FA280 Digital Feces Analyzer vs Formalin-Ethyl Acetate Concentration Technique: A Comprehensive Technical Analysis for Biomedical Research
This comprehensive analysis compares the diagnostic performance, operational efficiency, and practical applications of the automated FA280 digital feces analyzer against the traditional formalin-ethyl acetate concentration technique (FECT) for intestinal parasite...
FA280 Digital Feces Analyzer vs Formalin-Ethyl Acetate Concentration Technique: A Comprehensive Technical Analysis for Biomedical Research
Abstract
This comprehensive analysis compares the diagnostic performance, operational efficiency, and practical applications of the automated FA280 digital feces analyzer against the traditional formalin-ethyl acetate concentration technique (FECT) for intestinal parasite detection. Through systematic evaluation of recent validation studies, we examine the technological foundations, methodological workflows, optimization strategies, and comparative performance metrics relevant to researchers, scientists, and drug development professionals. The analysis reveals that while the FA280 offers significant advantages in throughput, automation, and reduced technician dependency, it demonstrates variable sensitivity compared to FECT, particularly in low-intensity infections. This review synthesizes evidence from multiple recent studies to provide actionable insights for laboratory implementation and future diagnostic development in parasitology research.
Technological Foundations: Understanding Automated vs Traditional Parasitology Methods
Parasitology diagnostics are undergoing a significant transformation, moving from traditional, labor-intensive microscopic methods toward automated, digital solutions. For decades, the formalin-ethyl acetate concentration technique (FECT) has served as a cornerstone diagnostic method for intestinal parasitic infections, which affect billions of people worldwide and cause substantial health burdens including malnutrition, anemia, and impaired cognitive development [1]. The emergence of fully automated digital analyzers like the Orienter Model FA280 represents a technological shift aimed at addressing the limitations of conventional microscopy. This guide provides an objective comparison of these two methodologies, supported by experimental data, to inform researchers, scientists, and drug development professionals about their respective performances, applications, and limitations within modern parasitology.
Performance Comparison: FA280 vs. FECT
Detection Sensitivity and Agreement
Table 1: Comparison of Detection Sensitivity and Agreement Between FA280 and Reference Methods
| Study Reference | Sample Size | Comparator Method | Overall Agreement (κ statistic) | Key Findings |
|---|---|---|---|---|
| Boonyong et al. [1] | 200 fresh samples | FECT (with user audit) | 100% (κ = 1.00) | No significant difference in pairwise agreements (exact binomial test, P = 1) |
| Boonyong et al. [1] | 200 fresh samples | FECT (AI report only) | 75.5% (κ = 0.367) | Fair agreement; statistically significant difference from FECT (McNemar's test, P < 0.001) |
| Boonyong et al. [1] | 800 preserved samples | FECT (with user audit) | N/A (Significant difference) | FECT detected significantly more positives (McNemar's test, P < 0.001) |
| Community Study [2] | 1000 participants | Kato-Katz (KK) method | 96.8% (κ = 0.82) | No significant difference in positive rate (10.0% for both; P > 0.999) |
The diagnostic performance of the FA280 system varies considerably depending on the use of its artificial intelligence (AI) versus expert human oversight. When a skilled medical technologist conducts a user audit of the FA280's digital images, agreement with FECT can be perfect for fresh stool samples [1] [3]. However, the system's standalone AI report shows only fair agreement (κ = 0.367), indicating that the technology currently benefits substantially from human expert validation [1] [4].
In a large community-based study, the FA280 demonstrated strong agreement (κ = 0.82) with the Kato-Katz method for detecting Clonorchis sinensis, with no statistically significant difference in positive detection rates [2]. This suggests that the FA280's performance is robust for specific helminth infections in field settings. However, for broader parasite detection in preserved samples, FECT demonstrated higher sensitivity, likely because it processes a larger stool sample (2 g for FECT versus 0.5 g for FA280), increasing the probability of detecting low-burden infections [1].
