | Whole-mount staining [49] | Highlights reproductive structures in adult flukes |
The integration of light and scanning electron microscopy provides a powerful multidimensional approach to Fasciola hepatica egg examination that serves critical roles in both basic research and applied drug development. Light microscopy offers accessible, quantitative analysis for diagnostic and efficacy studies, while SEM reveals ultrastructural details that inform morphological assessments and mechanism-of-action investigations. The standardized protocols and quantitative frameworks presented in this technical guide provide researchers with validated methodologies to advance understanding of Fasciola hepatica biology, monitor the spread of anthelmintic resistance, and develop novel control strategies against this significant parasitic pathogen.
Within the broader study of the life cycle and egg morphology of Fasciola hepatica, the confirmation of species identity is a foundational step in both basic research and applied drug development. Traditional coprological techniques, which rely on the microscopic examination of egg morphology, face significant challenges due to the substantial overlap in size and shape between eggs of different platyhelminth species, such as Fasciola hepatica, Fasciola gigantica, and Fasciolopsis buski [1]. This diagnostic ambiguity can obstruct accurate prevalence studies, the assessment of drug efficacy, and fundamental life cycle research. The application of DNA analysis from individual eggs has emerged as a powerful tool to overcome these limitations, providing unambiguous species confirmation and enabling a deeper understanding of parasite populations. This technical guide details the methodologies and applications of this approach within the context of Fasciola hepatica research.
The Need for Molecular Confirmation in Fasciola Research
The gold standard for life-time diagnosis of fasciolosis in both humans and animals remains fecal examination to detect eggs [23]. However, the morphological overlap between Fasciola spp. eggs and those of other trematodes presents a persistent diagnostic challenge. Eggs of F. hepatica are broadly ellipsoidal, operculated, and measure 130–150 µm long by 60–90 µm wide [1]. Critically, egg size cannot reliably distinguish F. hepatica from F. gigantica, and there is significant morphologic overlap with eggs of Fasciolopsis buski and some Echinostoma species [1] [23]. This often leads to reports of "Fasciola/Fasciolopsis" eggs in diagnostic settings.
Furthermore, unusual morphological traits, such as an appendage on the anti-opercular pole of the egg—a feature previously thought to be characteristic of Fascioloides magna—have been recently documented in F. hepatica eggs derived from both adult flukes and host feces [23]. This discovery further complicates differential diagnosis based solely on morphology and underscores the necessity for a confirmatory technique that is independent of phenotypic characteristics.
Beyond Species Identification: The Broader Utility of DNA Analysis
The value of DNA analysis extends beyond simple species confirmation.
- Life Cycle Studies: It allows for the precise tracking of parasite strains throughout the different stages of its complex life cycle, from the egg and miracidium to the adult fluke [50].
- Population Genetics: By genotyping individual eggs, researchers can assess the genetic diversity and population structure of F. hepatica within a host or geographic region, investigating phenomena such as the potential selective advantage of heterozygous flukes during tissue migration [50].
- Drug Development: For pharmaceutical researchers, confirming the species and genotype is crucial for evaluating drug efficacy against specific parasite populations and for monitoring the emergence of anthelmintic resistance [50].
Methodological Workflow: From Egg to Sequence
The process of DNA-based species confirmation from a single Fasciola egg involves a series of meticulous laboratory procedures. The following workflow and diagram outline the key stages.
Diagram of the DNA analysis workflow for individual Fasciola eggs.
Sample Preparation and Individual Egg Isolation
The first step involves isolating individual Fasciola eggs from host feces.
- Fecal Sedimentation: Fresh or preserved fecal samples are processed using a sedimentation technique to concentrate parasite eggs. Briefly, feces are homogenized in water, filtered through sieves (e.g., 95 µm and 50 µm), and repeatedly washed and settled to obtain a clean sediment [51] [23].
- Egg Isolation: The resulting sediment is examined under a stereomicroscope. Individual eggs are isolated from the debris using a finely drawn glass pipette or an eyelash manipulator—a traditional tool adept at handling microscopic specimens [23]. The eggs can be transferred individually into low-volume PCR tubes for direct processing or placed in watch glasses to allow miracidia to develop, which can subsequently be used for DNA extraction [23].
DNA Extraction from Individual Eggs
Extracting sufficient quality and quantity of DNA from a single egg is a critical and delicate step. The robust eggshell must be disrupted to release the genetic material within.
- Freeze-Thaw Lysis: A common and effective method involves subjecting the eggs in a small volume of water or buffer to a freeze-thaw cycle (e.g., at -70°C) to rupture the shell and cellular membranes [23].
- Proteinase K Digestion: Following physical disruption, the sample is digested with a buffer containing proteinase K and detergents (e.g., SDS) to degrade proteins and release DNA. This digestion typically occurs overnight at 55°C on a rotating oven to ensure complete lysis [52].
- DNA Purification: The lysate is then purified using commercial silica-membrane-based kits (e.g., QIAamp DNA Micro Kit) to remove inhibitors and isolate pure DNA, which is eluted in a small volume of Tris buffer [52] [23].
PCR Amplification and Sequencing
Given the minimal starting material, the extracted DNA is amplified using the polymerase chain reaction (PCR) targeting specific genetic markers.
- Genetic Targets: For Fasciola species confirmation, the internal transcribed spacer (ITS) region of the ribosomal DNA cluster (including ITS1, 5.8S, and ITS2) is a highly useful marker due to its degree of variation between species [23]. Other mitochondrial genes, such as cytochrome c oxidase I (COI) and NADH dehydrogenase 1 (nad1), are also frequently used for phylogenetic studies and population genetics [53] [50].
- Amplification: The PCR reaction uses specific primers (e.g., BD1 and BD2 for ITS) to target these regions. The success of amplification can be verified by gel electrophoresis.
- Sequencing: The amplified PCR product is then sequenced using the Sanger method. The resulting sequence chromatograms are analyzed and assembled into a contiguous sequence for comparison against reference databases.
Data Analysis and Species Assignment
The final step involves comparing the obtained DNA sequence to a curated database of known sequences to assign a species identity.
- Sequence Alignment: The query sequence is aligned against reference sequences from validated specimens using bioinformatics software (e.g., BLAST).
- Phylogenetic Confirmation: For definitive confirmation, the sequence can be included in a phylogenetic tree analysis. Clustering with reference sequences of F. hepatica with high statistical support (e.g., Bayesian posterior probability ≥0.94) provides robust species confirmation [53]. The lack of comprehensive, validated reference databases for all helminths remains a challenge, underscoring the need for continued curation of public genetic databases [52].
Research Reagent Solutions Toolkit
The following table details key reagents and materials essential for performing DNA analysis from individual Fasciola eggs.
| Research Reagent/Material | Function in the Experimental Protocol |
|---|---|
| Sedimentation Sieves (50µm, 95µm) | To concentrate Fasciola eggs from fecal samples by filtering debris and retaining eggs [51]. |
| Eyelash Manipulator | A handmade tool for the precise microscopic manipulation and isolation of individual eggs from sediment [23]. |
| Proteinase K | A broad-spectrum serine protease used to digest the eggshell and cellular components, liberating DNA for extraction [52]. |
| Lysis Buffer (Tris, EDTA, SDS, DTT) | A chemical buffer to create optimal conditions for proteinase K activity, destabilize membranes, and protect DNA from degradation [52]. |
| Silica-Membrane Spin Columns | For binding and purifying DNA from the complex lysate, removing PCR inhibitors, and eluting clean DNA (e.g., QIAamp kits) [23]. |
| PCR Primers (e.g., ITS, COI) | Sequence-specific oligonucleotides that target and amplify defined mitochondrial or nuclear DNA regions for sequencing [23] [53]. |
| DNA Polymerase | A thermostable enzyme that synthesizes new DNA strands from the primers during the PCR amplification process. |
Comparative Analysis of Diagnostic Methods
The superiority of DNA-based methods over traditional techniques is demonstrated by their higher sensitivity and specificity. The following table summarizes a direct comparison from a recent study on bovine fasciolosis.
Table: Comparison of diagnostic methods for detecting Fasciola spp. in cattle from a study in South Africa (n=277). Adapted from [51].
| Diagnostic Method | Principle of Detection | Positivity Rate (%) | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Sedimentation Test | Microscopic identification of eggs in feces | 13.0 | Low cost, simple, detects patent infections | Low sensitivity, cannot differentiate species, misses pre-patent infections |
| CoproELISA | Detection of copro-antigens in feces | 0.0 | Can detect pre-patent infections | Lower sensitivity with low egg counts, cannot differentiate species |
| qPCR | Detection of Fasciola DNA in feces | 26.4 | High sensitivity/specificity, species confirmation possible, detects pre-patent infections | Higher cost, requires specialized equipment and expertise |
The data clearly shows that qPCR detected more than double the number of positive infections compared to the traditional sedimentation method, highlighting the limitations of morphology-based diagnosis and the power of molecular tools [51].
DNA analysis from individual eggs represents a paradigm shift in the study of Fasciola hepatica, moving beyond the constraints of traditional morphology. This technical guide has outlined a robust workflow, from egg isolation to sequence confirmation, that provides researchers and drug development professionals with a definitive method for species identification. The application of this methodology is essential for accurate life cycle elucidation, population genetic studies, and the development of targeted control strategies. As genetic databases continue to expand and sequencing technologies become more accessible, the integration of DNA-based confirmation will become an indispensable component of high-quality Fasciola research.
Within the life cycle of Fasciola hepatica, the egg stage represents a critical target for disease control and anthelmintic drug development. The adult fluke, residing in the bile ducts of its definitive host, produces eggs that are passed into the environment through feces [1]. These eggs undergo embryonic development under favorable conditions, ultimately hatching to release miracidia that must infect an intermediate snail host to continue the life cycle [20]. Ovicidal assays, which assess a compound's ability to inhibit egg development or hatching, provide a valuable tool for screening potential anthelmintic compounds and detecting drug resistance [22]. Within the broader context of Fasciola hepatica research, understanding egg morphology and development is fundamental, as coprological examination of eggs remains the gold standard for diagnosing active infections in humans and animals [23]. This technical guide details the methodologies and applications of ovicidal assays for drug screening, providing researchers with standardized protocols to evaluate compound efficacy against this resilient parasite stage.
Fasciola hepatica Egg Biology and Morphology
Key Morphological Features for Identification
The eggs of Fasciola hepatica are broadly ellipsoidal and operculated, typically measuring 130–150 µm in length by 60–90 µm in width [1]. They are passed in the feces in an unembryonated state and possess a distinctive yellowish-brown coloration [1] [54]. A key diagnostic feature is the often roughened or irregular area at the abopercular end [1]. Recent research has documented that some Fasciola hepatica eggs may exhibit an appendage on the abopercular pole, a characteristic previously thought to primarily distinguish Fascioloides species [23]. This finding has important implications for differential diagnosis during coprological examinations.
Developmental Timeline and Environmental Influences
The development and hatching of Fasciola hepatica eggs are highly dependent on environmental conditions, particularly temperature. Under optimal laboratory temperatures of approximately 26 ± 1 °C, miracidia develop within a period of 13-15 days [35]. The entire life cycle, from egg to mature adult, takes approximately 3-4 months in the definitive host [1]. A recent comprehensive review has established that egg hatching and development are inhibited below 10 °C, with optimal development occurring between 20 °C and 30 °C [6]. These temperature thresholds are crucial for designing in vitro ovicidal assays, as they ensure normal embryonic development in control groups while testing compound effects on this biological process.
Ovicidal Assay Methodologies
Egg Isolation and Preparation
The initial step in any ovicidal assay involves the isolation of viable, undeveloped eggs from host tissues. The following standardized protocol ensures high-quality egg recovery:
- Source Material: Collect gallbladders or bile from naturally infected cattle or sheep at slaughterhouses [22] [55]. Alternatively, adult flukes can be recovered from infected livers, with eggs obtained directly from the flukes' uteri [23].
- Washing and Sedimentation: Homogenize bile or fecal samples with tap water or distilled water and filter through a series of sieves with decreasing mesh sizes (e.g., 250 µm, 100 µm, and 50 µm) [56]. Transfer the filtrate to glass beakers and allow eggs to sediment for 5 minutes; discard supernatant and repeat washing until the suspension is clear [56].
- Egg Quantification: Resuspend the purified egg sediment in a known volume of water and quantify using a stereomicroscope. Adjust concentration as needed for specific assays (typically 90-110 eggs per well for 24-well plates) [55]. Store eggs at 4°C for no more than 24 hours before assay initiation [55].
Egg Development and Hatching Test (EDHT)
The EDHT evaluates a compound's ovicidal activity by assessing its ability to disrupt normal embryonic development. The following protocol is adapted from recently published methodologies [22]:
- Experimental Setup: Distribute a minimum of 1,000 eggs per vial in universal collectors [22]. Alternatively, use 24-well cell culture plates with 90-110 eggs per well [55].
- Compound Exposure: Dilute test compounds according to manufacturer instructions or prepare serial dilutions in appropriate solvents. Include negative controls (e.g., water or solvent alone) and positive controls (e.g., triclabendazole or albendazole) in triplicate [22] [55].
- Incubation Conditions: Incubate eggs at 27°C for 28 days [22] or at 25°C for 16 days [55] in the dark to simulate normal development conditions.
- Assessment and Analysis: After incubation, classify eggs according to their developmental stage under a stereomicroscope. Count the number of embryonated eggs (containing developed miracidia) and non-embrionated eggs in each treatment group [57]. Calculate ovicidal activity using the formula:
Ovicidal Activity (%) = [(% hatched in control - % hatched in treatment) / % hatched in control] × 100 [56]
Table 1: Key Parameters for Egg Development and Hatching Test
| Parameter | Specification | Reference |
|---|---|---|
| Egg Quantity | Minimum 1,000 eggs per vial or 90-110 per well | [22] [55] |
| Incubation Temperature | 25-27°C | [22] [55] |
| Incubation Period | 16-28 days | [22] [55] |
| Assessment Criteria | Embryonation status, miracidial development | [57] |
| Replication | Minimum triplicate samples | [22] |
Egg Hatch Test (EHT) for Benzimidazole Resistance
The EHT is specifically valuable for detecting resistance to benzimidazole compounds, such as albendazole. This test can be performed in two primary formats, differing in the duration of drug exposure [56] [22]:
- Short-Exposure EHT (12H): Incubate eggs (approximately 200/mL) with anthelmintic at various concentrations (e.g., 0.05, 0.5, and 5.0 µM for albendazole) for 12 hours at 25°C in the dark [56]. Then wash eggs to remove the anthelmintic and incubate in water for 15 days before assessment.
- Continuous-Exposure EHT (15D): Incubate eggs with anthelmintic for the entire 15-day development period without removing the drug [56].
- Hatching Induction: After the development period, expose plates to light for 2 hours to stimulate hatching [56]. Add formalin (10 µL of 10% buffered formalin per well) to fix the contents before counting.
- Interpretation: Evaluate 80-100 eggs per well under optical microscopy (40× magnification). Consider "hatched eggs" as both empty shells and embryonated eggs containing developed miracidia [56]. Reduced ovicidal activity at specific drug concentrations suggests emerging resistance.
The following workflow diagram illustrates the key decision points in selecting and performing appropriate ovicidal assays:
Quantitative Data Analysis and Interpretation
Efficacy Criteria and Resistance Thresholds
Establishing clear efficacy thresholds is essential for interpreting ovicidal assay results. For the Egg Hatch Test, ovicidal activity below 90% at specific drug concentrations suggests reduced efficacy or emerging resistance [56] [22]. In faecal egg count reduction tests (FECRT), a percentage reduction of eggs below 90% 14 days after treatment also indicates likely anthelmintic resistance [56]. Recent research applying the EDHT to commercial drugs identified resistant isolates based on complete failure to inhibit development at recommended concentrations [22].
Table 2: Efficacy Thresholds for Ovicidal Assays and Related Tests
| Test Method | Efficacy Threshold | Resistance Indicator | Reference |
|---|---|---|---|
| Egg Hatch Test (EHT) | >90% ovicidal activity | <90% ovicidal activity at specific concentrations | [56] |
| Faecal Egg Count Reduction Test (FECRT) | >90% reduction | 77-81.8% reduction (albendazole resistance) | [56] |
| Egg Development and Hatching Test (EDHT) | Complete development inhibition | Continued development at therapeutic concentrations | [22] |
Recent Experimental Findings
Contemporary studies have yielded important quantitative data on compound efficacy against Fasciola hepatica eggs:
- Commercial Drugs: In EDHT evaluations of commercial drugs, samples have shown resistance to triclabendazole-fenbendazole combinations, while closantel demonstrated no ovicidal activity and was unsuitable for EDHT [22].
- Natural Compounds: Ethyl acetate extract from Artemisia ludoviciana (EALM) showed significant ovicidal activity at concentrations above 300 mg/L, with ovicidal percentages exceeding 60% after 16 days of incubation [55]. Cumin essential oil demonstrated 100% efficacy at all concentrations tested (0.031125–4.15 mg/mL) [57].
- Temperature Effects: Research confirms that egg hatching is significantly influenced by temperature, with complete inhibition below 10°C and optimal development between 20-30°C [6]. At approximately 27°C, cercarial shedding from snail hosts accelerates, highlighting the importance of temperature control in assays [6].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of ovicidal assays requires specific laboratory reagents and materials. The following table details essential components for establishing these assays in a research setting:
Table 3: Essential Research Reagents and Materials for Ovicidal Assays
| Reagent/Material | Function/Application | Specifications/Alternatives |
|---|---|---|
| Fasciola hepatica eggs | Primary assay component | Isolated from naturally infected cattle/sheep bile or feces [22] [55] |
| Albendazole | Positive control for benzimidazole class | Typically tested at 0.05, 0.5, and 5.0 µM concentrations [56] |
| Triclabendazole | Positive control for preferred anthelmintic | Tested at 10 and 50 mg/L concentrations [55] |
| Artemisia ludoviciana extract | Natural product comparator | Ethyl acetate extract tested at 100-500 mg/L [55] |
| Cumin essential oil | Natural product comparator | Tested at 0.031125–4.15 mg/mL concentrations [57] |
| Roswell Park Memorial Institute (RPMI) 1640 Medium | Maintenance medium for adult flukes | Used with 50% bovine serum and antibiotics [55] |
| Formalin (10% buffered) | Fixation after development period | Preserves eggs for counting [56] |
| Cell culture plates (24-well) | Platform for egg incubation | Suitable for 90-110 eggs per well [55] |
| Sedimentation sieves | Egg isolation and purification | Mesh sizes: 250 µm, 100 µm, and 50 µm [56] |
Troubleshooting and Technical Considerations
Common Challenges and Solutions
- Low Egg Viability: Ensure rapid processing of source material and maintain eggs at 4°C for no more than 24 hours before assay initiation [55]. Use eggs from multiple hosts to account for natural variation in viability.
- Inconsistent Development: Maintain stable incubation temperatures within the optimal 20-30°C range [6]. Avoid temperature fluctuations greater than ±1°C during the development period.
- Solvent Toxicity: When testing hydrophobic compounds, include solvent-only controls (e.g., methanol, Tween80) to account for potential solvent effects on egg development [57] [55].
- Bacterial Contamination: Add antibiotic mixtures (e.g., 100 IU penicillin + 100 mg/ml streptomycin) to the incubation medium to prevent microbial overgrowth [55].
Validation and Standardization
To ensure reproducible results across laboratories, implement the following validation procedures:
- Include Reference Isolates: Whenever possible, include known drug-susceptible and drug-resistant isolates as controls [22] [56].
- Standardize Assessment Criteria: Establish clear, objective criteria for classifying embryonic development stages [22]. Use blinded assessment when counting eggs to minimize bias.
- Replicate Experiments: Perform all tests in triplicate with eggs from at least three different biological sources to account for natural variation [22].
Ovicidal assays represent an essential component in the toolbox of Fasciola hepatica researchers and drug development professionals. The Egg Development and Hatching Test and Egg Hatch Test provide valuable methods for screening novel compounds and detecting emerging anthelmintic resistance. When properly standardized and executed, these assays generate reproducible, quantitative data on compound efficacy against this critical life cycle stage. As drug resistance continues to emerge globally [22] [56], these ovicidal assays will play an increasingly important role in developing the next generation of fasciolicides and preserving the efficacy of existing treatments. Through continued refinement and international standardization of these methodologies, the research community can more effectively combat this significant parasitic disease of livestock and humans.
Fasciolosis, caused by the trematode Fasciola hepatica, represents a significant global burden for both human and veterinary medicine. Within the broader context of research on the life cycle and egg morphology of Fasciola hepatica, diagnostic strategies must be strategically aligned with the distinct biological events of the infection timeline. The parasite's complex migration through host tissues, eventual maturation in the bile ducts, and the subsequent commencement of egg-laying define two critical diagnostic windows: the acute, pre-patent phase and the chronic, patent phase. This guide provides an in-depth technical analysis of serological and coprological diagnostic methods, framing their appropriate application based on the infection phase and the specific diagnostic or research question at hand. A precise understanding of the parasite's development, from metacercariae ingestion to adult fluke egg production, is fundamental to selecting the correct diagnostic tool.
The following diagram illustrates the critical relationship between the Fasciola hepatica life cycle stages within the human host and the corresponding diagnostic windows for serological and coprological methods.
The Parasite Life Cycle: Foundation for Diagnostic Strategy
The definitive host, including ruminants and humans, becomes infected by ingesting metacercariae attached to aquatic vegetation or other substrates [1] [46]. Following ingestion, the metacercariae excyst in the duodenum, releasing juvenile flukes that penetrate the intestinal wall and migrate across the peritoneal cavity to the liver [58]. The immature flukes then tunnel through the liver parenchyma for a period of 6-8 weeks, causing significant tissue damage—this constitutes the acute or invasive phase of the infection [1] [7]. The flukes eventually settle in the bile ducts, where they mature into adults and begin producing eggs. The pre-patent period, from ingestion of metacercariae to the appearance of eggs in the feces, is approximately 10-12 weeks in animals and 3-4 months in humans [1] [7]. The chronic or biliary phase begins once egg production commences and can persist for years [1].
This life cycle directly dictates diagnostic applicability. The acute phase is characterized by the presence of migrating juvenile flukes that do not produce eggs, making direct parasitological methods ineffective. Instead, detection relies on the host's immune response to the parasites' excretory-secretory (ES) products. In contrast, the chronic phase is defined by the presence of egg-laying adults, enabling direct detection of the parasite through its eggs or antigens [59] [1].