Operational Characteristics and Cost-Analysis
Table 2: Operational and Economic Comparison of Diagnostic Methods
| Parameter | Direct Wet Smear | FECT | FA280 with User Audit |
|---|---|---|---|
| Weight of Stool Used | 0.2 g [5] | 2 g [5] | 0.5–1 g [5] |
| Technique | Manual [5] | Manual [5] | Automatic [5] |
| Process Simplicity | Less complicated [5] | More complicated [5] | Less complicated [5] |
| Processing Time per Sample | ~2 min [5] | 8-10 min [5] | ~2 min [5] |
| Parasite Observation Time | 5-10 min [5] | 5-10 min [5] | 3-5 min [5] |
| Result Storage | No [5] | No [5] | Yes, digital images [5] |
| Cost per Test | ~USD 0.25 [5] | ~USD 0.50 [5] | ~USD 2.00 [5] |
Operational metrics reveal the FA280's advantages in throughput and efficiency. The system can process a batch of 40 stool samples in a single run of approximately 30 minutes, significantly accelerating workflow compared to manual techniques [1]. This high-throughput capability, combined with reduced hands-on time and automated digital reporting, makes it suitable for laboratories with substantial sample volumes.
The primary operational disadvantage of the FA280 is its higher cost per test—approximately USD 2.00 compared to USD 0.50 for FECT [5]. This cost differential must be weighed against labor savings, reduced biohazard exposure, and digital record-keeping benefits. The FA280 also reduces laboratory contamination risk by automating sample handling [1].
Experimental Protocols and Workflows
Formalin-Ethyl Acetate Concentration Technique (FECT) Protocol
The FECT method, as a standard sedimentation technique, leverages differences in specific gravity to concentrate parasitic elements from fecal debris [6]. The detailed protocol is as follows:
- Sample Preparation: Mix 2 g of stool sample with 10 ml of 10% formalin [1].
- Filtration: Strain the fecal suspension through a 2-layer gauze into a 15-ml conical centrifuge tube [1] [6]. Add saline or formalin to bring the volume to 15 ml [6].
- Centrifugation: Centrifuge at 500 × g for 10 minutes. Decant the supernatant [6].
- Solvent Addition: Add 10 ml of 10% formalin to the sediment and mix thoroughly. Add 4 ml of ethyl acetate, stopper the tube, and shake vigorously for 30 seconds [6].
- Second Centrifugation: Centrifuge at 500 × g for 10 minutes [6]. The process forms four layers: ethyl acetate, debris plug, formalin, and sediment [6].
- Examination: Free the debris plug, decant the top layers, and use a cotton-tipped applicator to clean the tube sides. The final sediment is pipetted onto a glass slide for microscopic examination [1] [6].
This method's effectiveness stems from processing larger stool quantities, but it requires skilled technicians for both processing and microscopic interpretation [1].
FA280 Automated Digital Feces Analysis Protocol
The FA280 employs a fully automated workflow based on a simple sedimentation principle, integrating digital imaging and AI analysis [1]:
Figure 1: The FA280 automated digital feces analysis workflow.
The process begins with sample loading, where approximately 0.5 g of stool is placed in a filtered collection tube [1] [2]. The system then performs pneumatic mixing with a diluent to ensure homogeneity [1]. A high-resolution camera captures macroscopic attributes including color, shape, and consistency [1] [2]. The microscope unit automatically captures high-resolution images through multi-field tomography at different magnifications [1]. Finally, the software analyzes images using an AI program to generate a report, with an optional user audit by a skilled technologist for verification [1] [3].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagents and Materials for Parasitological Diagnosis
| Item | Function | Application in Protocols |
|---|---|---|
| 10% Formalin | Preservative and fixative; maintains structural integrity of parasites. | Used in FECT for initial sample fixation and sedimentation [1] [6]. |
| Ethyl Acetate | Solvent for extraction and purification; removes fats and debris. | Used in FECT as a safer alternative to diethyl ether [6] [7]. |
| Formalin-Based Fixatives | Preserve parasitic morphology for later analysis. | Used for storing stool samples before processing with FECT or FA280 [1]. |
| Alcorfix (Alcohol-Based Fixative) | Formalin-free fixative integrated in closed concentration systems. | Used in Parasep SF technique; eliminates formalin hazards [8]. |
| Ethyl Acetate Substitutes (Tween, Acetone) | Alternative solvents; more stable and safer than ether. | Can be used in modified concentration techniques (FTC, FAC) [7]. |
| Glycerol-Malachite Green | Staining and clearing agent for microscopic visualization. | Used in Kato-Katz method for preparing slides for helminth egg counting [2]. |
This toolkit comprises critical reagents that facilitate both traditional and modern diagnostic approaches. The shift toward solvent-free and formalin-free alternatives like Alcorfix reflects an ongoing effort to improve laboratory safety without compromising diagnostic accuracy [8]. Similarly, substitutes for ethyl acetate, such as Tween and acetone, offer more stable and less flammable options for concentration procedures [7].
The evolution from manual microscopy to digital automation in parasitological diagnosis presents researchers and laboratory professionals with meaningful choices between established and emerging technologies. The FECT method remains a highly sensitive reference technique, particularly valuable for detecting low-intensity infections and in research settings where cost is a primary consideration. In contrast, the FA280 automated analyzer offers substantial advantages in throughput, standardization, and operational safety, making it suitable for clinical laboratories with high sample volumes, despite its higher per-test cost and current reliance on expert auditing for optimal accuracy.
The future of parasitological diagnosis will likely involve continued refinement of AI algorithms to improve autonomous detection capabilities, along with the development of integrated systems that combine the sensitivity of traditional concentration methods with the efficiency of digital automation. This technological progression promises to enhance global capacity for diagnosing and monitoring parasitic infections, ultimately contributing to improved public health outcomes.
Intestinal parasitic infections remain a significant global health challenge, particularly in tropical regions, affecting billions of people and causing substantial morbidity including malnutrition, anemia, and impaired cognitive development [1]. The accurate diagnosis of these infections relies heavily on microscopic examination of stool samples, a process that has evolved considerably since the early 20th century with the development of various concentration techniques to improve detection sensitivity [9]. Among these methods, the Formalin-Ethyl Acetate Concentration Technique (FECT) has emerged as a widely adopted standard in clinical parasitology due to its effectiveness in recovering diverse parasitic forms [6].
This guide examines the core sedimentation dynamics and parasite recovery mechanisms of FECT while contextualizing its performance against emerging automated technologies, particularly the FA280 fully automatic digital feces analyzer. As diagnostic paradigms shift toward automation and digital imaging, understanding the fundamental principles, advantages, and limitations of established techniques like FECT becomes crucial for evaluating new diagnostic platforms and their appropriate application in both clinical and research settings [3] [2].
Core Principles and Sedimentation Dynamics of FECT
The Formalin-Ethyl Acetate Concentration Technique operates on the principle of differential sedimentation, leveraging variations in specific gravity between parasitic structures and fecal debris to achieve separation and concentration. This process enhances detection sensitivity by increasing the density of diagnostically relevant elements in the final sediment [6] [9].
Specific Gravity and Sedimentation Dynamics
The effectiveness of FECT relies on carefully engineered specific gravity relationships between the solution and parasitic elements. Formalin (10%) serves as both a preservative and a suspension medium with a specific gravity of approximately 1.03, which is sufficient to maintain most parasitic cysts, ova, and oocysts in suspension while allowing heavier fecal debris to sediment more rapidly [9]. Ethyl acetate, with a specific gravity of 0.89, functions as an extractant that forms a distinct upper layer during centrifugation, effectively trapping lighter debris and lipids at the interface while parasitic structures sediment through the formalin layer [6].
The sedimentation dynamics follow Stokes' law principles, where the terminal velocity of particles in a fluid medium depends on their size, density difference with the medium, and the applied centrifugal force. Parasitic structures, with their characteristic specific gravities (typically ranging from 1.05 to 1.20), sediment efficiently under standardized centrifugal forces of 500 × g for 10 minutes as recommended by CDC protocols [6]. This differential sedimentation results in a cleaner sediment enriched with parasites while simultaneously reducing obscuring debris that could complicate microscopic identification [8].