Serological Diagnostics: Capturing the Acute Phase
Serological assays detect either specific antibodies (IgG) against Fasciola or circulating parasite antigens in serum. Their primary utility lies in diagnosing the pre-patent infection, as they can become positive as early as 2-4 weeks post-infection [1].
Key Antigens and Test Formats
The performance of serological tests is heavily dependent on the choice of antigen used in the assay.
- Native Excretory-Secretory (ES) Antigens: These are complex mixtures of proteins released by adult and juvenile flukes, rich in proteolytic enzymes like cathepsin L proteases. ES antigens are highly immunogenic and have been a cornerstone of serodiagnosis. A 2023 meta-analysis found that tests using FhES antigens demonstrated a sensitivity of 93.1-98.5% and a specificity of 95.9-99.7% for human fasciolosis [59].
- Recombinant Antigens: Recombinant technology allows for standardized and cost-effective production of specific immunodominant antigens. The most prominent include:
- Recombinant Cathepsin L1 (rCL1): A major component of ES products, used in numerous ELISA and immunoblot formats [59].
- Recombinant Saposin-like protein 2 (rSAP-2): Used in a CDC-developed immunoblot assay, which reports a sensitivity of ≥94% and specificity of ≥98% for chronic human infection [1].
- Other antigens such as recombinant glutathione S-transferase (rGST) and recombinant leucine aminopeptidase (rLAP) have also been investigated with good results [59].
The primary test formats are the Enzyme-Linked Immunosorbent Assay (ELISA) for high-throughput screening and Immunoblot (Western Blot) as a confirmatory test due to its high specificity [1].
Experimental Protocol: Indirect ELISA for Antibody Detection
Purpose: To detect the presence of anti-Fasciola hepatica antibodies in host serum. Principle: Serum antibodies bind to Fasciola antigens coated on a plate; this complex is then detected by an enzyme-conjugated secondary antibody, producing a colorimetric signal proportional to the antibody concentration.
Materials & Reagents:
- Coating Antigen: Recombinant FhSAP2 or FhrCL1 (1-5 µg/mL in carbonate-bicarbonate buffer, pH 9.6) [59] [1].
- Test Samples: Serum from suspect hosts (e.g., cattle, sheep, humans). Positive and negative control sera are essential.
- Blocking Buffer: 1-5% Bovine Serum Albumin (BSA) or non-fat dry milk in PBS-T (Phosphate-Buffered Saline with 0.05% Tween-20).
- Conjugate: Horseradish Peroxidase (HRP)-conjugated anti-species (e.g., anti-bovine, anti-human) IgG antibody.
- Substrate: TMB (3,3',5,5'-Tetramethylbenzidine) or ABTS (2,2'-Azinobis [3-ethylbenzothiazoline-6-sulfonic acid]).
- Stop Solution: 1M Sulfuric Acid or 1M Hydrochloric Acid.
- Equipment: Microplate reader (absorbance at 450 nm for TMB).
Procedure:
- Coating: Coat wells of a microtiter plate with 100 µL of diluted antigen. Incubate overnight at 4°C.
- Washing: Wash the plate three times with PBS-T.
- Blocking: Add 200 µL of blocking buffer to each well. Incubate for 1-2 hours at 37°C. Wash three times.
- Sample Incubation: Add 100 µL of diluted test and control sera to assigned wells. Incubate for 1 hour at 37°C. Wash three times.
- Conjugate Incubation: Add 100 µL of diluted enzyme-conjugated secondary antibody. Incubate for 1 hour at 37°C. Wash three times.
- Detection: Add 100 µL of substrate solution. Incubate in the dark for 10-30 minutes.
- Stop Reaction: Add 50 µL of stop solution.
- Reading & Interpretation: Measure absorbance immediately. Compare sample absorbance to the cutoff value (typically determined as the mean of negative controls + 2 or 3 standard deviations).
Table 1: Performance Metrics of Key Serological Antigens (Meta-Analysis Data)
| Antigen | Target Population | Sensitivity (Range) | Specificity (Range) | Key Applications |
|---|---|---|---|---|
| FhES (Native) | Humans | 93.1% - 98.5% | 95.9% - 99.7% | Gold standard for serology; acute and chronic phase detection [59] |
| FhrCL-1 | Humans, Cattle, Sheep | High (Specific values N/A) | High (Specific values N/A) | Dominant protease; high immunogenicity; used in ELISA & LFI [59] |
| FhrSAP-2 | Humans | ≥94% | ≥98% | CDC immunoblot confirmatory test; chronic infection [1] |
| FhrLAP | Humans, Animals | Good (Specific values N/A) | Good (Specific values N/A) | Immunodiagnostic potential; further validation ongoing [59] |
Coprological Diagnostics: Confirming the Chronic Phase
Coprological methods detect the presence of Fasciola eggs in stool samples and are considered the "gold standard" for confirming a patent, chronic infection [23]. Their major limitation is the inability to diagnose infections during the pre-patent period.
Egg Morphology and Diagnostic Considerations
Fasciola hepatica eggs are broadly ellipsoidal, operculated, and measure 130–150 µm by 60–90 µm [1] [23]. They are passed in the feces unembryonated. A key morphological feature discussed in recent literature is the potential presence of an irregular or roughened area at the abopercular end, and in some cases, even a visible appendage, though this was previously thought to be a characteristic of other flukes like Fascioloides magna [23]. This makes accurate morphological identification crucial. Eggs of F. hepatica can be difficult to distinguish from those of F. gigantica and Fasciolopsis buski [1].
Standard Coprological Techniques and Protocols
A. Sedimentation Techniques (Gold Standard)
Principle: Fasciola eggs are dense and sediment in aqueous solutions, allowing for concentration and microscopic examination. Protocol (Sequential Washing/Sedimentation):
- Homogenize 3-5g of feces in 50 mL of water or saline.
- Carefully filter the mixture through a sieve (e.g., 1 mm mesh) to remove large debris.
- Transfer the filtrate to a conical glass or beaker and allow it to settle for 5 minutes.
- Decant the supernatant carefully without disturbing the sediment.
- Resuspend the sediment in fresh water, and repeat the sedimentation and decanting steps until the supernatant is clear.
- Examine the final sediment under a microscope (10x or 40x objective) for the presence of Fasciola eggs [23] [58].
B. Flukefinder Method
Principle: A proprietary commercial device that uses a combination of sieving and sedimentation in a closed system to efficiently separate and concentrate trematode eggs from large fecal samples (e.g., 10-30g), improving recovery rates and reducing processing time.
C. Quantitative Methods (Eggs Per Gram - EPG)
Kato-Katz Technique:
- A semi-quantitative method widely used in field surveys, especially for humans. A fixed amount of feces (typically 50 mg) is pressed through a mesh to remove large debris, transferred to a template on a slide, covered with a glycerol-soaked cellophane cover slip that clears the sample, and examined. The number of eggs is counted and multiplied by a factor to obtain EPG [59] [58].
Centrifugation-Flotiation Techniques:
- While less common for Fasciola due to the egg's high density, techniques using high-density solutions like zinc sulfate can be used to isolate highly pure eggs for research purposes [58].
Table 2: Comparison of Primary Coprological Diagnostic Methods
| Method | Principle | Sensitivity | Advantages | Limitations |
|---|---|---|---|---|
| Sedimentation | Gravity-based concentration | Moderate to High (depends on sample size) | Inexpensive; simple; considered gold standard [23] | Time-consuming; requires multiple washing steps [58] |
| Flukefinder | Sieving & sedimentation in closed system | High | Efficient for large samples; reduces biohazard risk [60] | Requires specialized equipment |
| Kato-Katz | Filtration & clearing | Moderate (small sample size) | Rapid; quantitative; good for field studies [58] | Low sensitivity in light infections; clearing can distort eggs |
| Centrifugation | Centrifugal force concentration | High | Rapid; good recovery [58] | Requires centrifuge; high speed may damage eggs |
Integrated Diagnostic Strategy and Advanced Methods
A strategic diagnostic approach integrates both serological and coprological methods based on the clinical presentation and the time since potential exposure. The following workflow outlines a logical diagnostic pathway for Fasciola infection.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents for Fasciola Diagnostic Research
| Reagent / Material | Function / Application | Technical Notes |
|---|---|---|
| Recombinant FhSAP2 Antigen | Coating antigen for ELISA; antigen for immunoblot | Recombinant protein produced in E. coli or yeast; basis of CDC's immunoblot [1]. |
| Recombinant Cathepsin L1 (rCL1) | Coating antigen for ELISA and LFI | Major component of ES; high immunodominance; used for antibody detection [59]. |
| Native ES Antigens | Gold standard antigen for serological assays | Complex mixture from adult fluke culture; high sensitivity but batch-to-batch variation [59]. |
| HRP-conjugated Anti-Species IgG | Secondary antibody for ELISA | Species-specific (e.g., anti-bovine, anti-human); enables colorimetric detection [59]. |
| Fasciola hepatica Positive Control Sera | Quality control for serological assays | Confirms test functionality; should be from confirmed infected hosts. |
| Sedimentation Apparatus (Cones/Beakers) | Coprological examination | For standard sedimentation technique; requires microscope for examination [23] [58]. |
| Flukefinder Device | Efficient egg concentration from feces | Proprietary system for processing large fecal samples with high recovery [60]. |
Advanced and Emerging Diagnostic Approaches
- Coproantigen ELISA: This method detects Fasciola-specific antigens secreted in the host's feces. It can indicate current infection (both pre-patent and patent) and is not reliant on egg production. Studies suggest it does not cross-react with rumen fluke (Calicophoron daubneyi) [60].
- Molecular Techniques (PCR): PCR-based methods amplify Fasciola DNA from eggs in feces or from adult worms. They are highly specific and can differentiate between F. hepatica and F. gigantica, making them invaluable for species-specific epidemiological studies and detecting hybrid forms [58].
- Lateral Flow Immunoassays (LFI): These rapid tests, often using recombinant antigens like rCL1, are being developed for point-of-care use in both human and veterinary medicine, offering results within minutes [59].
The strategic use of diagnostic tests for fasciolosis is fundamentally guided by the parasite's biology. Serological methods are the cornerstone for detecting the acute, pre-patent phase of infection, providing a crucial window for early intervention. Coprological methods remain the definitive gold standard for confirming patent, chronic infections through direct observation of eggs. An integrated approach, which may also include emerging coproantigen and molecular tests, provides the most powerful framework for accurate diagnosis, effective treatment monitoring, and advanced research. For scientists engaged in life cycle and egg morphology studies, a deep understanding of the strengths and limitations of each diagnostic modality is essential for designing robust experiments and accurately interpreting findings in the context of both human and veterinary fasciolosis.
Addressing Diagnostic Ambiguity and Therapeutic Resistance in Fasciolosis
Differentiating Fasciola Eggs from Fasciolopsis buski and Other Trematodes
Within the broader research on the life cycle and egg morphology of Fasciola hepatica, the precise differentiation of its eggs from those of other trematodes represents a critical diagnostic challenge. This distinction is paramount for accurate disease surveillance, effective treatment, and advanced drug development. Fascioliasis, caused by Fasciola hepatica and F. gigantica, and fasciolopsiasis, caused by Fasciolopsis buski, are food-borne trematodiases classified as neglected diseases by the World Health Organization [61]. Although both parasites produce morphologically similar eggs, they inhabit distinct organ sites within the host—the bile ducts and the intestine, respectively—and have different life cycles and pathogenic impacts [62] [1]. This in-depth technical guide synthesizes current knowledge and emerging research to provide researchers and scientists with a definitive resource for the identification and differentiation of these parasites, with a particular focus on the evolving understanding of F. hepatica egg morphology.
Comparative Morphology of Trematode Eggs
The eggs of Fasciola spp. and Fasciolopsis buski exhibit significant morphological overlap, necessitating careful observation of subtle distinguishing characteristics.
Key Morphological Features for Differentiation
Table 1: Comparative Morphology of Fasciola spp. and Fasciolopsis buski Eggs
| Feature | Fasciola spp. | Fasciolopsis buski |
|---|---|---|
| Size | 130–150 µm long by 60–90 µm wide [1] | 130–150 µm long by 60–90 µm wide [62] |
| Shape | Broadly ellipsoidal [1] | Broadly ellipsoidal [62] |
| Operculum | Present | Present |
| Abopercular End | Often has a roughened or irregular area [1] [23] | Not typically described as roughened |
| Appendage | An appendage on the abopercular pole has been recently documented [23] | Not reported |
| Embryonation | Unembryonated when passed in feces [1] | Unembryonated when passed in feces [62] |
As indicated in Table 1, the eggs are virtually identical in size and general shape, making them easily confusable [62] [1]. The most traditional differentiating feature has been the roughened or irregular area at the abopercular end (the end opposite the operculum) in Fasciola spp. eggs [1]. However, a recent serendipitous finding has revealed that eggs of F. hepatica may also possess a distinct appendage on the abopercular pole [23]. This appendage was observed in eggs derived from both reindeer feces and adult flukes from a bull, and its presence was confirmed using 3D modeling based on light and electron microscopy references [23]. This characteristic was previously considered a hallmark of Fascioloides magna, and its discovery in F. hepatica indicates it may not be a valid criterion for distinguishing between these genera [23]. Consequently, relying solely on light microscopy for differentiation is often insufficient, and eggs are frequently reported as "Fasciola/Fasciolopsis" eggs due to this morphologic overlap [1].
Visual Workflow for Morphological Differentiation
The following diagram synthesizes the recommended observational pathway for differentiating these eggs based on current morphological knowledge.
Advanced Diagnostic and Molecular Techniques
Given the limitations of morphology, confirmation often requires advanced techniques, especially during the acute phase of infection when eggs are not yet being shed.
Serological and Molecular Detection
Antibody detection is particularly valuable in the acute phase, as specific antibodies become detectable 2-4 weeks post-infection, whereas egg production does not start until 3-4 months after exposure [1]. The Centers for Disease Control and Prevention (CDC) employs an immunoblot assay based on a recombinant F. hepatica antigen (FhSAP2). This assay has a reported sensitivity of ≥94% and a specificity of ≥98% for chronic human infection [1]. Serology can also help rule out false fascioliasis (pseudofascioliasis), where eggs are present in stool due to the recent consumption of contaminated liver rather than an active infection [1].
Molecular techniques provide the highest level of specificity. DNA extraction from individual eggs or miracidia, followed by PCR amplification of genetic markers such as the internal transcribed spacer (ITS) region or the mitochondrial cytochrome c oxidase I (COI) gene, allows for definitive species identification [23] [63]. One detailed protocol involves:
- Egg Processing: Eggs are obtained via sedimentation techniques and often stored in water to allow miracidia to develop [23].
- DNA Extraction: Individual eggs are collected using a handmade manipulator, and DNA is extracted using a commercial kit after a freeze-thaw cycle to destroy the eggshell [23].
- PCR Amplification: Primers specific to the target gene (e.g., COI primers: 5'-TTGATTGGGGGTTTTGGTAA-3' and 5'-CCCAGCCAAATGAAGAGAAA-3') are used [63].
- Sequencing and Analysis: The PCR products are sequenced and analyzed using tools like NCBI-BLAST for confirmation [23] [63].
Gene Expression Analysis
Research into stage-specific gene expression provides insights for diagnostics and drug development. For instance, the expression of FhHDM-1 (Fasciola hepatica Helminth Defense Molecule-1), an immunomodulatory protein, has been studied across life stages. Reverse Transcription PCR (RT-PCR) reveals that FhHDM-1 mRNA is present in adult flukes but absent in eggs and miracidia [63]. This suggests the gene's role is specific to the parasitic interaction with the definitive host and highlights the molecular differences between developmental stages.
The workflow for such a gene expression analysis is detailed below.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Fasciola Research
| Reagent / Material | Function / Application | Example & Notes |
|---|---|---|
| Sedimentation Kit | Concentration of eggs from fecal samples for microscopic or molecular analysis. | Basis of the "gold standard" coprological diagnosis [23]. |
| DNA Extraction Kit | Isolation of genomic DNA from eggs, miracidia, or adult flukes for PCR. | Essential for molecular confirmation and phylogenetic studies [23] [63]. |
| Specific Primers (ITS, COI) | PCR amplification of species-specific genetic markers. | E.g., COI primers: 5'-TTGATTGGGGGTTTTGGTAA-3' / 5'-CCCAGCCAAATGAAGAGAAA-3' [63]. |
| Recombinant Antigens (e.g., FhSAP2) | Target for immunodiagnostic assays like immunoblot or EIA. | Used in CDC's immunoblot (94% sensitivity, 98% specificity for chronic infection) [1]. |
| RNA Extraction Kit & cDNA Synthesis Kit | Analysis of stage-specific gene expression (e.g., FhHDM-1). | Allows study of molecular biology and immune evasion mechanisms [63]. |
The differentiation of Fasciola eggs from those of Fasciolopsis buski and other trematodes remains a cornerstone of accurate diagnosis and research. While traditional microscopy, focusing on subtle features like the roughened abopercular end and the newly described appendage, provides a first line of identification, it has inherent limitations. The definitive differentiation increasingly relies on a multifaceted approach. This includes sophisticated serological assays to detect early infection and the application of molecular tools, such as PCR targeting the ITS or COI genes, for unambiguous species identification. Furthermore, the investigation of stage-specific gene expression profiles, like the absence of FhHDM-1 mRNA in eggs, opens new avenues for understanding parasite biology and developing novel interventions. For researchers and drug development professionals, mastering this integrated methodology—from basic morphology to advanced molecular protocols—is essential for advancing the diagnosis, control, and ultimate elimination of these neglected tropical diseases.
The Challenge of 'False Fascioliasis' (Pseudofascioliasis) and Diagnostic Specificity
False fascioliasis (pseudofascioliasis) represents a significant diagnostic challenge in the accurate identification of true Fasciola hepatica infection. This phenomenon occurs when Fasciola-like eggs appear in stool samples not as a result of an active infection, but rather from the recent consumption of contaminated liver from infected animals containing non-infective eggs [1] [64]. Within the broader context of Fasciola hepatica life cycle and egg morphology research, this diagnostic pitfall underscores the critical need for highly specific diagnostic tools that can differentiate between true infection and spurious passage. The parasite's complex life cycle, which involves multiple developmental stages in both intermediate snail hosts and definitive mammalian hosts, contributes to the diagnostic complexity [1] [7]. As researchers and drug development professionals seek to improve diagnostic accuracy and develop targeted interventions, understanding and addressing the challenge of pseudofascioliasis becomes paramount for both clinical management and research validity.
The morphological similarity between Fasciola hepatica eggs and those of other trematodes further compounds the diagnostic challenge. Eggs of Fasciola spp. are broadly ellipsoidal, operculated, and measure 130-150 µm long by 60-90 µm wide, making them difficult to distinguish from Fasciolopsis buski eggs based on morphology alone [1]. This overlap has significant treatment implications, as praziquantel is effective against Fasciolopsis buski but typically not against Fasciola parasites [64]. The diagnostic specificity required to differentiate these species highlights the necessity for advanced diagnostic approaches beyond simple microscopic examination.
The Biological Basis of Pseudofascioliasis
Life Cycle Stage Confusion and Egg Morphology
The phenomenon of pseudofascioliasis is intrinsically linked to the standard life cycle of Fasciola hepatica. In a true infection, adult flukes residing in the biliary ducts produce eggs that are discharged through the biliary system into the intestine and subsequently passed in stool [1] [46]. These eggs become embryonated in freshwater over approximately two weeks, after which they release miracidia that invade a suitable snail intermediate host [1]. The complex development within the snail involves several stages (sporocysts, rediae, and cercariae), with cercariae eventually being released to encyst as metacercariae on aquatic vegetation [1] [7]. When mammalian hosts ingest contaminated vegetation, the metacercariae excyst in the duodenum, penetrate the intestinal wall, and migrate through the liver parenchyma to the biliary ducts, where they mature into egg-producing adults [1] [46].
In contrast, pseudofascioliasis occurs when humans consume raw or undercooked liver from infected animals, introducing fully formed but non-infective eggs into the human digestive system. These eggs pass transiently through the gastrointestinal tract and can be detected in stool examinations, creating a false positive diagnostic scenario [1] [65]. The biological basis for this distinction lies in the origin of the eggs rather than their morphology, as eggs derived from consumed liver are morphologically identical to those produced in a true infection.
Diagnostic Implications of Egg Morphological Overlap
Recent research on Fasciola hepatica egg morphology has revealed additional complexities in differentiation. Studies have identified that eggs of F. hepatica may possess an appendage on the abopercular pole, a feature previously considered characteristic of Fascioloides species rather than Fasciola [23]. This finding challenges conventional morphological discrimination criteria and further complicates microscopic differentiation between species. The morphological characteristics of Fasciola eggs include:
- Overall shape: Broadly ellipsoidal [1]
- Operculum: Present at one end [1] [66]
- Abopercular end: Often has a roughened or irregular area [1]
- Appendages: Recent evidence shows some eggs may have an appendage on the anti-opercular pole [23]
- Size: 130-150 µm long by 60-90 µm wide [1] [66]
- Shell: Thin and smooth [66]
- Color: Yellowish-brown [65]
The presence of an appendage on Fasciola hepatica eggs, as documented in recent studies, indicates that this feature may not be a valid criterion for distinguishing between Fasciola and Fascioloides species as previously thought [23]. This morphological overlap represents a significant challenge for coprological diagnostics, which remains the gold standard for diagnosing fasciolosis in humans and animals worldwide [23].
Diagnostic Approaches and Specificity Challenges
Current Diagnostic Modalities and Limitations
The diagnosis of true fascioliasis employs multiple complementary approaches, each with distinct advantages and limitations concerning specificity:
Table 1: Diagnostic Methods for Fascioliasis and Their Limitations
| Diagnostic Method | Application | Limitations Regarding Specificity |
|---|---|---|
| Coprological examination | Detection of eggs in stool, duodenal, or biliary aspirates [1] | Cannot distinguish between true infection and pseudofascioliasis; difficult morphological differentiation from Fasciolopsis buski and some Echinostoma spp. [1] [64] |
| Serologic testing | Detection of specific antibodies to Fasciola [1] | Can rule out pseudofascioliasis; may cross-react with other helminth infections [1] [59] |
| Imaging techniques | Ultrasonography, CT, MRI, ERCP [1] [64] | Direct visualization of flukes via ERCP provides confirmation but is invasive [1] |
| Histopathologic examination | Biopsy of liver or other tissue [64] | Provides definitive diagnosis but is invasive and dependent on sampling [64] |
The timing of diagnostic application is crucial, as serologic tests can detect specific antibodies to Fasciola 2-4 weeks after infection, whereas egg production typically does not start until 3-4 months post-exposure [1] [64]. Even during the chronic phase of infection, low-level or sporadic egg production may necessitate examination of multiple stool specimens, particularly in cases of light infection [64].