Table 1: Specific Gravity Ranges of Common Parasitic Structures in FECT
| Parasitic Structure | Specific Gravity Range | Sedimentation Behavior in FECT |
|---|---|---|
| Helminth eggs (Ascaris, Trichuris) | 1.10 - 1.20 | Excellent sedimentation with minimal loss |
| Protozoan cysts (Giardia, Entamoeba) | 1.05 - 1.15 | Good sedimentation, some fragile forms may be affected |
| Coccidian oocysts (Cryptosporidium) | 1.04 - 1.08 | Moderate sedimentation, may require specialized stains |
| Nematode larvae (Strongyloides) | 1.05 - 1.10 | Variable recovery, alternative methods often preferred |
Key Procedural Steps and Their Functions
The FECT procedure consists of several critical steps, each contributing to the overall efficiency of parasite recovery:
Sample Homogenization and Filtration: Approximately 2g of stool is emulsified in 10% formalin and strained through gauze or a specialized sieve (450-500μm pore size). This initial step removes large particulate matter while creating a uniform suspension of parasitic structures [6] [8].
Primary Sedimentation: The filtered suspension undergoes initial centrifugation at 500 × g for 10 minutes, allowing dense debris to form a firm pellet while parasitic structures remain in suspension or form a looser layer above the debris [6].
Solvent Extraction: After decanting the supernatant, the sediment is resuspended in formalin, and ethyl acetate is added. The tube is vigorously shaken to ensure thorough mixing, facilitating the dissolution of lipids and trapping of light debris at the solvent interface [6] [7].
Final Concentration: A second centrifugation step separates the mixture into distinct layers: an ethyl acetate plug containing extracted debris at the top, a formalin layer, and the concentrated parasitic sediment at the bottom. The debris plug is dislodged and removed along with the solvent layers, leaving a cleaned sediment for microscopic examination [6].
The following diagram illustrates the step-by-step FECT workflow and the separation dynamics during centrifugation:
Comparative Experimental Data: FECT vs. FA280
Recent studies have systematically compared the performance characteristics of traditional FECT with the automated FA280 digital feces analyzer, revealing significant differences in sensitivity, operational parameters, and diagnostic efficiency.
Parasite Detection Agreement
A comprehensive study examining 200 fresh stool samples found statistically significant differences in detection rates between FECT and the FA280's fully automated AI interpretation mode (McNemar's test, P < 0.001). The agreement for species identification between FA280 with AI report and FECT showed only fair agreement (overall agreement = 75.5%, kappa [κ] = 0.367) [3] [1]. However, when a skilled technologist conducted a user audit of the FA280 findings, the agreement with FECT improved dramatically to perfect agreement (overall agreement = 100%, κ = 1.00) [3] [1].
In a larger study involving 800 preserved stool samples, FECT detected significantly more positive samples for parasites than the FA280 with user audit (McNemar's test, P < 0.001) [3]. This sensitivity advantage was attributed to the substantially larger stool sample processed by FECT (2g) compared to the FA280 (0.5-1g), increasing the probability of detecting low-intensity infections [3] [5].
Table 2: Performance Comparison Between FECT and FA280 in Parasite Detection
| Performance Metric | FECT | FA280 with AI | FA280 with User Audit |
|---|---|---|---|
| Sample Size Processed | 2g [5] | 0.5-1g [5] | 0.5-1g [5] |
| Overall Agreement with FECT | Reference | 75.5% [3] | 100% (fresh samples) [3] |
| Kappa Statistic | Reference | 0.367 (fair agreement) [3] | 1.00 (perfect agreement) [3] |
| Helminth Identification Agreement | Reference | Not reported | κ = 0.857 (strong agreement) [3] |
| Protozoa Identification Agreement | Reference | Not reported | κ = 1.00 (perfect agreement) [3] |
| Processing Time | 8-10 minutes/sample [5] | 2 minutes/sample [5] | 2 minutes/sample [5] |
Sample Throughput and Operational Efficiency
The FA280 demonstrates significant advantages in processing speed and automation. While FECT requires 8-10 minutes of hands-on technician time per sample, the FA280 processes samples in approximately 2 minutes each [5]. This efficiency gain becomes particularly substantial in high-volume settings, with the FA280 capable of processing batches of 40 samples in approximately 30 minutes [1].