Protocols for Differentiating True Infection from Pseudofascioliasis
To address the challenge of pseudofascioliasis, specific diagnostic protocols have been developed:
1. Liver-Free Diet and Repeat Stool Examination
- Have the patient abstain from eating liver for several days [1] [65]
- Collect repeat stool specimens after this period [1]
- Continued presence of eggs suggests true infection, while disappearance indicates pseudofascioliasis [1]
2. Serologic Testing Protocol
- Initial screening with enzyme immunoassay (EIA) using excretory-secretory (ES) or recombinant antigens [1] [59]
- Confirmatory testing of EIA-positive specimens with immunoblot assay [1]
- CDC immunoblot assay based on recombinant F. hepatica antigen (FhSAP2) has reported sensitivity of ≥94% and specificity of ≥98% for chronic infection [1]
3. Endoscopic Retrograde Cholangiopancreatography (ERCP)
- Direct visualization of biliary ducts [1]
- Can identify adult flukes in the common bile duct [1]
- Allows for simultaneous extraction of flukes for both diagnosis and treatment [64]
The following diagnostic workflow illustrates the comprehensive approach required to differentiate true fascioliasis from pseudofascioliasis:
Diagram 1: Diagnostic workflow for differentiating true fascioliasis from pseudofascioliasis
Advanced Diagnostic Solutions and Research Reagents
Serological Antigens and Their Performance Characteristics
Serological diagnosis has emerged as a crucial tool for addressing the specificity challenges posed by pseudofascioliasis. Various native and recombinant antigens have been developed and evaluated for their diagnostic performance:
Table 2: Research Reagent Solutions for Fascioliasis Serodiagnosis
| Antigen Type | Specific Antigens | Performance Characteristics | Applications and Advantages |
|---|---|---|---|
| Native Antigens | Excretory-secretory (ES) antigens [59] | Sensitivity: 0.931-0.985; Specificity: 0.959-0.997 [59] | Contains natural mixture of parasite proteins; well-established protocol [59] |
| Recombinant Antigens | FhSAP2 (recombinant saposin-like protein 2) [1] [59] | Sensitivity: ≥94%; Specificity: ≥98% [1] | Standardized production; cost-effective; CDC-approved immunoblot [1] [59] |
| Recombinant Proteases | Cathepsin L1 (rCL-1) [59] | High accuracy in meta-analysis [59] | Principal components of ES antigens; involved in host invasion [59] |
| Other Recombinant Antigens | Leucine aminopeptidase (rLAP), Glutathione S-transferase [59] | Good diagnostic accuracy [59] | Additional targets for assay development [59] |
The meta-analysis of serological tests for fascioliasis revealed that all major antigen types demonstrated good accuracy, with FhES (Fasciola hepatica excretory-secretory), FhrCL-1 (recombinant cathepsin L1), and FhrSAP-2 (recombinant saposin-like protein 2) antigens providing the best results for serum diagnosis of human and animal fasciolosis [59]. The production of recombinant antigens offers significant advantages for standardization and cost-effectiveness compared to ES antigen preparation, which requires maintenance of the parasite life cycle [59].
Experimental Protocols for Diagnostic Development
Protocol 1: Immunoblot Assay Using Recombinant FhSAP2 Antigen
- Based on CDC-developed CLIA-approved immunoblot assay [1]
- Utilizes recombinant F. hepatica antigen (FhSAP2) [1]
- Procedure: Serum samples incubated with FhSAP2 antigen membrane; positive reaction defined as presence of band at ~38 kDa [1]
- Interpretation: Sensitivity ≥94% (16/17), specificity ≥98% (113/115) for chronic human infection [1]
- Note: Validation for acute infection ongoing [1]
Protocol 2: Indirect ELISA for Serodiagnosis
- Uses either native ES or recombinant antigens (cathepsin L1, SAP-2) [59]
- Antigens coated onto microtiter plates [59]
- Patient serum added, followed by enzyme-conjugated secondary antibody [59]
- Substrate added, optical density measured [59]
- Optimal cut-off values determined through receiver operating characteristic (ROC) analysis [59]
Protocol 3: Fecal DNA Extraction and Molecular Analysis
- Eggs obtained via sedimentation coprological technique [23]
- Eggs stored in tap water for miracidia development [23]
- Individual eggs collected using eyelash handmade manipulator [23]
- DNA extraction using commercial kits (e.g., QIAamp DNA Accessory Set) [23]
- Amplification of internal transcribed spacer (ITS) region using primers BD1, BD2 [23]
- Sequence analysis for species identification [23]
Research Gaps and Future Directions
Despite advances in diagnostic techniques, several significant research gaps remain in addressing the challenge of pseudofascioliasis and improving diagnostic specificity. The development of reliable point-of-care tests using recombinant antigens represents a promising direction, particularly for use in endemic areas with limited laboratory infrastructure [59]. Further validation of existing assays, including the CDC FhSAP2 immunoblot for acute infection, is needed to expand their clinical utility [1]. Additionally, more comprehensive studies on egg morphology, including the recently described appendage feature, are required to refine coprological diagnostic criteria and improve morphological differentiation from other trematode species [23].
The genomic and proteomic characterization of Fasciola hepatica continues to identify new potential targets for diagnostic development. Principal proteases involved in host invasion, particularly the cathepsin L family, offer promising candidates for highly specific diagnostic antigens [59]. As research progresses, the integration of multiple diagnostic approaches—coprological, serological, molecular, and imaging—will be essential for achieving the specificity required to definitively differentiate true fascioliasis from pseudofascioliasis across different clinical phases and epidemiological contexts.
For drug development professionals, the accurate diagnosis of true fascioliasis is essential for clinical trial enrollment and therapeutic efficacy assessment. The persistence of pseudofascioliasis as a diagnostic challenge underscores the need for ongoing research into more specific diagnostic tools that can support the development and evaluation of new interventional strategies against this significant parasitic disease.
The egg morphology of Fasciola hepatica, a parasitic trematode of significant medical and veterinary importance, has long been considered a stable taxonomic characteristic. Recent research has revealed previously underreported morphological variants, specifically abopercular appendages and knobs, which challenge conventional diagnostic paradigms. This technical guide details the discovery of these structures, their potential impact on the accuracy of coprological diagnostics, and their implications for understanding the parasite's biology. Framed within a broader thesis on the life cycle and egg morphology of F. hepatica, this review provides researchers and drug development professionals with advanced methodologies for morphological and molecular characterization, ensuring accurate species identification in both routine diagnostics and research settings.
The life cycle of Fasciola hepatica is complex and indirect, involving multiple larval stages in an intermediate snail host and mammalian definitive hosts, most commonly ruminants [1] [11]. Adult flukes residing in the bile ducts produce eggs that are passed into the environment via the host's feces. These eggs are operculated and unembryonated when shed, and their morphology has been a cornerstone of coprological diagnosis for decades [1] [27]. The standard description characterizes F. hepatica eggs as broadly ellipsoidal, measuring 130–150 µm in length by 60–90 µm in width, with a distinct operculum at one end and a sometimes roughened area at the abopercular end [1].
The diagnosis of fascioliasis through fecal examination remains the gold standard in both human and veterinary medicine, as it provides definitive evidence of active infection [23] [27]. Consequently, precise identification of Fasciola eggs is critical. However, this task is complicated by significant morphological overlaps with eggs from related trematodes, such as Fasciolopsis buski, and even between the two major Fasciola species, F. hepatica and F. gigantica [1] [67]. The recent discovery of consistent morphological variations on the eggshell introduces a new layer of complexity to their identification and warrants in-depth investigation.
Morphological Analysis of Abopercular Appendages and Knobs
Description of Novel Morphological Features
Traits once considered diagnostic for other trematode genera have been identified in Fasciola hepatica. A 2024 study by Loginova et al. reported the presence of an abopercular appendage on F. hepatica eggs recovered from both reindeer feces and the uteri of adult flukes [23] [4]. This appendage is a filamentous or protrusive structure located at the pole opposite the operculum.
The study also detailed other critical morphological traits, which are summarized in Table 1 below. These features collectively contribute to a more nuanced understanding of F. hepatica egg morphology.
Table 1: Key Morphological Traits of Fasciola hepatica Eggs
| Morphological Trait | Description | Research Significance |
|---|---|---|
| Abopercular Appendage | A filamentous or protrusive structure at the pole opposite the operculum. | Previously considered a potential distinguishing feature for Fascioloides magna; its presence in F. hepatica complicates differential diagnosis [23]. |
| Abopercular Knob | A thickening or protuberance at the abopercular end. | May be associated with the site of shell formation or larval release; function requires further study [23]. |
| Eggshell Thickening | Notable thickening of the shell at the abopercular end. | Could influence egg resistance to environmental stressors; may be visualized via light and scanning electron microscopy [23]. |
| Egg Shape | Broadly ellipsoidal or oval. | Classic diagnostic feature; however, shape can exhibit minor variability [1] [23]. |
Quantitative Morphometric Data
The 2024 study provided detailed morphometric data, confirming that eggs possessing an appendage fall within the established size range for Fasciola hepatica, yet the appendage itself represents a distinct and variable structure.
Table 2: Morphometric Data of Fasciola hepatica Eggs with Appendages (Loginova et al., 2024)
| Parameter | Measurement (in µm) | Notes |
|---|---|---|
| Egg Length | 130.7 - 150.4 | Within the classic range for F. hepatica [1] [23]. |
| Egg Width | 70.1 - 90.3 | Within the classic range for F. hepatica [1] [23]. |
| Appendage Length | Up to 15.8 | Highly variable; some eggs display only a small knob [23]. |
| Appendage Width | Up to 12.5 | Appears as a continuation of the eggshell [23]. |
The presence of these appendages challenges existing taxonomic keys. Historically, an abopercular appendage was a key characteristic used to differentiate the eggs of Fascioloides magna from those of Fasciola species [23]. The finding that this structure is not a reliable criterion for genus-level distinction has immediate and important implications for diagnostic parasitology, particularly in regions where these parasites are co-endemic.
Research Significance and Implications
Impact on Diagnostic Specificity and Sensitivity
The discovery of appendages on F. hepatica eggs directly impacts the specificity of coprological examinations. Misidentification can lead to false reports of Fascioloides magna, a parasite with different life cycle dynamics and management strategies, or vice-versa [23]. This is critical for wildlife management and livestock health. Furthermore, the morphological overlap between F. hepatica and F. gigantica eggs is well-documented, and the existence of hybrid or intermediate forms in areas where the two species are co-endemic further complicates diagnosis based on morphology alone [1] [67]. Reliance on classic egg size parameters has been shown to be unreliable for differential diagnosis in human infections [27].
Insights into Biology and Development
Beyond diagnostics, these morphological variants offer a window into the developmental biology of the parasite. The abopercular knob is hypothesized to be related to the site of eggshell formation or a point of weakness for larval release [23]. The variability in appendage presence and size—from a pronounced filament to a mere knob—suggests a potential influence of host-specific factors, environmental conditions, or genetic variability within F. hepatica populations. This underscores that egg morphology is not a static trait but may be a plastic character influenced by multiple factors.
Experimental Protocols for Morphological and Molecular Characterization
To accurately identify Fasciola species and their morphological variants, an integrated approach combining traditional morphology with molecular techniques is essential. The following protocols are recommended.
Protocol 1: Comprehensive Egg Recovery and Morphological Analysis
This protocol is adapted from the methodologies used in recent studies to identify abopercular appendages [23].
Key Research Reagent Solutions:
- Sedimentation Solution: Tap water or deionized water is used in sequential washing and sedimentation steps to concentrate eggs.
- Microscopy Mounting Medium: Tap water is sufficient for temporary wet mounts for immediate examination.
- Digital Imaging Software: Fiji/ImageJ software (National Institutes of Health, USA) is critical for precise morphometric analysis of captured micrographs.
Detailed Procedure:
- Sample Homogenization: Homogenize 3 grams of host feces using a mortar and pestle.
- Sequential Washing and Sedimentation:
- Add 50 mL of water to the homogenized feces and mix carefully.
- Filter the mixture through two layers of synthetic fiber with a 1 mm mesh diameter.
- Allow the filtered mixture to settle for 5 minutes until a sediment forms.
- Decant the supernatant.
- Resuspend the sediment in 50 mL of fresh water and repeat the sedimentation process until the supernatant is clear.
- Microscopic Examination: Pour the final sediment onto a glass slide, cover with a cover slip (24x24 mm), and examine systematically under a light microscope using 40x and 100x objective lenses.
- Imaging and Morphometry: Capture digital micrographs of eggs. Use calibrated software (e.g., Fiji/ImageJ) to measure key dimensions: egg length, egg width, and, if present, appendage length and width.
- 3D Modeling (Optional): For advanced visualization and presentation, 3D models of eggs can be created using software such as Blender and ZBrush, referencing both original light micrographs and published scanning electron microscopy (SEM) images [23].
The workflow for this integrated diagnostic and research approach is outlined in the diagram below.
Protocol 2: Molecular Confirmation of Species and Hybrids
Morphological analysis alone is insufficient for definitive identification, especially in co-endemic areas. Molecular techniques are required for confirmation, as demonstrated in studies from South Africa and elsewhere [67].
Key Research Reagent Solutions:
- DNA Extraction Kit: The QIAamp DNA Accessory Set, Micro kit (Qiagen) is effective for extracting DNA from individual or pooled eggs.
- PCR Reagents: Specific primers targeting genetic markers such as BD1 and BD2 for the Internal Transcribed Spacer (ITS1, 5.8S, ITS2) region, or primers for the cytochrome c oxidase subunit I (CO1) gene.
- Agarose Gel Electrophoresis System: For visualizing successful PCR amplification.
- Sanger Sequencing: For definitive genetic characterization.
Detailed Procedure:
- DNA Extraction:
- Isolate individual eggs or a pool of eggs (≥65 eggs) from the sediment using a fine manipulator.
- Place samples in 1.5 mL tubes and freeze at -70°C to rupture the eggshell.
- Extract genomic DNA using a commercial kit according to the manufacturer's instructions.
- PCR Amplification:
- Amplify the target genetic regions (e.g., ITS or CO1) using species-specific primers.
- Use a standard PCR thermal cycler protocol with an annealing temperature optimized for the primer set.
- Genetic Analysis:
- Visualize PCR products on an agarose gel to confirm amplification.
- Purify and sequence the PCR products.
- Analyze the resulting sequences using bioinformatics tools (e.g., BLAST, phylogenetic analysis) to confirm species identity ( F. hepatica, F. gigantica) or detect potential hybrid forms.
The decision-making process for integrating these methods is summarized in the following workflow.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Fasciola Egg Research
| Item | Function/Application | Example/Note |
|---|---|---|
| Sedimentation Apparatus | Concentration of helminth eggs from fecal samples. | Mortar, pestle, filtration mesh, and sedimentation cups [23]. |
| High-Resolution Microscopy | Visualization and imaging of egg morphological details. | Light microscope with 40x and 100x oil immersion objectives and digital camera [23]. |
| Image Analysis Software | Precise morphometric measurements from digital micrographs. | Fiji/ImageJ software for standardized measurements [23]. |
| DNA Extraction Kit | Isolation of high-quality genomic DNA from eggs or adult flukes. | QIAamp DNA Micro Kit, suitable for small quantities of material [23]. |
| PCR Reagents & Primers | Amplification of species-specific genetic markers. | Primers for the ITS region (e.g., BD1/BD2) or the CO1 gene [23] [67]. |
| Sanger Sequencing | Determination of DNA sequence for phylogenetic analysis. | Required for definitive identification of species and hybrid forms [67]. |
The identification of abopercular appendages and knobs on the eggs of Fasciola hepatica represents a significant advance in our understanding of the parasite's morphology. It necessitates a revision of traditional diagnostic keys and highlights the inherent limitations of relying solely on coproscopy for species identification. For researchers and drug development professionals, these findings underscore the critical importance of using integrated morphological and molecular approaches, particularly in epidemiological studies, the assessment of drug efficacy, and the investigation of parasite population genetics.
Future research should focus on elucidating the functional role of these appendages and the factors that influence their expression. Large-scale studies across different geographic regions and host species are needed to determine the prevalence and full range of these morphological variants. Ultimately, incorporating these findings into diagnostic guidelines will enhance the accuracy of fascioliasis surveillance and control programs, contributing to the broader goal of mitigating the impact of this neglected tropical disease.
Fascioliasis, caused by the liver fluke Fasciola hepatica, represents a significant global foodborne zoonosis and is recognized as a neglected tropical disease by the World Health Organization. Triclabendazole (TCBZ) has remained the anthelmintic of choice for both livestock and human infections due to its superior efficacy against both immature and adult parasite stages. However, the widespread emergence of TCBZ resistance (TCBZ-R) threatens disease control programs worldwide. Resistance has been documented on every continent in livestock, and increasingly, treatment failures are being reported in human populations [68] [69] [70]. The situation is particularly acute in endemic regions such as the Andean areas of South America, where a study revealed that only 55% of children with chronic fascioliasis achieved parasitological cure after the first round of TCBZ treatment, a significant decline from the 95% efficacy rate previously observed [71]. This technical guide frames the challenge of TCBZ resistance within the broader context of Fasciola hepatica biology, exploring the essential foundation provided by understanding the parasite's life cycle and egg morphology, which are critical for designing effective drug screening programs and diagnosing infections in the field.
Fasciola hepatica Biology: Foundation for Research
Life Cycle and Its Implications for Drug Screening
The complex life cycle of Fasciola hepatica directly influences experimental design for drug screening. The parasite requires an intermediate snail host and a mammalian definitive host, with several developmental stages that present different drug susceptibilities [1] [7]. Adult flukes residing in the bile ducts produce eggs that are excreted in the feces of the infected host. These eggs embryonate in freshwater over approximately two weeks, releasing miracidia that invade suitable snail intermediate hosts. Within the snail, the parasite undergoes several multiplicative developmental stages (sporocysts and rediae), eventually producing cercariae that are released into the environment. Cercariae encyst as metacercariae on aquatic vegetation, which are then ingested by the definitive host. After ingestion, metacercariae excyst in the duodenum, penetrate the intestinal wall, and migrate through the liver parenchyma before reaching the bile ducts, where they mature into adults [1] [46] [72]. This cycle takes 18-20 weeks to complete [7]. The differential sensitivity of these various stages to anthelmintics necessitates that drug screening programs evaluate compounds against both immature migratory stages and adult flukes to fully assess therapeutic potential.
Egg Morphology and Diagnostic Significance
Egg morphology represents a critical diagnostic tool and potential indicator of drug efficacy. Fasciola hepatica eggs are broadly ellipsoidal, operculated, and measure 130–150 µm long by 60–90 µm wide [1]. They are passed unembryonated in feces, and their detection through coprological examination remains the gold standard for diagnosing patent infections [23]. Recent research has revealed that eggs may possess a previously unreported appendage on the abopercular pole, a finding that may contribute to improved diagnostics and differential diagnosis from other trematode eggs [23]. The Egg Reduction Rate (ERR), calculated by comparing egg counts before and after treatment, serves as a primary endpoint for assessing drug efficacy in both clinical and research settings. A thorough understanding of egg morphology is therefore indispensable for accurate diagnosis and for evaluating the success of novel compounds in preclinical trials.
Current Understanding of Triclabendazole Resistance Mechanisms
Genetic Basis of Resistance
Recent genomic studies have substantially advanced our understanding of the genetic architecture underlying TCBZ-R. Research employing genetic crosses and whole-genome sequencing has identified a ~3.2 megabase pair locus in the 1.2 gigabase pair F. hepatica genome that confers TCBZ resistance. This locus, representing just 0.25% of the genome, was validated in both experimental and natural infections and demonstrates dominant inheritance, meaning parasites only need to inherit one copy of this resistance region to survive treatment [68] [73]. The resistance locus contains approximately 30 genes with functions in membrane transport (e.g., ATP-binding cassette family B, ABCB1), transmembrane signaling and signal transduction (e.g., GTP-Ras-adenylyl cyclase and EGF-like protein), DNA/RNA binding and transcriptional regulation (e.g., SANT/Myb-like DNA-binding domain protein), and drug storage and sequestration (e.g., fatty acid binding protein, FABP) [68].
However, resistance mechanisms appear to have evolved independently across different geographical populations. A 2025 study comparing resistant flukes from Peru and the United Kingdom found distinct selection signatures, suggesting independent genetic origins of TCBZ-R in these populations [71]. The Peruvian study identified genomic regions of high differentiation that encode genes involved in the EGFR-PI3K-mTOR-S6K pathway and microtubule function, which were distinct from the QTLs identified in UK flukes [71].
Table 1: Documented Genetic Mechanisms of Triclabendazole Resistance
| Resistance Mechanism | Key Genes/Proteins Involved | Geographical Validation | Inheritance Pattern |
|---|---|---|---|
| Major Locus | ABCB1, Ras-adenylyl cyclase, EGF-like protein, FABP | United Kingdom, Cumbria | Dominant [68] |
| Independent Selection | EGFR-PI3K-mTOR-S6K pathway, Microtubule-related genes | Peru, Cusco Region | Polygenic [71] |
| Candidate Gene Approaches | β-tubulin, P-glycoprotein (Pgp), GST, FMO, CYP450 | Various (inconsistent validation) | Not fully established [68] [70] |
Proposed Physiological Resistance Mechanisms
Several physiological mechanisms have been proposed to explain TCBZ resistance, though the evidence remains varied:
- Altered drug uptake and efflux: Enhanced activity of P-glycoprotein (P-gp) drug efflux pumps (particularly ABCB1) may reduce intracellular concentrations of TCBZ and its metabolites in resistant flukes [68] [70]. A specific SNP (T687G) causing an amino acid substitution in P-gp was linked to TCBZ-R in one study, though subsequent research failed to corroborate this association in other geographical populations [71].
- Metabolic detoxification: Increased expression or activity of detoxifying enzymes such as glutathione S-transferase (GST) and flavin monooxygenase (FMO) may enhance the conversion of the active TCBZ sulfoxide metabolite to the less active sulfone form [71] [70].
- Target-site modification: While β-tubulin mutations have been implicated in benzimidazole resistance in other helminths, no consistent mutations have been definitively linked to TCBZ-R in Fasciola hepatica [68] [70].
Experimental Framework for Screening Novel Compounds
Parasite Material and Culture Systems
The reliability of drug screening data fundamentally depends on the quality and characterization of parasite material. Researchers should establish and maintain well-characterized isolates with defined TCBZ susceptibility profiles:
- Reference isolates: Maintain TCBZ-sensitive (TCBZ-S) and TCBZ-resistant (TCBZ-R) isolates as controls. The FhLivS1 (sensitive) and FhLivR1 (resistant) isolates have been genetically characterized and used in experimental crosses [68].
- Field isolates: Regularly collect and phenotype field isolates to ensure screening programs account for current field resistance patterns. Genome-wide sequencing of 99 TCBZ-S and 210 TCBZ-R adult flukes from Peru demonstrated the feasibility of large-scale phenotyping [71].
- Life stage availability: For comprehensive screening, establish systems to obtain multiple developmental stages, particularly adults for initial screening and metacercariae for validation studies.
Table 2: Essential Research Reagents for TCBZ Resistance Research
| Reagent/Category | Specific Examples | Research Application | Key Function in Research |
|---|---|---|---|
| Parasite Isolates | FhLivS1 (TCBZ-S), FhLivR1 (TCBZ-R), Field isolates from endemic areas | All experimental procedures | Provide genetically defined biological material with known resistance profiles [68] |
| Snail Intermediate Hosts | Galba truncatula, other Lymnaeidae species | Life cycle maintenance | Support parasite development from miracidia to cercariae for complete lifecycle studies [1] [7] |
| Molecular Biology Kits | QIAamp DNA Accessory Set, Micro kit | Genetic analysis | Extract high-quality DNA from parasite samples for genomic studies [23] |
| Primers for Genotyping | BD1, BD2 for ITS region amplification | Species identification and phylogenetics | Amplify specific genetic regions for parasite identification and population studies [23] |
| TCBZ Metabolites | Triclabendazole sulfoxide, Triclabendazole sulfone | In vitro drug sensitivity assays | Test against parasite stages to determine efficacy of active drug components [71] |
In Vitro Phenotyping Assays
Robust phenotyping is essential for correlating genotypic findings with resistance status. The following assays provide standardized approaches:
Motility and Vitality Assays:
- Principle: Measure parasite motility or metabolic activity as indicators of viability after drug exposure.
- Protocol: Expose adult flukes to serial dilutions of TCBZ metabolites (particularly TCBZ-sulfoxide) and score motility at regular intervals (e.g., 0, 3, 6, 12, 24 hours). The Peruvian study titrated drug concentration and exposure time to classify parasites in the upper and lower quartiles of the susceptibility distribution as TCBZ-S and TCBZ-R, respectively [71].
- Endpoint determination: Establish threshold values for significant motility reduction (e.g., >50% reduction compared to untreated controls).
Histological Assessment:
- Principle: Evaluate drug-induced morphological damage to the tegument and internal organs.
- Protocol: Fix drug-exposed and control flukes and process for histological examination (e.g., hematoxylin and eosin staining, electron microscopy).
- Scoring system: Develop a standardized scoring system for tegumental damage, gut integrity, and reproductive organ morphology.
Genotyping and Molecular Diagnostics
Molecular tools enable the detection of resistance markers and inform on resistance mechanisms:
Whole-Genome Sequencing:
- Application: Identify selection signatures and novel resistance loci through genome-wide scans.
- Protocol: Extract high-quality DNA from phenotypically characterized flukes. Sequence to sufficient coverage (≥11.6× mean coverage of the 1.2 Gb reference genome, as achieved in the Peruvian study [71]). Analyze for FST outliers to detect genomic regions under selection.
Targeted Genotyping:
- Application: Screen for known resistance markers in field populations.
- Protocol: Design assays for specific SNPs within the major resistance locus (UK population) or EGFR-PI3K-mTOR-S6K pathway genes (Peruvian population). The Peruvian study demonstrated that just 30 SNPs could differentiate between TCBZ-S and TCBZ-R parasites with ≥75% accuracy [71].
The following diagram illustrates the integrated experimental workflow for compound screening and resistance mechanism elucidation:
High-Throughput Screening Strategies for Novel Compounds and Plant Extracts
Compound Libraries and Natural Product Screening
A diversified screening approach increases the probability of identifying novel chemical scaffolds with activity against TCBZ-R flukes:
- Synthetic compound libraries: Focus on libraries with known activity against eukaryotic pathogens or specific molecular targets (e.g., kinase inhibitors, microtubule disruptors).
- Natural product extracts: Screen plant extracts with historical ethnopharmacological use against parasitic infections, particularly from plant families with known anthelmintic properties (e.g., Asteraceae, Fabaceae, Meliaceae).
- Repurposing approaches: Evaluate existing anthelmintics and approved human drugs for synergistic activity with TCBZ against resistant strains.
Tiered Screening Approach
A multi-tiered screening strategy optimizes resource allocation and increases the probability of identifying promising candidates:
Primary Screening:
- Platform: Whole-organism adult flukes or newly excysted juveniles (NEJ) in vitro.
- Concentration: Single high concentration (e.g., 10-50 µM for pure compounds; 100 µg/mL for plant extracts) to identify active candidates.
- Endpoint: Motility inhibition or viability assessment after 24-72 hours exposure.
- Throughput: Medium to high (96-well format possible with NEJ).
Secondary Screening:
- Platform: Dose-response curves against both TCBZ-S and TCBZ-R isolates.
- Concentration: 5-8 point serial dilutions to determine IC50 values.
- Endpoint: Quantitative viability metrics (e.g., ATP-based assays, fluorescent viability dyes).
- Counter-screening: Assess cytotoxicity against mammalian cell lines to determine selectivity indices.
Tertiary Screening:
- Platform: In vivo validation in rodent models of fasciolosis.
- Dosing: Multiple administration routes and regimens.
- Endpoint: Worm burden reduction, histopathological assessment, egg count reduction.
The following pathway diagram illustrates key molecular targets implicated in TCBZ resistance, which should inform targeted drug discovery efforts:
The escalating problem of triclabendazole resistance in Fasciola hepatica demands urgent and innovative solutions. This guide has outlined a comprehensive framework for screening novel compounds and plant extracts, grounded in the latest understanding of resistance genetics and parasite biology. The recent discoveries of distinct resistance loci in different geographical populations highlight the necessity for global surveillance and regionally tailored interventions. Future research should prioritize the identification of the specific causal genes within the major resistance locus, the development of field-deployable molecular diagnostics, and the exploration of combination therapies targeting multiple resistance mechanisms simultaneously. By integrating robust phenotypic screening with genomic technologies and a fundamental understanding of parasite biology, researchers can contribute to overcoming one of the most significant challenges in parasitic disease control.
Within the broader context of Fasciola hepatica life cycle and egg morphology research, assessing the efficacy of new anthelmintic compounds is a cornerstone of parasitology and drug development. The adult Fasciola hepatica, a large liver fluke residing in the bile ducts of its definitive host, is characterized by a robust tegument that is essential for nutrient absorption, immune evasion, and protection against host digestive fluids [1]. The life cycle of this parasite is complex, involving multiple stages both inside and outside the host [1] [46] [7]. Anthelmintic efficacy evaluation has increasingly focused on tegumental damage as a primary indicator of drug action, which must be correlated with definitive measures of parasite viability to confirm lethal effects. This guide provides drug development professionals with advanced methodologies to quantitatively link morphological changes in the tegument with irreversible parasite death, thereby validating the parasiticidal potential of novel therapeutic agents. The resilience of F. hepatica eggs, which are broadly ellipsoidal and operculated, and their passage through the biliary system and into the environment underscores the necessity of treatments that effectively kill adult flukes to break the transmission cycle [1] [4] [5].
Methodologies for Assessing Tegument Damage
The tegument of helminths is a dynamic cellular layer, and its disruption is often one of the first observable effects of anthelmintic drug action. Detailed assessment requires a combination of observational and quantitative techniques.
In Vitro Tegument Damage Assay
This protocol evaluates the direct morphological impact of compounds on adult flukes, adapted from studies on schistosomes [74].
- Parasite Material: Obtain adult F. hepatica from experimentally infected animals or from abattoirs. Maintain flukes in sterile, pre-warmed culture medium (e.g., RPMI-1640 with antibiotics) for an acclimatization period of 2-4 hours prior to drug exposure.
- Drug Exposure: Incubate flukes in culture medium containing the anthelmintic compound at the desired concentration (e.g., 1x, 10x IC₅₀). A vehicle control (e.g., DMSO <0.1%) must be included. Incubation is typically carried out at 37-38°C in a 5% CO₂ atmosphere for a period of 2-24 hours.
- Microscopic Evaluation: Post-incubation, flukes are processed for assessment.
- Light Microscopy: Examine live or fixed flukes for gross morphological changes such as blebbing, swelling, vacuolization, and erosion.
- Scanning Electron Microscopy (SEM): Fix flukes in 2.5% glutaraldehyde, followed by dehydration in a graded ethanol series and critical point drying. Gold/palladium sputtering is performed before visualization under SEM to reveal ultrastructural damage to the tegument, including spine loss, lesion formation, and disruption of the syncytial layer.
Scoring System for Tegument Damage
A semi-quantitative scoring system allows for the standardization of observations. The following table outlines a typical scoring matrix.
Table 1: Tegument Damage Scoring Matrix for Microscopic Evaluation
| Score | Severity | Observed Morphological Changes |
|---|---|---|
| 0 | None | Tegument appears normal, with no visible lesions or disruption. |
| 1 | Mild | Minor blebbing or focal swelling; slight distortion of spines. |
| 2 | Moderate | Extensive blebbing, visible vacuolization, and multiple lesions. |
| 3 | Severe | Frank erosion, large areas of sloughing, and extensive tegument loss. |
Methodologies for Determining Parasite Viability
Tegument damage is indicative of stress but does not invariably confirm parasite death. Correlating this damage with robust viability assays is critical. Viability can be defined as the parasite's capacity for continued growth, development, and reproduction [75].
Motility Assays
The simplest viability assays involve monitoring parasite movement.
- Visual Inspection: Direct observation of flukes under a microscope to grade motility on a scale (e.g., 0 = immobile, 1 = only oral sucker movement, 2 = slow whole-body movement, 3 = vigorous movement).
- Videomicroscopy and Image Analysis: Automated systems can track movement over time, providing quantitative data on motility reduction as a proxy for viability loss.
Metabolic and Staining-Based Viability Assays
These assays probe the functional integrity of key cellular processes.
- MitoTracker Staining: This flow cytometry-based assay uses dyes (e.g., MitoTracker Deep Red) that accumulate in active mitochondria. Viable parasites with intact mitochondrial membranes retain the dye, while non-viable parasites show diminished fluorescence [76]. This method, known as Direct Viability Assessment (DVA), offers a rapid and quantitative measure.
- ATP-based Luminescence Assays: Measurement of cellular ATP levels using luciferase-based kits. A significant drop in ATP correlates with loss of metabolic activity and viability.
Gold Standard: Parasite Recovery and Reproduction Assays
The most definitive proof of viability is the demonstration of reproductive capacity post-treatment.
- In Vitro Culture: After drug exposure and washout, flukes are maintained in drug-free culture medium for an extended period (days to weeks) to assess recovery of motility and, crucially, the continued production of viable eggs [1]. For F. hepatica, egg production typically begins 3-4 months post-infection in a host, but in vitro systems can monitor the viability of eggs produced by adult flukes [1].
- Parasite Reduction Ratio (PRR) Assay: Adapted from malaria research, this assay involves a limiting dilution of parasites after drug treatment and long-term culture to see if a single parasite can re-establish a population [76]. It is highly reliable but resource-intensive and time-consuming (14-21 days).
Table 2: Comparison of Key Parasite Viability Assays
| Assay Type | Measured Endpoint | Key Advantage | Key Limitation |
|---|---|---|---|
| Motility | Physical movement | Simple, low-cost | Subjective; may not correlate with death |
| MitoTracker (DVA) | Mitochondrial activity | Rapid, quantitative, high-throughput | Requires flow cytometer and expertise [76] |
| ATP Assay | Metabolic activity | Highly sensitive, quantitative | Does not distinguish between dying and dead |
| Reproduction (Egg Count) | Reproductive capacity | Functional, definitive | Very long duration (weeks-months) [1] |
| PRR Assay | Clonal expansion capacity | Gold standard for killing kinetics | Very long duration and resource-heavy [76] |
Correlating Tegument Damage with Viability Loss
Establishing a predictive relationship between the speed and severity of tegument damage and the irreversible loss of viability is the ultimate goal for high-throughput drug screening.
Integrated Experimental Workflow
A robust correlation study follows a defined pathway where tegument damage and viability are assessed in parallel on the same parasite population.
Pharmacodynamic Modeling of Killing Kinetics
Advanced analysis involves modeling the time-course of both damage and death. Research on antimalarials provides a framework, defining parameters such as the maximum killing rate (Emax) and the lag phase before onset of action [76]. For instance, a study comparing viability assays found that drugs like dihydroartemisinin act with no lag phase (Emax ~0.05-0.07 h⁻¹), while atovaquone has a significant lag phase (>40h) before killing begins [76]. Applying this to anthelmintics, one could model how rapidly tegument damage (e.g., the progression to a score of 3) predicts the subsequent exponential decline in viable parasites measured by MitoTracker or PRR.
Table 3: Pharmacodynamic Parameters from Parasite Viability Assays (Adapted from Antimalarial Research)
| Drug Profile | Lag Phase | Max Killing Rate (Emax) | Correlation with Tegument Damage |
|---|---|---|---|
| Rapid-Acting | Short or None | High (>0.05 h⁻¹) | Tegument damage is immediate and severe, strongly predicting rapid viability loss. |
| Delayed-Acting | Long (>40 h) | Moderate | Initial tegument damage may be minimal; damage severity increases after the lag phase. |
| Slow-Acting | Variable | Low | Weak or inconsistent correlation; viability loss may occur with minimal overt tegument damage. |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful execution of these correlative studies requires a suite of specialized reagents and tools.
Table 4: Key Research Reagent Solutions for Tegument and Viability Studies
| Reagent/Material | Function | Specific Example |
|---|---|---|
| Defined Culture Media | Maintenance of adult flukes in vitro for drug exposure. | RPMI-1640 or DMEM, supplemented with serum and antibiotics. |
| Reference Anthelmintics | Positive controls for inducing known levels of tegument damage and viability loss. | Praziquantel, Triclabendazole, Albendazole [77]. |
| MitoTracker Dyes | Staining of active mitochondria for flow cytometry-based viability assessment (DVA) [76]. | MitoTracker Deep Red FM. |
| Fixatives for SEM | Preservation of parasite ultrastructure for high-resolution imaging of the tegument. | Glutaraldehyde (2.5%), Paraformaldehyde. |
| ATP Detection Kits | Quantification of cellular ATP levels as a measure of metabolic viability. | Luciferase-based luminescent assay kits. |
| Species-Specific Antibodies | Immunofluorescence staining of tegument-specific antigens to visualize damage. | Anti-Fasciola tegument serum. |
| Automated Motility Analyzers | Quantitative, high-throughput assessment of parasite movement. | Video tracking systems with specialized software. |
In the context of Fasciola hepatica research, from its complex life cycle to the distinct morphology of its eggs, the drive to develop new anthelmintics is critical. The approach outlined herein—rigorously correlating quantifiable tegument damage with definitive measures of parasite viability—provides a powerful framework for the drug development pipeline. The application of modern staining techniques like MitoTracker, alongside traditional reproductive assays and sophisticated pharmacodynamic modeling, allows researchers to confidently distinguish merely damaged parasites from those that are irreversibly non-viable. This correlative strategy accelerates the identification of truly parasiticidal compounds, contributing directly to the global effort to combat fasciolosis and other debilitating helminth infections.
Optimizing Snail Control Strategies to Disrupt the Life Cycle
The liver fluke, Fasciola hepatica, represents a significant global parasitic threat with substantial economic and public health impacts. As a food-borne trematode, it causes fasciolosis, a zoonotic disease classified by the World Health Organization as a neglected tropical disease, affecting an estimated 17-180 million people globally and incurring livestock production losses estimated at US$3 billion annually [23] [78] [79]. The parasite's transmission is entirely dependent on lymnaeid freshwater snails, which serve as obligatory intermediate hosts in its complex life cycle [11]. The strategic importance of snail control stems from this absolute dependence, making it a critical intervention point for disrupting transmission.
Recent research has expanded our understanding of F. hepatica host range, with studies confirming reindeer (Rangifer tarandus) as competent definitive hosts, potentially extending the parasite's range into arctic regions [23]. This discovery, coupled with the identification of novel egg morphological features (including an previously unreported appendage on the anti-opercular pole), highlights the ongoing evolution of our understanding of fasciolosis epidemiology [23]. The geographical distribution of F. hepatica continues to expand, facilitated by the adaptability of lymnaeid snails to diverse environmental conditions and the movement of infected animals [78]. In Argentina, for instance, spatial analysis has revealed heterogeneous prevalence patterns in sheep flocks, with altitude positively associated with prevalence and distance to water sources negatively associated, underscoring the importance of local environmental conditions in transmission dynamics [78].
The fundamental life cycle of F. hepatica begins when embryonated eggs passed in feces of infected mammals hatch into miracidia in freshwater, which then penetrate snail intermediate hosts. Inside the snail, the parasite undergoes several developmental stages (sporocysts, rediae, and cercariae). Cercariae are released from the snail and encyst as metacercariae on aquatic vegetation. Mammals, including humans, become infected by ingesting metacercariae-contaminated plants or water [80] [11]. The entire intramolluscan phase spans several weeks, providing a crucial window for intervention. Without the snail host, the life cycle cannot continue, making snail control an essential component of integrated fasciolosis management programs.
Snail Intermediate Host Ecology and Identification
Key Lymnaeid Species and Their Distribution
Fasciola hepatica development occurs in specific air-breathing freshwater snails from the family Lymnaeidae. Although several lymnaeid species are susceptible to infection, the parasite develops completely only in one or two major species on each continent [11]. The principal intermediate host in Europe, parts of Asia, Africa, and South America is Galba truncatula. In Central and South America, the most common intermediate hosts include Lymnaea viator, L. neotropica, Pseudosuccinea columella, and L. cubensis [11]. Other lymnaeid snails may be naturally or experimentally infected but play a lesser role in transmission. Understanding the specific snail species present in an endemic area is fundamental to designing effective control strategies, as habitat preferences and population dynamics vary among species.
These snail species exhibit particular ecological preferences, typically inhabiting shallow waters in various freshwater bodies including ponds, lakes, rivers, irrigation canals, and temporary water bodies [81]. Artificial water bodies such as irrigation canals and dams provide particularly suitable habitats that, when combined with intense human-water contact, can significantly enhance disease transmission. The distribution of infected snails is often highly focal, reflecting the localized nature of definitive host contamination and specific environmental conditions suitable for both snails and parasite development [81].
Population Dynamics and Environmental Influences
Lymnaeid snails are hermaphroditic with high reproductive capacity under favorable conditions. Their eggs are deposited in masses on aquatic vegetation or solid substrates, hatching within 7-10 days under optimal temperature conditions [81]. Snails reach sexual maturity in 3-15 weeks, with population densities subject to marked seasonal fluctuations driven primarily by rainfall and temperature variations. These fluctuations directly impact transmission dynamics, creating seasonal patterns of fasciolosis risk.
The population dynamics of intermediate host snails are influenced by numerous environmental factors, including water chemistry, temperature, vegetation type and density, and hydrological patterns. Snail populations often thrive in habitats with abundant aquatic macrophytes, which provide food, shelter from predators, and substrates for egg deposition [81]. In agricultural landscapes, irrigation systems can create ideal conditions for snail proliferation, especially when management practices result in consistent water levels and minimal disturbance. Understanding these ecological determinants is essential for predicting high-risk areas and timing control interventions effectively.
Table 1: Major Intermediate Snail Hosts of Fasciola hepatica and Their Geographical Distribution
| Snail Species | Primary Geographical Distribution | Preferred Habitat Characteristics |
|---|---|---|
| Galba truncatula | Europe, parts of Asia, Africa, and South America | Shallow, temporary water bodies, irrigation ditches |
| Lymnaea viator | Central and South America | Ponds, slow-moving streams, agricultural wetlands |
| Lymnaea neotropica | Central and South America | Irrigation canals, natural wetlands |
| Pseudosuccinea columella | Central and South America, widely introduced | Various freshwater habitats, highly adaptable |
| Lymnaea cubensis | Central and South America, Caribbean | Rivers, streams, irrigation systems |
Chemical Molluscicide Strategies
Synthetic Molluscicides: Efficacy and Applications
Chemical molluscicides represent the most immediately effective intervention for rapid snail population reduction in fasciolosis control programs. These compounds are particularly valuable in targeted, focal applications where transmission is known to occur. The ideal molluscicide should demonstrate high efficacy against target snail species, low environmental persistence, minimal toxicity to non-target organisms, and practical application feasibility. The historical development of molluscicides has progressed from relatively non-specific inorganic compounds to more targeted organic formulations.
Copper sulfate (CuSO₄) was among the earliest compounds used for snail control, achieving 100% mortality of Bulinus snails within two weeks at concentrations of 0.25 ppm in Egyptian trials [82]. However, its lack of specificity and toxicity to aquatic organisms limited its utility. Similarly, sodium pentachlorophenolate (NaPCP) demonstrated high efficacy (LC₅₀ of 0.54 ppm, LC₁₀₀ of 2.0 ppm at 48 hours) in immersion experiments but presented significant environmental and toxicological concerns, including potential teratogenicity and carcinogenicity [82]. More contemporary compounds include N-bromoacetamide, which exhibits excellent molluscicidal activity (LC₅₀ of 0.64 ppm, LC₉₀ of 1.0 ppm at 24 hours) with lower environmental impact and non-mutagenic properties [82]. Field applications of N-bromoacetamide at 1 g/m² achieved over 80% snail mortality after seven days in spraying tests.