However, this operational efficiency comes with economic tradeoffs. The cost per test for FECT is approximately USD 0.50, compared to USD 2.00 for the FA280 [5]. Additionally, FECT maintains superior sensitivity for low-intensity infections due to its larger sample size, whereas the FA280's smaller sample volume (0.5-1g) may limit its detection capability for parasites present in low numbers [3].
Technical Protocols and Methodologies
Standard FECT Protocol
The following detailed methodology outlines the standard FECT procedure as recommended by the CDC and employed in comparative studies [6]:
Sample Preparation: Emulsify 2g of fresh or preserved stool in 10mL of 10% formalin in a 15mL conical centrifuge tube. For preserved specimens, ensure proper fixation in 10% formalin at a 3:1 preservative-to-fecal material ratio [6] [9].
Filtration: Strain the fecal suspension through wet cheesecloth-type gauze or a specialized sieve (450-500μm pore size) into a clean 15mL conical centrifuge tube. Add additional 10% formalin through the debris on the gauze to bring the total volume to 15mL [6] [8].
Primary Centrifugation: Centrifuge at 500 × g for 10 minutes. Decant the supernatant carefully without disturbing the sediment [6].
Solvent Extraction: Resuspend the sediment in 10mL of 10% formalin. Add 4mL of ethyl acetate, stopper the tube securely, and shake vigorously in an inverted position for 30 seconds. Carefully remove the stopper to release pressure [6].
Final Centrifugation: Recentrifuge at 500 × g for 10 minutes. Four distinct layers will form: ethyl acetate at the top, a debris plug, formalin, and sediment at the bottom [6].
Sediment Recovery: Free the debris plug from the tube walls using an applicator stick. Decant the top three layers, leaving the sediment. Use a cotton-tipped applicator to remove residual debris from the tube sides [6].
Microscopic Examination: Resuspend the final sediment in a few drops of 10% formalin. Prepare wet mounts with saline and iodine for microscopic examination at 100× and 400× magnification [6] [8].
FA280 Automated Protocol
The FA280 employs an automated sedimentation and concentration approach with the following workflow [2] [1]:
Sample Loading: Approximately 0.5g of stool is placed in a filtered sample collection tube containing diluent. A batch of 40 samples can be loaded simultaneously [1].
Automated Processing: The instrument performs automatic sample mixing through high-frequency pneumatic agitation, followed by intelligent sample dilution and transfer to the examination system [1].
Digital Imaging: High-resolution cameras capture sample characteristics and color attributes. The microscope unit automatically captures multi-field tomographic images using high- and low-power objectives [1].
AI Analysis: Acquired digital images are automatically analyzed by artificial intelligence software that identifies and classifies parasitic structures based on morphological parameters [3] [1].
Result Verification: A user audit function allows skilled technologists to review and verify AI-generated findings, with digital archiving of all images for quality control and documentation [3] [1].
Research Reagent Solutions and Essential Materials
Successful implementation of parasitological diagnostic methods requires specific reagents and materials optimized for parasite recovery and morphological preservation.