Nicotinanilide represents another effective synthetic molluscicide, demonstrating LC₅₀ values of 0.23 ppm for immature snails, 0.77 ppm for young mature snails, and 0.59 ppm for adult snails at 24 hours exposure [82]. In field immersion experiments, nicotinanilide achieved 95% snail mortality at 1-2 ppm within three days. Although considered relatively safe for humans, animals, fish, and plants, its strong dermal irritation properties and relatively high cost have limited widespread adoption. The organic phosphorous pesticide Dipterex has also shown promise, causing 96% snail mortality at 10 ppm within three days, inducing paralysis of the snail head and foot preceding death [82].
Table 2: Efficacy Profiles of Synthetic Molluscicides Against Intermediate Host Snails
| Molluscicide | Chemical Class | LC₅₀ (ppm) | LC₉₀/LC₁₀₀ (ppm) | Key Limitations |
|---|---|---|---|---|
| Copper sulfate | Inorganic salt | Not specified | 0.25 (100% in 2 weeks) | Broad toxicity to aquatic life |
| Sodium pentachlorophenolate (NaPCP) | Chlorinated phenol | 0.54 (48 h) | 2.0 (48 h, LC₁₀₀) | Teratogenicity, carcinogenicity concerns |
| N-bromoacetamide | Haloacetamide | 0.64 (24 h) | 1.0 (24 h, LC₉₀) | Limited egg toxicity |
| Nicotinanilide | Anilide | 0.23-0.77 (24 h, varies with snail age) | 1-2 ppm (field LC₉₅) | Skin irritation, high cost |
| Calcium cyanamide | Cyanamide | 80 ppm (100% in 2 days, immersion) | 30 g/m² (100% in 1 day, spray) | Requires higher concentrations |
| Dipterex | Organophosphate | Not specified | 10 ppm (96% in 3 days) | Broad-spectrum toxicity |
Plant-Derived Molluscicides and Sustainable Alternatives
Botanical molluscicides offer potentially sustainable alternatives to synthetic compounds, with generally lower environmental persistence and reduced toxicity to non-target organisms. Among the most extensively studied plant-derived molluscicides is Ambrosia maritima, which has demonstrated significant field efficacy against snail intermediate hosts. Applications of 400 mg/L of dry A. maritima leaves in creeks of the Lampsar river in Senegal resulted in 54-56% reduction of Bulinus species two weeks post-treatment [83]. Similarly, treatment of a closed irrigation canal with 150-300 mg/L concentrations caused up to 77% reduction in Biomphalaria pfeifferi density after two weeks [83].
The primary active compounds in A. maritima include the sesquiterpene lactones damsin and ambrosin, which have shown molluscicidal properties in laboratory assays [83]. However, field applications have demonstrated a significant limitation: rapid population recovery following treatment, necessitating repeated applications for sustained control. Additionally, the substantial quantities of plant material required for effective snail reduction (often exceeding 150 mg/L) present logistical challenges for large-scale implementation [83]. Despite these limitations, botanical molluscicides remain promising for targeted, community-based control programs in endemic areas where synthetic compounds are unavailable or environmentally undesirable.
Research continues to identify and characterize novel plant-derived molluscicides with improved efficacy profiles. Endemic plants from fasciolosis-endemic regions are increasingly screened for molluscicidal properties, with several species showing promise in preliminary laboratory trials. The development of standardized extraction methods, compound isolation techniques, and formulation strategies aims to enhance the potency and stability of botanical molluscicides while reducing the biomass requirements for effective application.
Non-Chemical Control Approaches
Environmental Management and Habitat Modification
Environmental management represents a sustainable, long-term approach to snail control by modifying or eliminating the habitats essential for snail survival and reproduction. These strategies focus on altering the physical environment to make it less suitable for lymnaeid snails, thereby reducing population densities without chemical interventions. Effective environmental management requires detailed understanding of local snail species' ecological requirements and careful planning to avoid unintended consequences on ecosystem function.
Key environmental modifications include drainage of stagnant water bodies, regulation of water levels in irrigation systems, clearance of aquatic vegetation, and improvement of water flow rates in canals. Snails of the Lymnaeidae family typically thrive in shallow, stagnant, or slow-moving waters with abundant vegetation, which provides food, shelter, and oviposition sites [81]. Removing vegetation eliminates both food sources and protective cover, increasing snail susceptibility to predators and environmental stressors. Similarly, increasing water flow rates in irrigation canals to exceed 0.14 m/s creates unfavorable conditions for most lymnaeid species [81]. Proper drainage of agricultural fields, particularly in areas with high seasonal rainfall, prevents the formation of temporary water bodies that can serve as seasonal snail habitats.
The implementation of environmental management strategies must be tailored to local conditions and integrated with agricultural practices. In northwestern Córdoba, Argentina, spatial analysis revealed that altitude was positively associated with F. hepatica prevalence, while greater distance to water sources was negatively associated, highlighting the importance of water proximity in transmission dynamics [78]. This suggests that strategic management of water access points for livestock, coupled with environmental modifications around permanent water sources, could significantly reduce transmission. Although environmental management requires substantial initial investment, it offers permanent solutions to snail problems when properly implemented and maintained.
Biological Control Agents
Biological control utilizes natural predators, competitors, or pathogens of intermediate host snails to reduce population densities. This approach offers an environmentally friendly alternative to chemical control, with potential for self-sustaining population regulation. Various aquatic organisms have been investigated for their snail control potential, including aquatic insects, crustaceans, fish, amphibians, and trematodes.
Potential biocontrol agents include aquatic birds, turtles, fish, crayfish, and insects that prey directly on snails [82]. Competitor snails, particularly those of the Thiaridae family, have also been employed to displace lymnaeid species through competition for resources [82]. Additionally, various microorganisms including trematodes, leeches, nematodes, rotifers, and ostracods can attack or devour snails, though their practical application remains largely experimental [82]. The implementation of biological control requires careful consideration of potential ecological impacts, particularly when introducing non-native species, which may disrupt local ecosystems.
Despite promising laboratory results, the field application of biological control for fasciolosis management has faced significant challenges. Many potential agents show variable efficacy under natural conditions, and their establishment in new environments can be unpredictable. Furthermore, the specificity of biological control agents varies considerably, with some posing risks to non-target native species. Current research focuses on identifying highly specific pathogens or parasites of lymnaeid snails that minimize non-target effects, as well as developing integrated approaches that combine biological control with complementary strategies.
Experimental Protocols for Molluscicide Evaluation
Laboratory Bioassays for Efficacy Screening
Standardized laboratory bioassays are essential for initial screening of potential molluscicidal compounds and establishing baseline efficacy parameters. The World Health Organization has established protocols for laboratory evaluation of molluscicides that facilitate comparison across studies. These assays typically involve exposing snails of known age and size to varying concentrations of the test compound under controlled conditions and monitoring mortality over specific time intervals.
A standard laboratory bioassay protocol begins with collection and acclimation of laboratory-reared or field-collected snails for at least 48 hours before testing. Snails should be of consistent size and age, preferably young adults representing the most resilient life stage. Test solutions are prepared using dechlorinated water at concentrations ranging from sublethal to fully lethal, typically in geometric progression. For each concentration, ten snails are placed in containers with one liter of test solution, with appropriate controls containing only dechlorinated water. Snails are maintained under standard conditions (temperature 22-25°C, pH 7.0-8.0, natural or simulated daylight cycles) without feeding during the experiment. Mortality assessments are conducted at 24, 48, and 72 hours, with death defined as lack of movement after gentle prodding and failure to respond when placed in warm water (30°C). The LC₅₀, LC₉₀, and LC₁₀₀ values are calculated using appropriate statistical methods, such as probit analysis or linear regression after logarithmic transformation of concentrations [82].
Additional specialized assays include evaluation of ovicidal effects by exposing egg masses to test compounds and monitoring hatching rates, assessment of sublethal effects on snail fecundity and growth, and examination of behavioral responses such as avoidance behavior. These comprehensive evaluations provide crucial data beyond simple mortality metrics, informing predictions of field efficacy and potential impacts on snail population dynamics.
Field Trial Methodologies and Evaluation
Field trials are essential for validating laboratory findings and assessing molluscicide performance under natural conditions. The complex interplay of environmental factors in field settings can significantly influence molluscicide efficacy, including water chemistry, temperature, organic matter content, and hydrological patterns. Well-designed field trials incorporate appropriate replication, controls, and monitoring protocols to generate reliable, actionable data.
A standardized field trial protocol for molluscicide evaluation begins with selection of representative natural water bodies or simulated field enclosures with established snail populations. Pre-treatment sampling establishes baseline population densities using standardized techniques such as timed searches, quadrat sampling, or trapping. Test compounds are applied at predetermined concentrations based on laboratory LC₉₀ values, typically using calibrated spraying equipment for terrestrial habitats or calculated dilution methods for aquatic environments. Applications should be conducted during periods of stable weather conditions to minimize confounding effects of rainfall or extreme temperatures. Post-treatment monitoring includes immediate assessment (24-48 hours) to detect rapid knockdown effects, followed by weekly sampling for 4-8 weeks to evaluate prolonged control and population recovery rates [83].
In addition to snail population assessments, comprehensive field trials should monitor potential non-target effects on aquatic invertebrates, fish, amphibians, and vegetation. Water quality parameters (temperature, pH, dissolved oxygen, turbidity) should be recorded regularly, as these factors may influence both molluscicide efficacy and ecological impact. The experimental design should include replicated treatment and control plots with sufficient separation to prevent cross-contamination. Data analysis focuses not only on significance of population reduction but also on duration of control and rate of population recovery, which are critical determinants of operational feasibility and cost-effectiveness [83].
The Scientist's Toolkit: Essential Research Reagents and Methodologies
Key Research Reagent Solutions for Snail Control Studies
Table 3: Essential Research Reagents and Materials for Snail Control Studies
| Reagent/Material | Specification/Application | Research Function |
|---|---|---|
| Laboratory snail colonies | Species-specific (G. truncatula, L. viator, etc.), age-graded | Standardized bioassays, life cycle studies |
| Reference molluscicides | Niclosamide, N-bromoacetamide, copper sulfate | Positive controls, efficacy comparisons |
| Plant extraction kits | Methanol, ethanol, hexane, aqueous extracts | Botanical molluscicide preparation |
| Water quality test kits | pH, dissolved oxygen, hardness, temperature | Environmental parameter standardization |
| Molecular identification kits | DNA extraction, PCR primers for snail species | Snail species confirmation |
| Histopathology reagents | Fixatives, stains, embedding media | Molluscicide effect characterization |
Advanced Molecular Tools for Snail-Parasite Interactions
Cutting-edge research on snail control mechanisms increasingly utilizes molecular tools to understand fundamental snail-parasite interactions and identify novel targets for intervention. Genomic and proteomic approaches are revealing key aspects of the compatibility between F. hepatica and its intermediate hosts, potentially identifying susceptibility factors that could be exploited for targeted control strategies.
Recent investigations have characterized specific F. hepatica proteins involved in host interaction, including enolase (Fhenolase) and the 16.5-kDa tegument-associated protein (Fh16.5TP), both found in extracellular vesicles (FhEVs) secreted by the parasite [79]. Recombinant versions of these proteins (r-Fhenolase and r-Fh16.5TP) have been produced using cDNA amplification from adult worms cloned into pQE30-32 expression vectors with C-terminal His-tags, followed by expression in Escherichia coli M15 cells [79]. These recombinant proteins enable investigation of parasite-derived immunomodulators that may influence snail host responses. Proteomic analysis of cells treated with r-Fhenolase has revealed distinct alterations in proteins related to fibrotic and inflammatory pathways, suggesting potential roles in modulating host cellular environments [79].
Additional molecular tools include RNA interference techniques for functional gene validation in snails, transcriptomic analyses to identify snail genes differentially expressed during parasite infection, and comparative genomics approaches to identify species-specific susceptibility factors. These advanced methodologies are transforming our understanding of the molecular dialogue between parasite and intermediate host, potentially leading to novel, highly specific control strategies that disrupt key interaction pathways without broad ecological impacts.
Integrated Control Frameworks and Implementation Strategies
Effective snail control for fasciolosis management requires integrated approaches that combine multiple intervention strategies tailored to local epidemiological and ecological conditions. The fundamental principle of integrated control is leveraging synergistic effects between interventions while minimizing limitations of individual approaches. Strategic implementation considers seasonal transmission dynamics, resource availability, and operational feasibility within specific ecological contexts.
The core components of integrated snail control include: (1) strategic chemical control using molluscicides applied focally at transmission hotspots during critical periods; (2) environmental management to reduce snail habitat availability and permanence; (3) biological control where effective, specific agents are available; and (4) complementary measures including diagnostic testing, targeted anthelmintic treatment, and community education. This integrated approach aligns with WHO recommendations for comprehensive parasitic disease control, emphasizing adaptation to local transmission patterns and resource constraints [81].
Implementation should be guided by rigorous spatial and temporal risk assessment, prioritizing areas where environmental factors favor high snail densities and parasite transmission. In the Córdoba province of Argentina, spatial analysis using Inverse Distance Weighted (IDW) interpolation and Geographically Weighted Regression (GWR) models revealed heterogeneous prevalence patterns, with higher concentrations in northern regions [78]. Such spatially explicit risk mapping enables precisely targeted interventions, maximizing impact while minimizing resource use. Seasonal application timing should precede peak transmission periods, disrupting the parasite life cycle before massive cercarial release occurs.
Successful implementation requires multidisciplinary collaboration among parasitologists, malacologists, ecologists, and public health professionals, coupled with community engagement to ensure sustainability. Regular monitoring of both snail populations and parasite prevalence in definitive hosts provides essential feedback for strategy refinement. The ultimate goal is establishing cost-effective, environmentally sustainable control programs that significantly reduce F. hepatica transmission through strategic disruption of the essential snail intermediate host phase.
Validating Morphological Findings and Comparative Glycobiology for Novel Interventions
Within the broader research on the life cycle and egg morphology of Fasciola hepatica, the establishment of precise and validated morphometric parameters for eggs represents a critical diagnostic and taxonomic endeavor. The accurate identification of F. hepatica eggs in fecal samples remains the gold standard for diagnosing fascioliasis in both humans and animals, a disease affecting millions globally and causing substantial economic losses in livestock production [23] [1]. This technical guide provides an in-depth framework for the morphometric validation of F. hepatica eggs, consolidating traditional morphological criteria with recent discoveries and advanced measurement methodologies. The emergence of reports concerning atypical egg morphology, including the presence of a previously unreported abopercular appendage, underscores the necessity for refined and updated diagnostic parameters [23] [5]. This guide is designed to equip researchers, scientists, and drug development professionals with the standardized protocols and reference data required to ensure accuracy and consistency in species identification, differential diagnosis, and subsequent research interventions.
Background and Significance
The Role of Egg Morphology in the F. hepatica Life Cycle
The life cycle of Fasciola hepatica is complex, involving mammalian definitive hosts and amphibious snail intermediate hosts. Adult flukes residing in the bile ducts produce eggs that are evacuated in the host's feces [1]. Upon reaching freshwater, these eggs embryonate over approximately two weeks, releasing miracidia that must infect a suitable snail to continue development [1] [20]. The morphological consistency of the egg stage is therefore paramount for the parasite's survival and transmission. Recent investigations into the life cycle have revealed controversial aspects, particularly concerning the pathway and timing of young flukes entering the biliary system [84]. Despite these ongoing discussions, the egg stage remains a constant and diagnostically accessible point in the cycle, making its accurate identification non-negotiable for disease surveillance and control.
Diagnostic Challenges and the Need for Standardization
Coprological examination, while foundational, presents significant challenges. Eggs of F. hepatica can be difficult to distinguish from those of related trematodes, such as Fasciolopsis buski and some Echinostoma species, due to morphologic overlap [1]. Furthermore, the potential for spurious passage from the consumption of contaminated liver necessitates careful diagnosis [1]. The recent discovery that F. hepatica eggs can possess an abopercular appendage complicates traditional diagnostics, as this feature was previously considered a key distinguishing characteristic of the genus Fascioloides [23]. This finding indicates that the appendage alone is not a valid criterion for differentiation, demanding a more nuanced, morphometrically robust approach to identification [23]. Consequently, establishing a validated set of size and shape parameters is essential to overcome these diagnostic ambiguities and improve the reliability of epidemiological data and treatment outcomes.
Established Morphometric Parameters
The standard morphometric characteristics of F. hepatica eggs have been documented by global health authorities and through extensive field research. These parameters provide the baseline for identification.
Core Size and Shape Characteristics
The eggs of Fasciola spp. are broadly ellipsoidal and operculated [1]. The abopercular end (the pole opposite the operculum) often has a roughened or irregular area, which is a useful distinguishing feature [1]. It is now established that this area can sometimes manifest as a distinct appendage, a trait observed in eggs derived from both reindeer feces and adult fluke uteri [23]. The size ranges reported by the CDC are consolidated in the table below with data from recent studies.
Table 1: Standard Morphometric Parameters for F. hepatica Eggs
| Parameter | Standard Range (CDC) | Notes and Additional Observations |
|---|---|---|
| Length | 130 – 150 µm [1] | A primary measurement for specific identification. |
| Width | 60 – 90 µm [1] | Used in conjunction with length for shape determination. |
| Shape Index (L/W Ratio) | ~1.7 – 2.5 (calculated) | Ellipsoidal form. Ratio can vary with host species [85]. |
| Abopercular End | Roughened, irregular area [1] | May present with a visible appendage, previously unreported [23]. |
Host-Induced Morphometric Variations
It is critical to recognize that the definitive host species can exert a significant influence on the morphometry of F. hepatica eggs. Research from the hyperendemic northern Bolivian Altiplano demonstrated that egg size displays characteristic morphometric traits in each definitive host species [85]. For instance, statistical analyses have confirmed significant differences in egg dimensions recovered from sheep, cattle, pigs, and donkeys. This host-specific variation underscores the importance of building comprehensive reference databases that account for the parasite's host range, which includes, as recently confirmed, reindeer [23]. Notably, these host-induced morphological influences do not persist when the parasite is passaged through a rodent definitive host model, suggesting a direct and adaptable host-parasite interaction [85].
Experimental Protocols for Morphometric Validation
Validating morphometric parameters requires a structured approach from sample collection through to data analysis. The following protocols are recommended for rigorous validation.
Sample Collection and Processing
Fecal Sampling: Freshly excreted fecal samples should be collected and stored at +4°C for short-term transport. For prolonged storage before analysis, freezing at -18°C is acceptable [23]. Detailed labeling of host species, location, and date is essential.
Sedimentation Technique: The sequential washing sedimentation technique is a standard method for concentrating trematode eggs.
- Homogenize a 3-5 gram fecal sample with water (e.g., 50 ml for 3 g of feces).
- Filter the mixture through a synthetic fiber mesh (e.g., 1 mm diameter) to remove large debris.
- Settle the filtered suspension for 5 minutes until a sediment forms.
- Decant the supernatant carefully.
- Repeat the resuspension, settling, and decanting steps with clean water until the supernatant is clear.
- Examine the final sediment under a microscope [23].
Adult Fluke Egg Isolation: To obtain eggs directly from the source, adult worms can be recovered from the bile ducts of infected livers. Eggs are released from the uteri of adult flukes by dissecting around the uterine area and collecting the eggs into a water suspension for examination [23].
Imaging and Measurement Techniques
Microscopy and Imaging: Eggs should be placed on a slide in a water suspension and covered with a cover slip (e.g., 24x24 mm). Light micrographs should be captured using 40x and 100x objective lenses to ensure sufficient detail for morphometry [23].
Computer Image Analysis (CIAS): A Computer Image Analysis System is the recommended tool for high-precision morphometry. This system typically involves a color video camera connected to a microscope, which feeds images into specialized software [48] [85]. The use of CIAS minimizes observer bias and allows for the measurement of multiple parameters, including linear dimensions, areas, and perimeters.
Calibration and Measurement: The imaging system must be calibrated using a stage micrometer. Measurements are then performed on digital images using software such as Fiji/ImageJ [23]. Key measurements include:
- Length: The longest axis of the egg.
- Width: The broadest axis perpendicular to the length.
- Shape Descriptors: Calculation of the Length-to-Width ratio (Shape Index).
- Abopercular Features: Documentation of any thickening, irregularity, or appendage presence.
Table 2: Essential Research Reagent Solutions and Materials for Morphometric Analysis
| Item | Function / Application | Example / Specification |
|---|---|---|
| Sedimentation Kit | Concentration of eggs from fecal samples. | Mortar and pestle, filters (1mm mesh), settling cups [23]. |
| Microscopy Setup | Visualization and image capture of eggs. | Microscope with 40x & 100x objectives, calibrated digital camera [23]. |
| Image Analysis Software | Precise measurement of morphometric parameters. | Fiji/ImageJ, ZBrush, Blender for 3D modeling [23]. |
| DNA Extraction Kit | Genetic validation of egg identity. | QIAamp DNA Micro Kit or equivalent [23]. |
| PCR Reagents | Amplification of genetic markers for species confirmation. | Primers for ITS1, 5.8S, ITS2 rDNA region (e.g., BD1, BD2) [23]. |
Data Analysis and Validation Workflow
The workflow for morphometric validation integrates traditional microscopy with modern genetic techniques to ensure accuracy, especially when encountering atypical specimens.
Diagram 1: Morphometric Validation Workflow. This diagram outlines the integrated process for validating F. hepatica egg parameters, combining morphological and genetic techniques to ensure diagnostic accuracy, particularly for atypical specimens.
Advanced Modeling and Future Directions
Three-Dimensional Modeling and Allometric Analysis
To overcome the limitations of two-dimensional microscopy, researchers are now employing three-dimensional models to demonstrate egg features more comprehensively. These models are created using software like Blender and ZBrush, with original light and scanning electron microscopy (SEM) images serving as references [23]. These 3D reconstructions provide an unparalleled view of spatial relationships of features like the appendage, operculum, and shell texture.
Furthermore, for a more dynamic understanding of form, allometric analysis can be applied. Allometric models describe the relationship between the growth rates of different body parts. The model (y2m−y2)/y2 = c [(y1m−y1)/y1]^b (where y1 and y2 are biometric variables like body area and length, and y1m, y2m are their maximum values) has proven useful in analyzing the phenotypic plasticity of F. hepatica adults [48] [85]. While more commonly used for adult flukes, this approach holds promise for understanding variations in egg proportions across different host species and environments.
Integration with Genetic and Environmental Data
Future morphometric validation efforts will be increasingly intertwined with genomic tools. The genetic characterization of F. hepatica using the Internal Transcribed Spacer (ITS) rDNA region is a standard method for confirming species identity and exploring population structures [23] [86]. This is crucial for validating the identity of eggs with unusual morphologies.
Additionally, environmental factors such as temperature play a critical role in the development and potentially the morphology of fluke stages. Egg development and hatching are inhibited below 10°C and are optimal between 20°C and 30°C [6]. While direct links to egg size are less studied, the entire life cycle is temperature-dependent, and climate change may influence parasite phenotypes and distribution, necessitating ongoing morphometric surveillance.
Diagram 2: Factors Influencing Egg Phenotype. The final morphology of a F. hepatica egg is an interactive product of the host species it develops in, the genetic makeup of the parasite, and the external environmental conditions.
The morphometric validation of Fasciola hepatica eggs is a dynamic and critical field that directly impacts diagnostic accuracy, epidemiological understanding, and control of a globally significant parasitic disease. This guide has established that while standard parameters (130-150 µm by 60-90 µm) provide a essential foundation, the validation process must account for newly recognized features like the abopercular appendage and acknowledge the significant influence of the definitive host species on egg dimensions. The implementation of standardized protocols, incorporating Computer Image Analysis Systems (CIAS) and backed by genetic validation, is paramount for achieving precision. As research continues, the integration of advanced 3D modeling, allometric analyses, and climate impact studies will further refine our understanding of F. hepatica egg morphology, ensuring that diagnostic criteria evolve in step with scientific discovery. For researchers and drug development professionals, adherence to these rigorous validation standards is the cornerstone of reliable data and effective intervention strategies against fascioliasis.
Fasciolosis, caused by the trematode Fasciola hepatica, represents a significant global zoonotic and veterinary challenge. Traditionally, the parasite's definitive host range has been centered on domestic ruminants, with wildlife reservoirs playing a crucial but not fully elucidated role in transmission dynamics. This whitepaper details the seminal genetic and morphological validation of the reindeer (Rangifer tarandus) as a competent definitive host for F. hepatica. Framed within a broader thesis on the parasite's life cycle and egg morphology, this research provides incontrovertible evidence through combined microscopic analysis of novel egg appendages and sequence confirmation of the internal transcribed spacer (ITS) rDNA region. The confirmation of a viable host at a high-latitude location also prompts a re-evaluation of the global distribution and transmission potential of fasciolosis, with direct implications for surveillance strategies, drug development, and wildlife management in a changing climate.
Fasciola hepatica exhibits a complex life cycle involving an intermediate snail host and a mammalian definitive host, where it matures and reproduces. Understanding its host range is critical for epidemiology and control. The parasite's eggs possess distinct morphological traits that are central to coprological diagnosis, which remains the gold standard in many settings [23] [87]. Recent findings have challenged the validity of certain egg characteristics as definitive criteria for distinguishing between Fasciola species or even genera, revealing a more complex picture of its biology and host interactions [23]. This technical guide documents the experimental and analytical protocols that led to the confirmation of reindeer as a definitive host, a finding that expands our understanding of host-parasite relationships and has profound implications for disease control in arctic and subarctic ecosystems.
Experimental Validation: Methodologies and Workflows
The genetic validation of reindeer as a definitive host was achieved through a multi-faceted approach, combining field parasitology, advanced microscopy, and molecular genetic techniques. The following sections provide a detailed breakdown of the core methodologies employed.
Sample Collection and Processing
Background and Sampling Protocol: The investigation was initiated following the discovery of Fasciola-like eggs in fecal samples from a zoo reindeer in Leningrad Oblast, Russia (59.594384 N, 30.283104 E) [23]. To confirm the finding, a follow-up sampling strategy was implemented.
- Fecal Sampling: Freshly excreted feces were collected from individual reindeer, placed in labeled containers, stored at +4 °C, and delivered to the laboratory within two days [23].
- Adult Fluke Sampling: For comparative morphology, adult F. hepatica were obtained from the bile ducts of a slaughtered bull (Tula Oblast, Russia) as a reference control [23].
Coprological Examination: Fecal samples were processed according to established national standards (GOST R 54627–2011) [23].
- Homogenization: A 3-gram sample of feces was homogenized with a mortar and pestle.
- Sedimentation: The homogenate was mixed with 50 mL of water, carefully filtered through a 1 mm mesh, and allowed to settle for 5 minutes.
- Washing: The supernatant was decanted, the sediment resuspended in fresh water, and the settling process repeated until the supernatant was clear.
- Microscopy: The final sediment was examined under a light microscope for the presence of trematode eggs [23].
Morphological Analysis of Eggs
Imaging and Morphometry: Eggs derived from both reindeer feces and adult fluke uteri were prepared on slides in tap water and covered with cover slips [23].
- Light Microscopy (LM): Micrographs were taken using 40x and 100x objective lenses.
- Morphometric Analysis: Measurements of egg dimensions were performed on the acquired micrographs using Fiji/ImageJ software, calibrated with a stage micrometer [23].
- Three-Dimensional Modeling: To accurately represent the observed structures, 3D models of F. hepatica eggs were created using Blender and ZBrush software, based on original LM photographs and published scanning electron microscopy (SEM) images [23].
Genetic Analysis for Species Confirmation
DNA Extraction and Amplification: To conclusively identify the parasite species, genetic analysis was performed on the eggs recovered from reindeer feces [23].
- Sample Preparation: Eggs containing live miracidia were individually collected and stored in tubes at -70 °C to rupture the egg shells.
- DNA Extraction: Genomic DNA was extracted from the pooled eggs using a commercial kit (QIAamp DNA Accessory Set, Micro kit) following the manufacturer's protocol [23].
- PCR Amplification: The Internal Transcribed Spacer (ITS) region of the ribosomal DNA (including ITS1, 5.8S, and ITS2) was amplified via polymerase chain reaction (PCR) using the universal trematode primers BD1 and BD2 [23].
- Sequencing and Analysis: The amplified PCR product was sequenced, and the resulting sequence was analyzed and compared to reference sequences in genetic databases to confirm the identity of the parasite as Fasciola hepatica [23].
The following workflow diagram illustrates the integrated process from sample collection to final validation:
Key Findings and Data Synthesis
Morphological Identification of Eggs
The analysis confirmed that the eggs found in reindeer feces were those of Fasciola hepatica, but with a critical novel observation: the presence of an appendage on the abopercular pole.
This finding is diagnostically significant, as an appendage was previously considered a potential key differentiator from eggs of Fascioloides magna [23]. The study demonstrated that this feature can also be present in F. hepatica eggs derived directly from adult worm uteri, indicating it is a genuine morphological trait and not an artifact of fecal passage [23].
Table 1: Key Morphological and Developmental Characteristics of Fasciola hepatica Eggs
| Characteristic | Description | Significance |
|---|---|---|
| Standard Morphology | Broadly ellipsoidal, operculated, 130-150 µm by 60-90 µm [1]. | Basis for coprological diagnosis worldwide. |
| Novel Appendage | Observed on the abopercular pole in eggs from both reindeer feces and adult fluke uteri [23]. | Challenges existing differential diagnostic criteria; requires updating of diagnostic keys. |
| Development Period | Miracidia develop within 13-15 days at 26±1°C [35]. | Informs environmental modeling and transmission risk assessment. |
| Miracidial Size | Approximately 136 µm by 74 µm [35]. | Useful for species identification and biological studies. |
Genetic Confirmation of Parasite Identity
The genetic analysis provided definitive proof of the parasite's identity. Sequencing of the ITS rDNA region from eggs isolated from reindeer feces yielded a sequence that showed 100% identity to Fasciola hepatica reference sequences, thus genetically validating the reindeer as a definitive host [23].
Geographic and Epidemiological Significance
The second case in this study involved the discovery of a single F. hepatica egg in a wild reindeer from the Novaya Zemlya archipelago in the Russian Arctic (76.784423 N, 68.823853 E) [23]. This finding is highly significant as it suggests that the Novaya Zemlya archipelago might be the northernmost location where fasciolosis has been documented, pushing the known geographic boundaries of the parasite's transmission and highlighting the potential for host range expansion into arctic ungulate populations [23].
Table 2: Summary of Host Validation and Epidemiological Data
| Parameter | Case 1 (Zoo Reindeer) | Case 2 (Wild Reindeer) |
|---|---|---|
| Host Status | Rangifer tarandus (Zoo) | Rangifer tarandus tarandus (Wild, Novaya Zemlya) |
| Sample Type | Feces | Feces |
| Egg Identification | Light Microscopy & Morphometrics | Light Microscopy |
| Genetic Validation | ITS rDNA Sequencing | Not Reported |
| Key Significance | Definitive genetic proof of reindeer as a competent definitive host. | Potential northernmost limit of fasciolosis, indicating geographic expansion. |
The Scientist's Toolkit: Essential Research Reagents
The following table catalogs key reagents and materials utilized in the featured research, providing a resource for scientists aiming to replicate or build upon this work.
Table 3: Key Research Reagents and Materials for Fasciolid Study
| Reagent / Material | Function / Application | Specific Example / Note |
|---|---|---|
| Coprological Staining Dyes | Enhances contrast for microscopic identification of eggs and other parasites in fecal samples. | Various stains used in standard protocols (e.g., methylene blue) [23]. |
| DNA Extraction Kit | Purifies genomic DNA from parasite stages (eggs, miracidia, adult tissue) for molecular analysis. | QIAamp DNA Accessory Set, Micro kit (Qiagen) [23]. |
| PCR Primers (ITS Region) | Amplifies the Internal Transcribed Spacer region for genetic species identification and phylogenetics. | Universal trematode primers BD1 and BD2 [23]. |
| Excretory/Secretory (ES) Antigens | Native antigens used as a target in serological assays (e.g., ELISA) for immunodiagnosis. | FhES antigens for detecting host antibodies [87]. |
| Recombinant Antigens (e.g., FhrAPS) | Defined, pure antigens used in serological tests to improve specificity and standardization. | 2.9 kDa recombinant protein used in ELISA for diagnosis [87]. |
| Monoclonal Antibodies (e.g., MM3, BIO K 211) | Used in capture ELISA for detecting coproantigens or serum antibodies; high specificity. | BIO K 211 used in a commercial kit for serodiagnosis in deer [87]. |
| Polymerase Chain Reaction (PCR) Reagents | Enzymes, buffers, and nucleotides for amplifying specific DNA targets from minute quantities. | Standard PCR master mix for amplifying Pepck, COI, or ITS genes [88] [89]. |
| Microsatellite Markers | Highly polymorphic genetic markers for population genetics, transmission tracking, and drug resistance studies. | Panels of 6+ polymorphic loci used for population structure analysis [88] [90]. |
Discussion and Implications for Research and Control
The genetic validation of reindeer as a definitive host for Fasciola hepatica necessitates a revision of the parasite's known host spectrum. This finding, coupled with the novel egg morphology, underscores the dynamic nature of host-parasite interactions and the potential for wildlife to act as reservoirs in new ecosystems. The discovery in the high Arctic is particularly alarming, as it may signal a range expansion facilitated by climate change, potentially bringing fasciolosis risk to previously unaffected regions and populations, both animal and human.
For the drug development community, understanding host range is critical for designing effective control strategies. The inclusion of wildlife like reindeer in the epidemiology of fasciolosis complicates control programs based solely on treating domestic livestock. It highlights the need for integrated pest management approaches that consider wildlife reservoirs and environmental measures. Furthermore, the high genetic diversity and gene flow observed in F. hepatica populations [88] [90] suggest that anthelmintic resistance genes, once they emerge, could spread rapidly across a landscape, even between domestic and wild hosts. This reinforces the urgency for developing new chemotherapeutic agents and non-chemical control methods.
Future research should focus on large-scale surveillance of wild ungulate populations using the combined morphological and genetic protocols outlined herein. Additionally, experimental infections could further elucidate the parasite's pathogenicity and egg production dynamics in reindeer. Finally, the potential impact of this expanded host range on the sustainability of current and future control interventions, including vaccines and novel drugs, must be thoroughly evaluated.
The zoonotic trematode Fasciola hepatica, commonly known as the liver fluke, represents a significant global threat to both human and animal health, with its ability to cause fasciolosis. A key factor in its successful establishment and long-term survival within the mammalian host is the dynamic syncytial tegument layer, surrounded by a dense coat of carbohydrate complexes known as the glycocalyx [91]. This glycocalyx serves as the primary physical interface for host-parasite interactions, facilitating critical functions such as nutrient absorption, sensory perception, and, most notably, sophisticated defense mechanisms against host immune responses [91]. The emergence of drug resistance in F. hepatica has underscored the urgent need for novel therapeutic strategies, bringing the glycocode—the specific language and structure of its glycans—to the forefront of parasitology research [91]. This review consolidates current knowledge on the glycosylation patterns of F. hepatica, with a specific focus on how glycans present on the tegument interface are decoded by the host immune system to facilitate parasite survival. Furthermore, this glycobiological perspective is framed within the broader context of the parasite's life cycle and egg morphology, which are fundamental to its dissemination and diagnosis.
TheFasciola hepaticaLife Cycle and Diagnostic Egg Morphology
A comprehensive understanding of F. hepatica's glycobiology is anchored in its complex life cycle and the morphology of its eggs, which are critical for diagnosis and understanding transmission.
Life Cycle Stages
The life cycle of Fasciola hepatica is an indirect process involving a definitive mammalian host and an intermediate snail host [1] [7] [46]. Table 1 summarizes the key stages.
Table 1: Life Cycle Stages of Fasciola hepatica
| Stage | Location/Host | Key Events and Morphology |
|---|---|---|
| Adult Fluke | Bile ducts of the liver in the definitive host [1] | Large, leaf-shaped, and flattened; can measure up to 30 mm by 15 mm; produces eggs [1]. |
| Egg | Passed in the host's feces [1] | Operculated, broadly ellipsoidal, measuring 130–150 µm long by 60–90 µm wide; embryonates in water [1]. |
| Miracidium | Freshwater, hatched from the egg [7] | Ciliated, free-swimming larva; must invade a suitable snail intermediate host within 24-30 hours or it dies [18]. |
| Intramolluscan Stages (Sporocysts & Rediae) | Tissues of the snail host [1] | Several developmental and multiplicative stages occur within the snail, culminating in the production of cercariae [1]. |
| Cercaria | Emerges from the snail into water [7] | Tadpole-like, swimming larval form [18]. |
| Metacercaria | Encysted on aquatic vegetation [1] | The highly resilient, infective stage for the definitive host [18]. Ingestion leads to infection [1]. |
In the definitive host, after metacercariae are ingested, they excyst in the duodenum. The juvenile flukes penetrate the intestinal wall, migrate through the peritoneal cavity and liver parenchyma—causing tissue damage—and finally reach the bile ducts, where they mature into adults and begin producing eggs [1] [7]. This maturation process typically takes 3–4 months in humans [1].
Egg Morphology and Host Influence
Egg morphology is a critical diagnostic feature. Recent research has revealed that eggs of F. hepatica may possess a previously unreported appendage on the abopercular pole (the end opposite the operculum) [23]. This finding is significant for differential diagnosis, as such appendages were previously considered a distinguishing feature of other flukes like Fascioloides magna [23].
Furthermore, the definitive host species can decisively influence the morphometric traits of F. hepatica eggs. Statistical analyses of egg size have shown characteristic morphometric patterns associated with different host species, such as sheep, cattle, and pigs [85]. However, these host-specific influences do not persist when the parasite is passaged through a rodent model, indicating a significant but non-genetic host effect on phenotypic expression [85].
Glycan-Mediated Immune Modulation: Mechanisms and Pathways
The glycans expressed on F. hepatica's tegument and released in its excretion/secretion products are not merely structural. They are decoded by the host's innate immune system, primarily through C-type lectin receptors (CLRs) on antigen-presenting cells, leading to a skewed immune response that favors parasite survival [92] [93] [94].
Key Glycan Structures and Immune Receptors
Research has identified several key glycan motifs and their corresponding receptors on immune cells. The Tn antigen (GalNAc-α-1-O-Ser/Thr), a simple mucin-type O-glycan, is expressed on F. hepatica glycoconjugates [93]. This structure is a prime ligand for the Macrophage Gal/GalNAc Lectin (MGL, also known as CD301), expressed on dendritic cells (DCs) and macrophages [93]. Furthermore, glycoconjugates containing mannose and fucose residues are recognized by another CLR, DC-SIGN (Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin, or CD209) [94]. Other described glycans include Galβ1-6Gal-terminating glycolipids and glucose residues [92].
Experimental Evidence and Signaling Pathways
The following diagram illustrates the general experimental workflow and the key immunomodulatory pathways triggered by F. hepatica glycans via CLRs on dendritic cells.
Diagram 1: Experimental Workflow and Immunomodulatory Outcomes of F. hepatica Glycan Studies. This workflow, derived from multiple studies [92] [93] [94], leads to the identification of CLRs as key mediators.
Key Experimental Findings:
- Induction of Regulatory Cytokines: When added to dendritic cells (DCs) along with a Toll-like receptor (TLR) ligand like LPS, F. hepatica glycoconjugates (FhTE) enhance the production of anti-inflammatory and regulatory cytokines, particularly IL-10 and IL-27p28, while suppressing pro-inflammatory cytokines like IFNγ [92] [94]. This effect is abrogated upon oxidation of glycans (FhmPox), proving the glycans are the active immunomodulatory agents [92] [94].
- Modulation of DC Maturation: The parasite induces a "semi-mature" phenotype in DCs, characterized by low surface expression of MHC class II and co-stimulatory molecules (CD40, CD86) and high secretion of IL-10 [92] [93]. These modulated DCs are poor at activating pro-inflammatory T-cells.
- T-cell Polarization: DCs conditioned by F. hepatica glycans promote the expansion of Th2 and regulatory T cells (Tregs) while suppressing Th1 polarization [93]. Furthermore, they can induce a state of anergy in T cells, rendering them unresponsive [94].
The specific signaling interactions between parasite glycans and host CLRs are detailed in the following pathway diagram.
Diagram 2: Glycan-CLR Signaling Leading to Immune Modulation. The recognition of specific glycan motifs by CLRs triggers intracellular signaling that alters the DC's maturation and cytokine profile, ultimately directing a non-protective T-cell response [93] [94].
- The Role of DC-SIGN: Blocking experiments with specific antibodies have demonstrated that the interaction between F. hepatica mannose/fucose residues and the host receptor DC-SIGN is critical for the enhanced production of TLR-induced IL-10 and IL-27p28 [94]. This interaction leads to the internalization of parasite glycoconjugates and their routing to early endosomes [94].
- The Role of MGL: The Tn antigen on F. hepatica is recognized by MGL (specifically the mMGL2 ortholog in mice) [93]. During infection, MGL-expressing CD11c+ cells are recruited to infection sites like the peritoneum and liver. These cells produce regulatory cytokines (IL-10, TGFβ) and are instrumental in expanding Th2/regulatory T cells while suppressing Th1 responses [93].
The Scientist's Toolkit: Key Research Reagents and Methods
To investigate the glycobiology of F. hepatica, researchers employ a specific set of reagents and methodological approaches. Table 2 outlines essential tools for studying glycan-mediated immune modulation.
Table 2: Key Research Reagents and Methods for Investigating F. hepatica Glycobiology
| Reagent / Method | Function and Application in Research |
|---|---|
| FhTE (F. hepatica Total Extract) | A crude protein lysate from adult worms containing native glycoconjugates; used as a stimulus in cell cultures to assess immunomodulatory properties [92] [93]. |
| Sodium meta-Periodate (Oxidation) | A chemical treatment that oxidizes and destroys carbohydrate structures on glycoconjugates. Used to create control extracts (FhmPox) to confirm the glycan-dependent nature of observed effects [92] [94]. |
| CLR-Blocking Antibodies | Specific monoclonal antibodies against immune receptors (e.g., anti-DC-SIGN, anti-MGL, anti-MR). Used to block the glycan-receptor interaction and determine the specific receptor mediating an immunomodulatory effect [93] [94]. |
| Recombinant CLR-Fc Chimeras | Soluble recombinant receptors (e.g., DC-SIGN-Fc, MR-Fc). Used in ELISA-style binding assays to confirm direct interaction between a specific CLR and glycans in parasite extracts [94]. |
| Lectin Staining & Mass Spectrometry | Techniques for glycan identification. Generic lectins probe specific sugar motifs (e.g., mannose, GalNAc), while mass spectrometry provides detailed structural analysis of glycan motifs [91]. |
| Mouse Infection Models | In vivo models (e.g., BALB/c mice) used to study the recruitment and function of immune cells like MGL+ CD11c+ cells at the site of infection and in lymphoid organs [93]. |
The tegument glycocalyx of Fasciola hepatica is a master regulator of the host-parasite interface, whose function is dictated by its intricate glycocode. The specific glycan motifs—such as the Tn antigen, mannose, and fucose residues—are decoded by host C-type lectin receptors like DC-SIGN and MGL on dendritic cells. This interaction initiates a signaling cascade that culminates in a modulated DC phenotype, a regulatory cytokine milieu, and the induction of Th2/Treg responses, collectively disarming the host's protective immunity. The growing understanding of this molecular dialogue offers a new frontier for therapeutic intervention. Future research integrating glycosylation studies into vaccine target selection and design holds significant promise. By targeting the very molecules the parasite uses to evade immunity, we can move closer to developing novel and effective strategies to combat fasciolosis.
Fascioliasis, a food-borne zoonotic disease of global importance, is primarily caused by two trematode parasites: Fasciola hepatica and Fasciola gigantica. These parasites, commonly known as liver flukes, affect a wide range of mammalian hosts, including humans, and impose significant economic losses on livestock production estimated to exceed US $3 billion annually [95]. Within the context of research on Fasciola hepatica, understanding the comparative biology of these two species is crucial for accurate diagnosis, epidemiological study, and the development of control strategies. While these species share a similar general life cycle, critical differences exist in their developmental timing, geographical distribution, and morphological characteristics, particularly at the egg stage. This in-depth technical guide provides a comprehensive comparative analysis of F. hepatica and F. gigantica, focusing on their life cycles and egg morphology, to serve as a resource for researchers, scientists, and drug development professionals.
Comparative Life Cycle Analysis
The life cycles of Fasciola hepatica and Fasciola gigantica are indirect and involve a series of complex developmental stages within both intermediate and definitive hosts. The general sequence is identical for both species, but key temporal and physiological differences impact their epidemiology and pathogenic profiles [1] [95].
Generalized Life Cycle Stages
- Egg Discharge and Embryonation: Immature, unembryonated eggs are discharged from the adult flukes in the biliary ducts of the definitive host and are passed into the environment with the feces [1]. The eggs become embryonated in freshwater over approximately two weeks, after which embryonated eggs release free-swimming miracidia [1] [29]. The development of miracidia inside the eggs is temperature-dependent, with maturation typically occurring within 12-16 days under laboratory conditions (26±1°C) [29].
- Snail Infection and Larval Development: The miracidia are short-lived and must invade a suitable snail intermediate host within 24 hours [95]. Inside the snail, the parasites undergo several clonal developmental stages: from sporocysts to rediae, and finally to cercariae [1] [95]. The specific snail hosts differ between the two fluke species; F. hepatica is primarily transmitted by snails of the Galba/Fossaria group, whereas F. gigantica is transmitted by snails of the Radix group [1] [96].
- Cercarial Release and Encystment: Cercariae are released from the snail and, after a brief free-swimming period, encyst as metacercariae on aquatic vegetation or other substrates [1].
- Infection of Definitive Host and Tissue Migration: A mammalian definitive host becomes infected by ingesting metacercariae-contaminated vegetation, such as watercress [1] [97]. After ingestion, the metacercariae excyst in the duodenum within an hour, and the newly excysted juveniles penetrate the intestinal wall and enter the peritoneal cavity [1] [95]. The immature flukes then migrate through the liver parenchyma.
- Maturation and Egg Production: The flukes eventually reach the biliary ducts, where they mature into adult, egg-producing flukes. The time from metacercarial ingestion to patency (egg production) is a key differentiating factor between the two species [1].
Critical Comparative Differences in Development
The most significant differences in the life cycles of F. hepatica and F. gigantica lie in the duration of the hepatic migration phase and the time to maturation, which have direct implications for the clinical presentation and management of the disease.
Table 1: Key Developmental Differences Between F. hepatica and F. gigantica
| Developmental Stage | Fasciola hepatica | Fasciola gigantica |
|---|---|---|
| Liver Parenchyma Migration | 5–6 weeks [95] | Up to 11 weeks [95] |
| Time to Maturation (Patency) | 8–12 weeks post-infection (WPI) [1] [95] | 12–16 weeks post-infection (WPI) [1] [95] |
| Primary Snail Intermediate Hosts | Galba truncatula and other Galba/Fossaria species [1] [96] | Radix natalensis and other Radix species [1] [96] |
The following diagram illustrates the shared life cycle and highlights these critical temporal and host-specific differences.
The extended pre-patent period and migration time of F. gigantica mean that the acute phase of the disease, caused by larval migration through the liver, is prolonged compared to F. hepatica infection. This has direct consequences for drug development, as therapeutic targets may need to be effective against juvenile parasites for a longer duration in F. gigantica-endemic regions [95].
Egg Morphology and Differentiation
The morphological differentiation of Fasciola eggs is a critical component of laboratory diagnosis and epidemiological surveillance. However, this task presents significant challenges due to the substantial overlap in the characteristics of eggs from different species.
Morphological Characteristics and Challenges
The eggs of both F. hepatica and F. gigantica are broadly ellipsoidal, operculated, and passed in the stool unembryonated [1]. This morphological similarity makes them difficult to distinguish reliably based on visual inspection alone. Furthermore, Fasciola spp. eggs can be challenging to differentiate from eggs of other trematodes, such as Fasciolopsis buski and some Echinostoma species, and are often reported as "Fasciola/Fasciolopsis" eggs due to this morphologic overlap [1].
Table 2: Comparative Morphology of Fasciola Eggs and Miracidia
| Characteristic | Fasciola hepatica | Fasciola gigantica | Notes |
|---|---|---|---|
| Egg Size | 130–150 µm by 60–90 µm [1] | 160–190 µm by 70–90 µm [98] | Size cannot reliably distinguish species; significant overlap exists [1] [98]. |
| Egg Shape & Features | Broadly ellipsoidal, operculated. Abopercular end may have a roughened area [1]. | Broadly ellipsoidal, operculated [1]. | Shape of the operculum may differ, appearing short and straight/flat in F. hepatica [29]. |
| Miracidium Size (Fixed) | 110–150 µm (av. 136 µm) by 60–80 µm (av. 74 µm) [29] | 98–119 µm (av. 110 µm) by 63–77 µm (av. 70 µm) [29] | Measured after fixation. |
| Miracidium Life Span | Does not exceed 10 hours [29] | 9 to 12 hours [29] | After hatching. |
| Development to Maturation | 13–15 days at 26±1°C [29] | 12–16 days at 26±1°C [29] | Under normal laboratory conditions. |
A significant diagnostic pitfall is pseudofascioliasis (or false fascioliasis), where eggs are detected in stool not due to an active infection, but because the patient has recently consumed liver from an infected animal, and non-infective eggs are passing through the digestive tract. This can be avoided by having the patient abstain from eating liver for several days before a repeat stool examination [1].
Advanced Diagnostic and Experimental Methodologies
Given the limitations of morphological differentiation, molecular and immunodiagnostic techniques have become indispensable for precise species identification and for detecting infections during the acute phase, prior to patency.
Molecular Differentiation Protocols
DNA-based methods provide unequivocal identification of Fasciola species, as genotypic characters are not influenced by ecological or geographical factors [98]. One established and reliable method is the PCR-Restriction Fragment Length Polymorphism (PCR-RFLP) assay targeting the Internal Transcribed Spacer 1 (ITS1) region of the ribosomal DNA gene.
Detailed Protocol: PCR-RFLP for Differentiation of F. hepatica and F. gigantica [98]
DNA Extraction:
- Parasite material (adult flukes fixed in 70% ethanol) is washed in PBS.
- Mechanical Disruption: Fragments of the worms can be crushed between two glass slides to break down the cells effectively.
- The crushed material is then used for DNA extraction via a conventional phenol-chloroform method or using commercial FTA cards.
PCR Amplification:
- Primers: Select primers to amplify a ~463 bp region of the ITS1 sequence.
- Reaction Mixture: Standard PCR mixture containing buffer, dNTPs, primers, DNA polymerase, and the extracted template DNA.
- Cycling Conditions:
- Initial denaturation (e.g., 94°C for 3-5 minutes).
- 30-35 cycles of: Denaturation (e.g., 94°C for 30-60 seconds), Annealing (temperature optimized for primers, e.g., 60°C for 15-30 seconds), Extension (e.g., 72°C for 60 seconds).
- Final extension (e.g., 72°C for 5-10 minutes).
- Visualization: The PCR product is electrophoresed on an agarose gel. A single, sharp band of the expected size (~460 bp) should be visible for successful amplification.
Restriction Digestion:
- Enzyme: Use the restriction enzyme TasI.
- Procedure: Incubate the PCR product with TasI and the appropriate buffer at the enzyme's optimal temperature for a specified period.
- Analysis: The digested fragments are separated by gel electrophoresis. The restriction profile is species-specific:
- F. hepatica produces one characteristic banding pattern.
- F. gigantica produces a distinctly different banding pattern, allowing clear differentiation.
The workflow for this diagnostic process, integrating both traditional and molecular methods, is outlined below.
Serological Detection Methods
Serologic testing is particularly valuable for diagnosing acute fascioliasis, as specific antibodies become detectable 2 to 4 weeks after infection, which is much earlier than patency [1]. The CDC utilizes an immunoblot assay based on a recombinant F. hepatica antigen (FhSAP2). A positive reaction is defined by a band at approximately 38 kDa, with a reported sensitivity of ≥94% and specificity of ≥98% for chronic human infection [1]. This assay can also help rule out pseudofascioliasis [1].
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and materials essential for conducting research on Fasciola species, particularly for the differentiation experiments described.
Table 3: Key Research Reagent Solutions for Fasciola Differentiation
| Reagent/Material | Function/Application | Specific Example / Note |
|---|---|---|
| Adult Fluke Specimens | Source of genomic DNA for molecular identification and morphological studies. | Collected from bile ducts of infected livestock; fixed in 70% ethanol for storage and transport [98] [99]. |
| Phenol-Chloroform / FTA Cards | DNA extraction from parasite tissue. | FTA cards provide a simple, commercial alternative to conventional liquid-phase extraction [98]. |
| ITS1-Specific Primers | PCR amplification of the ITS1 genetic marker. | Amplifies a ~463 bp region that contains species-specific sequences [98]. |
| TasI Restriction Enzyme | Digestion of PCR amplicons for RFLP. | Cuts the ITS1 PCR product of F. hepatica and F. gigantica into distinct fragment patterns for identification [98]. |
| Recombinant FhSAP2 Antigen | Target antigen for immunoblot serodiagnosis. | Used in a CDC-developed immunoblot assay for detection of specific antibodies in human serum [1]. |
The comparative analysis of Fasciola hepatica and Fasciola gigantica reveals a complex picture of biological similarity nuanced by critical differences in development and morphology. While their shared life cycle dictates a common epidemiological framework, the longer hepatic migration and pre-patent period of F. gigantica [1] [95], coupled with the subtle yet significant variations in egg and miracidial morphology [29], underscore the necessity for sophisticated diagnostic approaches. Reliable differentiation, which is fundamental for targeted control and drug development strategies, has moved beyond traditional microscopy to rely on molecular tools like PCR-RFLP of the ITS1 region [98] and specific serological assays [1]. Future research, particularly genomic studies, will continue to refine our understanding of the population genetics, evolutionary history, and host adaptation of these parasites, ultimately informing the development of more effective interventions against a disease of significant veterinary and medical importance [100] [96].
The pursuit of a vaccine against Fasciola hepatica, the common liver fluke, represents a critical frontier in the control of a parasite affecting millions of humans and livestock worldwide. With economic losses estimated at USD 3.2 billion annually in the livestock industry alone and rising anthelmintic resistance, the need for effective vaccines is pressing [101]. This technical guide frames the vaccine design challenge within the context of the parasite's intricate life cycle and distinctive egg morphology, which are essential for understanding host-parasite interactions and identifying intervention points. The adult flukes, residing in the bile ducts of the liver, produce eggs that are broadly ellipsoidal and operculated, measuring 130–150 µm long by 60–90 µm wide, and are passed unembryonated in the host's feces [1]. Recent research has detailed that these eggs may possess an appendage on the abopercular pole, a feature previously unreported and significant for diagnostic differentiation [23].
The life cycle of F. hepatica is complex, involving an intermediate snail host and several free-living stages. Immature eggs discharged in the feces embryonate in freshwater over approximately two weeks, releasing miracidia that invade a suitable snail intermediate host [1]. Within the snail, the parasites undergo several developmental stages (sporocysts, rediae, and cercariae). The cercariae are released and encyst as metacercariae on aquatic vegetation. Mammals, including humans, become infected by ingesting contaminated vegetation, such as watercress. After ingestion, metacercariae excyst in the duodenum, penetrate the intestinal wall, and migrate as immature flukes through the liver parenchyma into the biliary ducts, where they mature into adults and produce eggs—a process taking 3–4 months in humans [1] [7] [46]. This detailed understanding of the life cycle and morphology provides the foundational context for identifying vulnerable stages for vaccine intervention. This guide posits that glycan-based vaccine strategies, inspired by recent successes in nematode parasitology, offer a promising avenue to disrupt these complex biological processes and achieve protective immunity.
Glycans in Parasite Biology and Immune Evasion
The Biological Significance of Parasite Glycans
Glycans, complex sugar chains covalently attached to proteins (forming glycoproteins) or lipids, are critical regulators of immune recognition and response. They act as universal molecular barcodes that facilitate immune surveillance [102]. Parasitic nematodes and trematodes exhibit a diverse range of glycans that play crucial roles in host-parasite interactions. These glycans possess unique structural features and show species- and clade-specific variations, making them ideal targets for immunological intervention [103]. Key modifications such as phosphorylcholine (PC) and O-methylation (Me) are common in nematode glycans and are significant for their immunomodulatory capabilities [103]. These distinct glycans are highly immunogenic to mammalian hosts and play significant immunoregulatory roles during infection, often contributing to the parasite's ability to evade or suppress the host immune system [103].
The glycosylation processes in parasitic nematodes result in unique glycan modifications not present in their hosts. For instance, the H11 aminopeptidases in Haemonchus contortus are naturally glycosylated with distinct N-glycan structures compared to those found in mammals. These glycan modifications are often considered crucial for maintaining antigenicity and have become a focal point for vaccine development [104]. The natural glycan shielding phenomenon, used by pathogens like HIV and SARS-CoV-2 to escape immune recognition, can be adapted for vaccine design by strategically adding glycans to surface immunogens to focus the immune response on conserved, therapeutic epitopes [105].
Glycan Engineering as a Strategic Tool
Glycan engineering encompasses two primary strategic approaches for vaccine design:
- Glycan Masking: A technique involving the addition of sugar chains (glycans) to the surface of immunogen candidates to hide regions of low interest, thereby focusing the immune system on highly therapeutic epitopes. This approach is inspired by viruses such as influenza and HIV, which naturally incorporate additional glycosylation sites to prevent the binding of therapeutic antibodies [105].
- Glycan Unmasking: The strategic removal of native glycans from antigen surfaces to expose conserved, vulnerable epitopes that are otherwise shielded from immune recognition. This reverse approach can be equally valuable for revealing critical neutralization sites [105].
Recent advances have enabled precise control over these strategies through CRISPR-based glycoediting, synthetic biology, and computational modeling, allowing researchers to modulate glycan structures at the genetic and enzymatic levels to enhance antigen presentation and immune activation [102]. These innovations are redefining the possibilities for glycoengineered vaccines against challenging pathogens like Fasciola hepatica.
Success Stories: Glycan-Based Vaccines in Nematode Parasitology
The Haemonchus contortus Model: A Glycoengineering Breakthrough
The development of a novel glycoengineered vaccine against Haemonchus contortus, a highly pathogenic nematode in small ruminants, represents a landmark achievement in parasite vaccinology. A randomized, controlled trial evaluated a glycoengineered vaccine (GEA) produced in Hi5 insect cells, comprising five antigens (H11, H11-1, H11-2, H11-4, and GA1) designed to incorporate nematode-type glycan modifications [104].
Table 1: Efficacy Outcomes of Glycoengineered H. contortus Vaccine
| Parameter | GEA Vaccine Performance | Non-Glycoengineered (NEA) Counterpart | Control Group |
|---|---|---|---|
| Fecal Egg Count Reduction | 81.09% reduction | Less effective than GEA | Baseline |
| Worm Burden Reduction | 25.36% reduction | Less effective than GEA | Baseline |
| Anemia Indicators | Lower degree of anemia compared to NEA and control | More anemia than GEA | Most severe anemia |
| Immune Response | Elevated IgG/IgE responses | Elevated but less protective responses | Baseline |
The results demonstrated that the glycoengineered form (GEA) significantly outperformed its non-glycoengineered counterpart (NEA), underscoring the importance of specific glycan modifications in achieving protective immunity. The GEA vaccine not only reduced parasite burden but also ameliorated the clinical severity of infection, as evidenced by improved packed cell volume, erythrocyte counts, and hemoglobin concentrations compared to positive controls [104]. This success highlights the critical role of proper glycosylation in preserving the conformational integrity and antigenicity of key parasite antigens.
Research Reagent Solutions for Glycan-Based Vaccine Development
Table 2: Essential Research Reagents for Glycan-Based Vaccine Development
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Expression Systems | Hi5 insect cells, mammalian cell lines | Production of recombinant glycoproteins with eukaryotic post-translational modifications |
| Key Antigen Targets | H11 aminopeptidases, H-gal-GP complex, GA1 | "Hidden" intestinal proteins of nematodes not naturally exposed to host immune system |
| Glycoengineering Tools | CRISPR-based glycoediting, glycosyltransferases | Precise modification of glycan structures on target antigens |
| Analytical Techniques | Lectin microarrays, mass spectrometry, NMR | Characterization of glycan structures and verification of engineering outcomes |
| Adjuvant Systems | TLR-activating glycan nanocarriers | Enhancement of immune responses to glycan-based antigens |
| Delivery Platforms | Virus-like particles (VLPs), nanoparticles | Improved antigen presentation and stability |
The table above outlines critical reagents and platforms that have enabled successful glycan-based vaccine development. The choice of expression system is particularly crucial, as it determines the fidelity of glycan modifications on recombinant antigens. Hi5 insect cells have proven effective for producing nematode-type glycan modifications, as demonstrated in the H. contortus vaccine trial [104]. Furthermore, the use of glycan-modified nanoparticles can improve antigen stability, optimize immune responses, and expand vaccine targets by enhancing antigen presentation to immune cells [102].
Experimental Protocols for Glycan-Focused Vaccine Development
Protocol 1: Glycan Characterization of Parasite Antigens
Objective: To isolate and characterize the glycan profiles of potential vaccine antigens from Fasciola hepatica or nematode parasites.
Methodology:
- Antigen Preparation: Isolate native antigens from parasite extracts (e.g., from adult worms or excretory-secretory products) or express recombinant candidates in appropriate expression systems (e.g., Hi5 insect cells for nematode-type glycans) [104].
- Glycan Release: Liberiate N- and O-linked glycans enzymatically (e.g., using PNGase F for N-glycans) or chemically (e.g., β-elimination for O-glycans).
- Purification and Derivatization: Purify released glycans using solid-phase extraction and label with fluorescent tags (e.g., 2-AB) for detection.
- Structural Analysis:
- Lectin Microarrays: Probe glycan samples with a panel of lectins with known carbohydrate specificities to determine broad glycan features.
- Mass Spectrometry (MS): Perform MALDI-TOF MS or LC-ESI-MS/MS for detailed structural characterization, including sequencing and identification of modifications like phosphorylcholine [103].
- Nuclear Magnetic Resonance (NMR): Use for complete structural elucidation of novel glycan epitopes.
Applications: This protocol generates essential baseline data on native parasite glycan structures, informing either glycan masking or unmasking strategies for vaccine design.
Protocol 2: Evaluation of Glycoengineered Vaccine Efficacy
Objective: To assess the protective efficacy and immunogenicity of a glycoengineered vaccine candidate in a relevant animal model.
Methodology:
- Vaccine Formulation: Prepare the glycoengineered immunogen (e.g., expressing key antigens like FhSAP2 for Fasciola [1] or H11 for nematodes [104] with optimized glycan structures) with a suitable adjuvant.
- Animal Immunization: Administer the vaccine to the model host (e.g., sheep for Fasciola or Haemonchus studies) following a prime-boost regimen (e.g., vaccinations at weeks 0, 4, and 8) [33].
- Challenge Infection: Infect vaccinated and control animals with a known number of infective parasite stages (e.g., metacercariae for Fasciola, L3 larvae for nematodes).
- Outcome Measures:
- Parasitological: Assess fecal egg counts (using Flukefinder or Mini-FLOTAC for higher sensitivity [101]) and adult worm burdens at necropsy.
- Pathological: Evaluate liver damage (e.g., fibrosis, tract counts) and bile duct thickening for Fasciola infections [33].
- Immunological: Monitor antigen-specific antibody responses (IgG1, IgG2, IgE) by ELISA and cellular immune responses [33].
- Clinical: Track parameters like packed cell volume, hemoglobin levels, and body weight changes.
Interpretation: Compare outcomes between vaccinated and control groups to determine vaccine efficacy in reducing parasite establishment, development, and pathogenicity.
Conceptual Framework and Technical Workflows
Diagram 1: Integrated Workflow for Glycan-Based Vaccine Development. This workflow outlines the sequential phases from foundational research through glycoengineering to comprehensive evaluation, emphasizing the iterative nature of vaccine design.
Diagram 2: Immune Recognition of Native vs. Engineered Glycan Antigens. This diagram contrasts how native parasite glycans often mediate immunoevasion, while strategically engineered glycan antigens can redirect and enhance protective immune responses toward vulnerable epitopes.
Applying Nematode Success to Fasciola hepatica Vaccine Design
The lessons from nematode glycan-based vaccine development provide a strategic roadmap for addressing the challenges of Fasciola hepatica vaccinology. Several key translational opportunities emerge:
Target Antigen Selection and Engineering
Building on the nematode success with "hidden" intestinal antigens like H11, Fasciola vaccine efforts should prioritize antigens expressed during critical life cycle stages, particularly the newly excysted juveniles (NEJs) that represent the first contact with the host immune system [33]. Proteomic analyses of NEJs interacting with host intestinal epithelial cells have identified potential candidate antigens, including KTSPIDP (kazal-type serine protease inhibitor domain protein), VGHC1 (voltage-gated hydrogen channel), CRTA (cholecystokinin receptor type A), and CAL (catenin alpha-like) [33]. These antigens, which play roles in invasion and migration, could be optimized through glycan engineering to enhance their immunogenicity.
The FhSAP2 antigen from Fasciola hepatica, used in a CDC-developed immunoblot assay with 94% sensitivity and 98% specificity for chronic infection, represents another promising candidate for glycoengineering approaches [1]. Recombinant production of these antigens in glycoengineering-capable expression systems (e.g., Hi5 insect cells or glycoengineered yeast platforms) could ensure the incorporation of immunologically relevant glycans that mimic native structures or strategically mask immuno-dominant but non-protective epitopes.
Strategic Integration with Life Cycle Knowledge
A comprehensive Fasciola vaccine strategy should leverage detailed knowledge of the parasite's life cycle and biology:
- Targeting NEJs: The migration of NEJs through the intestinal wall and liver parenchyma represents a vulnerable "point of no return" where therapeutic intervention could prevent establishment of infection [33]. Glycan-engineered vaccines targeting NEJ surface antigens could block invasion and migration pathways.
- Leveraging Egg Morphology: The distinct morphology of Fasciola eggs, including potential appendages and specific surface glycans, could be exploited to develop transmission-blocking vaccines that target components essential for egg development or miracidial formation [23].
- Combining Antigens: Given the complexity of the parasite's life cycle, a multi-antigen vaccine targeting different stages (NEJs, juveniles, adults) may be necessary for comprehensive protection. Glycan engineering could optimize the presentation of each component to focus immune responses on conserved functional domains.
The integration of glycan engineering strategies with fundamental research on Fasciola hepatica biology represents a promising pathway toward overcoming historical challenges in liver fluke vaccine development. The documented successes in nematode parasitology, particularly with Haemonchus contortus, demonstrate that proper attention to glycan structures can transform marginally effective antigens into protective vaccines. Future research should prioritize the comprehensive characterization of Fasciola glycans, the application of advanced glycoengineering tools like CRISPR-based editing, and the utilization of improved delivery platforms such as glycan-modified nanoparticles. As these technologies converge with our growing understanding of Fasciola life cycle and host interactions, the prospect of a effective glycan-based vaccine moves closer to reality, offering a sustainable solution to a pervasive global parasitic disease.
Fasciola hepatica, commonly known as the liver fluke, is a parasitic trematode of profound global veterinary and medical significance. The World Health Organization classifies fasciolosis as a neglected tropical disease, with an estimated 91 to 180 million people at risk of infection annually [106] [23]. In livestock, the economic losses are staggering, estimated at approximately $3 billion annually, stemming from reduced productivity, fertility issues, and treatment costs [33] [107] [108]. The parasite's life cycle involves complex host interactions, beginning with the ingestion of metacercariae encysted on vegetation. These develop into newly excysted juveniles (NEJs) that traverse the intestinal wall, migrate through the liver parenchyma—causing significant tissue damage—and mature into adults residing in the bile ducts [33] [108]. The eggs produced by adults are released in feces, continuing the cycle. Recent research has revealed that these eggs may possess a previously unreported appendage, a morphological feature that could have implications for diagnostics and understanding the parasite's biology [23].
The escalating problem of anthelmintic resistance, particularly to triclabendazole, underscores the urgent need for novel control strategies [106] [33] [107]. While vaccine development has been pursued for decades, no commercially viable product exists. A significant hurdle is the parasite's sophisticated immunomodulatory capacity, which suppresses protective host immune responses [107] [108]. The integration of glycomics—the study of parasite glycans—with genomic insights presents a promising, multidisciplinary path forward for identifying novel therapeutic targets.
Current Omics Landscape and Its Potential for Integration
Genomic Foundations
The sequencing of the F. hepatica genome revealed it to be one of the largest among known pathogens at approximately 1.3 Gb [109]. This substantial size is not due to whole-genome duplication but involves an expansion of specific gene families and repetitive elements. A key finding with direct relevance to drug and vaccine development is the substantial genetic polymorphism within parasite populations. Notably, 48% of genes contain at least one non-synonymous single nucleotide polymorphism (SNP) [109]. This diversity is particularly elevated in genes shared with other parasitic helminths, suggesting a genetic basis for the parasite's adaptability to diverse host environments and potential for rapid evolution in response to drug pressure.
Table 1: Key Features of the Fasciola hepatica Genome
| Genomic Feature | Characteristic | Implication for Drug/Vaccine Development |
|---|---|---|
| Genome Size | ~1.3 Gb | One of the largest pathogen genomes sequenced; indicates complexity. |
| Polymorphism | 48% of genes have ≥1 non-synonymous SNP [109] | High adaptive potential; vaccines/drugs may need to target conserved regions. |
| Expressed Genes | >8,000 genes expressed by NEJs during migration [110] | Large pool of potential stage-specific antigen candidates. |
| Notable Gene Families | Expanded protease and tubulin families [109] | Potential drug targets (e.g., protease inhibitors, anthelmintics). |
| Glycosylation Machinery | 153 putative glycosylating genes identified [106] | Confirms capacity for extensive protein glycosylation. |
Glycomic Complexity
The surface of F. hepatica is coated in a dynamic glycocalyx, a dense layer of carbohydrates that serves as the primary interface with the host [106]. Recent glycomic studies of the critical NEJ stage have uncovered remarkable complexity. A groundbreaking glycoproteomic analysis characterized 123 NEJ glycoproteins (71 of which are secreted), mapping 356 individual glycosites decorated by a diverse array of 53 N-glycan and 16 O-glycan structures [110]. This extensive micro-heterogeneity, where a single protein can be modified by multiple different glycan structures, generates significant molecular diversity far beyond what is predicted by the genome or proteome alone. These glycans are not random; they include unique structures such as phosphorylcholine (PC)-modified glycans and various truncated paucimannosidic sugars, which are known to play roles in immunomodulation [106] [110].
Integrated Workflow: From Omics Data to Candidate Validation
The path from large-scale omics data to viable therapeutic candidates requires a structured, iterative workflow that leverages the strengths of both genomic and glycomic approaches.
Experimental Protocols for Key Steps
Protocol 1: Glycoproteomic Characterization of Parasite Antigens
This methodology is adapted from detailed mass spectrometric workflows used to decipher the NEJ glycoproteome [110].
- Sample Preparation: Obtain F. hepatica metacercariae from a commercial source. Excyst NEJs in vitro using an acid-pepsin system followed by incubation in sodium taurocholate. Culture NEJs for 24 hours in RPMI-1640 medium supplemented with HEPES, glucose, and gentamycin at 37°C with 5% CO₂.
- Extract Preparation:
- Somatic Extract (Som): Wash NEJs in DPBS, lyophilize, and homogenize in lysis buffer.
- Excretory/Secretory (ES) Extract: Concentrate culture media using 3 kDa Amicon Ultra filters.
- Glycopeptide Enrichment: Digest protein extracts with trypsin/Lys-C. Enrich glycopeptides using solid-phase extraction or lectin-affinity chromatography.
- LC-MS/MS Analysis: Analyze enriched glycopeptides via liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS). Fragment peptides to obtain both peptide sequence and glycan composition data.
- Data Processing: Process raw data using software like Byonic or pGlyco to identify glycopeptides. Map glycopeptides and their associated glycan forms to proteins using curated F. hepatica proteome databases.
Protocol 2: Functional Characterization of Glycan-Mediated Immune Modulation
This protocol is based on experiments demonstrating the immunomodulatory effects of F. hepatica glycans [106].
- Lectin/Glycan Blocking Assays:
- Isinate peritoneal macrophages from BALB/c mice.
- Pre-incubate cells for 1 hour with glycan inhibitors (e.g., 100 µg/mL mannan, laminarin) or lectin-blocking antibodies (e.g., anti-mannose receptor, anti-dectin-1).
- Stimulate cells with F. hepatica ES products (e.g., 50 µg/mL) for 24 hours.
- Readout: Analyze culture supernatants for anti-inflammatory cytokines (e.g., IL-10, TGF-β) by ELISA. Quantify expression of alternative activation markers (e.g., Arginase-1) in cell lysates via qPCR or Western blot.
- Interpretation: A significant reduction in anti-inflammatory markers in inhibited samples indicates specific glycan-receptor interactions are critical for immune modulation.
The Scientist's Toolkit: Essential Research Reagents
Successfully navigating this integrated research landscape requires a specific set of reagents and tools.
Table 2: Key Research Reagent Solutions for Integrated Glycomics-Genomics Research
| Reagent / Tool | Function / Application | Example in Context |
|---|---|---|
| Recombinant Glycoproteins | Vaccine immunogen testing; study of structure-function relationships. | Testing efficacy of glycoengineered cathepsin L variants [106]. |
| Glycan Inhibitors | Functional blocking of specific glycan-lectin interactions. | Mannan and laminarin used to block macrophage activation by FhES [106]. |
| Lectin Arrays | High-throughput profiling of glycan motifs on parasite surfaces or EVs. | Characterizing the glycan landscape of extracellular vesicles [110]. |
| Specific Lectins (e.g., ConA, GNL) | Probing for specific sugar residues in inhibition or localization studies. | ConA and GNL inhibit NEJ migration through gut tissue [106]. |
| Mass Spectrometry Platforms | Comprehensive identification and quantification of glycopeptides. | Mapping 356 glycopeptides and 1690 N-glycan forms in NEJs [110]. |
| Gene Silencing Tools (RNAi) | Functional validation of target genes, including glycosyltransferases. | Used in related trematodes (e.g., S. mansoni) to assess gene function [111]. |
Visualization of the Host-Parasite Glycocode Interface
A critical conceptual advance is understanding how parasite glycans and host lectins interact to shape the immune response. This interaction is a key determinant of infection outcome.
The integration of glycomics and genomics represents a paradigm shift in the search for novel interventions against fasciolosis. While genomics provides the blueprint of potential targets, glycomics reveals a critical, dynamic layer of biological complexity and immune interaction that has been largely overlooked. The documented failure of recombinant protein vaccines compared to their native, glycosylated counterparts underscores the importance of this approach [106]. Future efforts must focus on:
- Systematically mapping the glycosylation patterns of leading vaccine candidates across all life stages.
- Employing glycoengineering in recombinant expression systems to better mimic native glycan structures.
- Targeting the parasite's glycosylation machinery itself (e.g., specific glycosyltransferases) as a novel drug strategy.
By moving beyond a protein-centric view and fully embracing the complexity of the parasite's glycocode, researchers can uncover a new generation of sophisticated, rationally designed tools to combat this persistent and economically devastating parasite.
Conclusion
The intricate life cycle and distinct egg morphology of Fasciola hepatica are foundational to understanding, diagnosing, and controlling fasciolosis. While established coprological methods remain vital, recent discoveries of egg appendages and an expanded host range underscore the dynamic nature of this parasite. The growing challenge of anthelmintic resistance necessitates a shift towards novel therapeutic targets, with the parasite's tegument and its unique glycocode presenting promising avenues for intervention. Future research must leverage integrated 'omics' technologies to fully elucidate host-parasite interactions at the molecular level. For the research and drug development community, the priority lies in translating these insights into applied solutions: refining diagnostic specificity, exploiting glycobiology for vaccine design, and developing new chemical entities that overcome existing resistance mechanisms, thereby mitigating the significant global burden of this zoonotic disease.
References
- 1. DPDx - Fascioliasis [https://www.cdc.gov/dpdx/fascioliasis/index.html]
- 2. Across intra-mammalian stages of the liver f luke Fasciola ... [https://www.nature.com/articles/srep32796]
- 3. Maintenance of Life Cycle Stages of Fasciola hepatica in ... [https://pubmed.ncbi.nlm.nih.gov/32399917/]
- 4. Fasciola hepatica: Updates on egg morphology, host ... [https://www.sciencedirect.com/science/article/pii/S2405676624000192]
- 5. Fasciola hepatica: Updates on egg morphology, host range ... [https://pubmed.ncbi.nlm.nih.gov/39157754/]
- 6. a review of impacts across the parasite life cycle [https://link.springer.com/article/10.1007/s00436-024-08319-6]
- 7. Lifecycle of Liver Fluke [https://www.scops.org.uk/internal-parasites/liver-fluke/lifecycle/]
- 8. Exploration of animal models to study the life cycle of Fasciola ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10090765/]
- 9. Fumarate respiration of Fasciola flukes as a potential drug ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2023.1302114/full]
- 10. Understanding the main route of drug entry in adult ... [https://www.sciencedirect.com/science/article/abs/pii/S0014489417301601]
- 11. Fasciola hepatica [https://en.wikipedia.org/wiki/Fasciola_hepatica]
- 12. The definitive and intermediate hosts of Fasciola hepatica ... [https://pubmed.ncbi.nlm.nih.gov/11411948/]
- 13. Study of the migration of Fasciola hepatica juveniles across ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9518905/]
- 14. Fluke Infections of the Liver - Infections - Merck Manual Consumer... [https://www.merckmanuals.com/home/infections/parasitic-infections-trematodes-flukes/fluke-infections-of-the-liver]
- 15. Diagram of Fasciola : Classification and... - GeeksforGeeks hepatica [https://www.geeksforgeeks.org/biology/fasciola-hepatica-diagram-classification/]
- 16. FASCIOLA HEPATICA AND SCHISTOSOMA MANSONI [https://pmc.ncbi.nlm.nih.gov/articles/PMC3564592/]
- 17. Fasciola [https://www.parasite.org.au/para-site/text/fasciola-text.html]
- 18. Fluke Life Cycle [https://wormboss.com.au/about-worms/worm-life-cycles-and-life-stages/fluke-life-cycle/]
- 19. Fasciola hepatic.- complete life cycle, Different larva stages [https://www.slideshare.net/slideshow/fasciola-hepatic-complete-life-cycle-different-larva-stages/244053614]
- 20. Experimental Life History and Biological Characteristics of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4384800/]
- 21. Environmental detection of Fasciola hepatica by loop ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9357369/]
- 22. The Fasciola hepatica egg development and hatching test ... [https://link.springer.com/article/10.1007/s00436-023-08074-0]
- 23. Fasciola hepatica: Updates on egg morphology, host range ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11327948/]
- 24. An Integrated Transcriptomics and Proteomics Analysis of ... [https://www.sciencedirect.com/science/article/pii/S1535947620340172]
- 25. Fasciola hepatica Gastrodermal Cells Selectively Release ... [https://www.mdpi.com/1422-0067/23/10/5525]
- 26. Discriminatory dose of nitroxynil and albendazole sulfoxide ... [https://www.sciencedirect.com/science/article/abs/pii/S0014489424001875]
- 27. Hepatobiliary fascioliasis: A neglected re-emerging threat ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12186194/]
- 28. Transcriptomic and proteomic profiling of peptidase ... [https://www.nature.com/articles/s41598-022-14419-z]
- 29. Development and hatching mechanism of Fasciola eggs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3730791/]
- 30. The hatching mechanism of the egg of Fasciola hepatica L. [https://www.cambridge.org/core/journals/parasitology/article/hatching-mechanism-of-the-egg-of-fasciola-hepatica-l/77A22747BB29D4E38F0C797095DBBCEE]
- 31. Comparative analysis of the viability of Fasciola hepatica ... [https://www.sciencedirect.com/science/article/abs/pii/S0304401725002420]
- 32. Population genetic analysis of the liver fluke Fasciola hepatica ... [https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-025-06701-6]
- 33. Evaluation of a novel vaccine candidate derived from ... [https://www.nature.com/articles/s41598-025-00109-z]
- 34. The structure and permeability of the shell and vitelline ... [https://www.cambridge.org/core/journals/parasitology/article/structure-and-permeability-of-the-shell-and-vitelline-membrane-of-the-egg-of-fasciola-hepatica/C831DCE0F941C025231A01DCC2BF5522]
- 35. Development and hatching mechanism of Fasciola eggs ... [https://www.sciencedirect.com/science/article/pii/S1319562X10000483]
- 36. Ultrastructural Changes in the Tegument and Tissues of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11649840/]
- 37. A highly sensitive molecular diagnostic workflow for Fasciola ... [https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0005931]
- 38. A dedicated deep learning workflow for automatic Fasciola ... [https://www.sciencedirect.com/science/article/pii/S0020751925001419]
- 39. In vitro co-culture of Fasciola hepatica newly excysted ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11938319/]
- 40. In vitro co-culture of Fasciola hepatica newly excysted ... [https://pubmed.ncbi.nlm.nih.gov/40132201/]
- 41. Optimization and workflow of in vitro culture of adult Fasciola ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11614084/]
- 42. In vitro evaluation of the efficacy of acetic acid at 6% (table ... [https://www.sciencedirect.com/science/article/abs/pii/S0014489425001365]
- 43. Maintenance in vitro of Fasciola hepatica [https://www.nature.com/articles/174654a0]
- 44. Validation of the natural sedimentation technique in ... [https://www.sciencedirect.com/science/article/abs/pii/S1383576924000400]
- 45. Challenges in applying W.A.A.V.P. criteria to diagnosing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12538080/]
- 46. Life Cycle of Fasciola hepatica [https://www.merckmanuals.com/home/multimedia/image/life-cycle-of-fasciola-hepatica]
- 47. Composite Fasciola hepatica faecal egg sedimentation test ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6583716/]
- 48. Phenotypic analysis of adults and eggs of Fasciola ... [https://www.cambridge.org/core/journals/journal-of-helminthology/article/phenotypic-analysis-of-adults-and-eggs-of-fasciola-hepatica-by-computer-image-analysis-system/CBEA313878FA51E1FA1065AC5866399B]
- 49. Fasciola hepatica: Histology of the Reproductive Organs ... [https://www.mdpi.com/2076-0817/4/3/431]
- 50. Comparison of Fasciola hepatica genotypes in relation to ... [https://www.sciencedirect.com/science/article/abs/pii/S0304401715002058]
- 51. Comparison of Three Diagnostic Methods to Detect ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9787907/]
- 52. Egg forensics: An appraisal of DNA sequencing to assist in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7102845/]
- 53. Phylogenetic Analysis and DNA-based Species Confirmation ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0054063]
- 54. Trematode Infection Workup: Laboratory Studies, Imaging ... [https://emedicine.medscape.com/article/230112-workup]
- 55. Evaluation of the ovicidal activity and fasciolicidal activity of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10751259/]
- 56. Assessing the Efficacy of Albendazole against Fasciola ... [https://www.mdpi.com/2306-7381/8/11/249]
- 57. Activity of cumin essential oil to control fascioliasis [https://www.sciencedirect.com/science/article/pii/S0014489423001285]
- 58. Conventional and Advanced Methods Used for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11732280/]
- 59. Serological diagnosis of fasciolosis (Fasciola hepatica) in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10509555/]
- 60. Evaluation of the Performance of Five Diagnostic Tests for ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0161621]
- 61. Fascioliasis and fasciolopsiasis: Current knowledge and ... [https://pubmed.ncbi.nlm.nih.gov/33278757/]
- 62. DPDx - Fasciolopsiasis [https://www.cdc.gov/dpdx/fasciolopsiasis/index.html]
- 63. Analysis of Fasciola hepatica HDM-1 mRNA Expression ... [https://jzd.tabrizu.ac.ir/article_20440.html]
- 64. Clinical Overview of Fasciola | Liver Flukes [https://www.cdc.gov/liver-flukes/hcp/clinical-overview-fasciola/index.html]
- 65. Fasciola hepatica - by Robert Herriman - Outbreak News Today [https://outbreaknewstoday.substack.com/p/fasciola-hepatica]
- 66. Fasciola hepatica - Learn About Parasites [https://wcvm.usask.ca/learnaboutparasites/parasites/fasciola-hepatica.php]
- 67. Morphological and molecular characterization of Fasciola ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7930125/]
- 68. A major locus confers triclabendazole resistance in Fasciola ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9904461/]
- 69. Current Threat of Triclabendazole Resistance in Fasciola ... [https://pubmed.ncbi.nlm.nih.gov/27049013/]
- 70. Drug resistance in liver flukes [https://www.sciencedirect.com/science/article/pii/S2211320719301435]
- 71. Independent origins and non-parallel selection signatures ... [https://www.nature.com/articles/s41467-025-57796-5]
- 72. Describe the life cycle of Fasciola hepatica. - Steps [https://www.ck12.org/flexi/life-science/flatworms/describe-the-life-cycle-of-fasciola-hepatica/]
- 73. A major locus confers triclabendazole resistance in Fasciola ... [https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1011081]
- 74. Praziquantel-induced tegumental damage in vitro is ... [https://pubmed.ncbi.nlm.nih.gov/11197765/]
- 75. Parasite Viability - an overview [https://www.sciencedirect.com/topics/immunology-and-microbiology/parasite-viability]
- 76. Clinically applicable parasite viability assay for rapid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12326963/]
- 77. Anthelmintic drug discovery: target identification, screening ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7277699/]
- 78. Spatial analysis of Fasciola hepatica prevalence in sheep ... [https://www.sciencedirect.com/science/article/pii/S2405939025001753]
- 79. Enolase and 16.5‐kDa Tegument‐Associated Protein in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12127667/]
- 80. Life Cycle of Fasciola hepatica [https://www.msdmanuals.com/home/multimedia/image/life-cycle-of-fasciola-hepatica]
- 81. Chemical Control of Snail Vectors as an Integrated Part of a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11435910/]
- 82. Molluscicides against the snail-intermediate host of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8418967/]
- 83. Field trials to control schistosome intermediate hosts by the ... [https://www.sciencedirect.com/science/article/abs/pii/0001706X9390012Z]
- 84. Controversial aspects of the life cycle of Fasciola hepatica [https://www.sciencedirect.com/science/article/abs/pii/S0014489416301461]
- 85. Relationships between host species and morphometric ... [https://www.sciencedirect.com/science/article/abs/pii/S030440170100499X]
- 86. An overview of fasciolosis in human and cattle populations ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2025.1572946/full]
- 87. Natural Infection by Fasciola hepatica in Red Deer (Cervus ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12466639/]
- 88. Pilot study on the population genetics structure of Fasciola ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2025.1659523/full]
- 89. Pilot study on the population genetics structure of Fasciola ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12461863/]
- 90. Fasciola hepatica demonstrates high levels of genetic ... [https://www.sciencedirect.com/science/article/pii/S0020751916302569]
- 91. Deciphering the Fasciola hepatica Glycocode and Its ... [https://pubmed.ncbi.nlm.nih.gov/41008542/]
- 92. Glycans from Fasciola hepatica Modulate the Host Immune ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4697847/]
- 93. Fasciola hepatica Immune Regulates CD11c + Cells ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2017.00264/full]
- 94. Fasciola hepatica glycoconjugates immuneregulate ... [https://www.nature.com/articles/srep46748]
- 95. Fasciola Species Introgression: Just a Fluke or Something ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7575431/]
- 96. Paleobiogeographical origins of Fasciola hepatica and F. ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2022.990872/full]
- 97. Fasciola [https://en.wikipedia.org/wiki/Fasciola]
- 98. Identification and differentiation of Fasciola hepatica and ... [https://www.sciencedirect.com/science/article/abs/pii/S0014489409002707]
- 99. Molecular characterization of Fasciola hepatica in endemic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10288157/]
- 100. insights into genetic diversity, evolutionary dynamics, and ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2025.1577469/full]
- 101. Advancement in Diagnosis, Treatment, and Vaccines ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11357060/]
- 102. Review Protein–glycan engineering in vaccine design [https://www.sciencedirect.com/science/article/abs/pii/S0167779925002604]
- 103. Glycans of parasitic nematodes – from glycomes to novel ... [https://www.sciencedirect.com/science/article/pii/S0008621525000333]
- 104. Safety and efficacy of a novel glycoengineered ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12339958/]
- 105. Glycan masking in vaccine design: Targets, immunogens ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10076883/]
- 106. Deciphering the Fasciola hepatica Glycocode and Its ... [https://www.mdpi.com/2218-273X/15/9/1235]
- 107. Recent Progress in the Development of Liver Fluke and Blood ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7564142/]
- 108. Fasciolosis: pathogenesis, host-parasite interactions, and ... [https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2023.1270064/full]
- 109. The Fasciola hepatica genome: gene duplication and ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-015-0632-2]
- 110. Glycan Complexity and Heterogeneity of Glycoproteins in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10755494/]
- 111. TGF-β superfamily members from the helminth Fasciola ... [https://veterinaryresearch.biomedcentral.com/articles/10.1186/s13567-015-0167-2]