Table 3: Essential Research Reagents for Stool Parasitology Methods
| Reagent/Material | Function | Application Notes |
|---|---|---|
| 10% Formalin | Preservative and suspension medium; fixes parasitic structures | Maintains morphology but destroys motile trophozoites; specific gravity ~1.03 [6] [9] |
| Ethyl Accetate | Solvent extractant; removes lipids and traps debris | Less flammable alternative to ether; specific gravity 0.89; forms distinct upper layer [6] [7] |
| Merthiolate-Iodine-Formalin (MIF) | Multipurpose fixative and stain | Simultaneously preserves and stains parasitic structures; suitable for quantitative methods [10] |
| Formalin-Tween | Alternative concentration reagent | Higher sensitivity for helminth eggs (71.7%) compared to FECT (55.8%) [7] |
| Formalin-Acetone | Alternative concentration reagent | Comparable to Formalin-Tween for helminth recovery (70% sensitivity) [7] |
| Alcorfix | Alcohol-based fixative in Parasep system | Formalin-free alternative; integrated filtration system reduces processing time [8] |
| Orienter FA280 Diluent | Proprietary suspension medium | Automated mixing and dilution; optimized for digital imaging [3] [1] |
Discussion and Comparative Analysis
Sedimentation Efficiency and Parasite Recovery Mechanisms
The fundamental mechanism of FECT relies on differential sedimentation rates between parasitic structures and fecal debris under standardized centrifugal forces. This process effectively concentrates diagnostically relevant organisms while reducing obscuring material that complicates microscopic examination [9]. The specific gravity parameters of the formalin-ethyl acetate system (ranging from 0.89 for ethyl acetate to 1.03 for formalin) create an optimal density gradient for recovering most clinically significant helminth eggs and protozoan cysts, which typically have specific gravities between 1.05 and 1.20 [9].
Comparative studies demonstrate that FECT consistently outperforms automated systems in recovery efficiency for low-intensity infections, primarily due to its larger sample size (2g vs. 0.5-1g in FA280) [3]. This difference becomes particularly significant in epidemiological surveys where infection intensities may be low, as the probability of detection correlates directly with the sample volume examined [3] [9]. However, the FA280's automated digital imaging offers advantages in standardization and documentation, creating permanent digital records that can be re-evaluated and used for quality assurance and training purposes [1].
Operational Considerations and Workflow Integration
From an operational perspective, FECT and the FA280 represent different paradigms in laboratory workflow. FECT requires substantial technical expertise, with processing times of 8-10 minutes per sample and additional microscopic examination time of 5-10 minutes [5]. In contrast, the FA280 reduces hands-on time to approximately 2 minutes per sample with examination time of 3-5 minutes, while also minimizing biohazard exposure through automated processing [5] [1].
The economic tradeoffs between these approaches are significant. While FECT costs approximately USD 0.50 per test compared to USD 2.00 for the FA280, the automated system offers substantially higher throughput potential [5]. This makes the FA280 particularly suitable for high-volume settings where technician time represents a major constraint, while FECT remains a cost-effective option for laboratories with limited resources or lower testing volumes [3] [2].
Limitations and Methodological Constraints
Both techniques present important limitations that influence their appropriate application. FECT's multi-step manual procedure introduces opportunities for technical error and increases biohazard exposure to laboratory personnel [3] [1]. The technique also shows variable sensitivity for certain parasites, particularly Strongyloides stercoralis larvae, where agar plate culture remains significantly more sensitive, especially for low-burden infections [10].
The FA280's limitations include higher per-test costs, reduced sensitivity for low-intensity infections due to smaller sample volume, and dependency on user verification for optimal accuracy, as the AI algorithm alone showed only fair agreement with FECT (κ = 0.367) [3]. Additionally, the system requires significant capital investment and technical infrastructure that may not be feasible in resource-limited settings where parasitic infections are most prevalent [2].
The Formalin-Ethyl Acetate Concentration Technique remains a cornerstone of parasitological diagnosis, with sedimentation dynamics based on well-established principles of differential specific gravity and centrifugal force. Its robust performance, particularly for detecting low-intensity infections, maintains its relevance in both clinical and research contexts. The FA280 automated fecal analyzer represents a significant advancement in diagnostic efficiency and standardization, offering substantial benefits in throughput, technician time reduction, and documentation capabilities.
The choice between these methodologies depends heavily on specific application requirements. For reference laboratories, epidemiological surveys, and settings where maximum sensitivity is paramount, FECT maintains distinct advantages. For high-volume clinical laboratories prioritizing workflow efficiency, standardized results, and reduced biohazard exposure, the FA280 offers compelling benefits. Future developments in automated digital morphology recognition, particularly improvements in AI algorithm training and sample processing methods, will likely narrow current sensitivity gaps while preserving the operational advantages of automation.
Understanding the core principles of FECT's sedimentation dynamics and parasite recovery mechanisms provides an essential foundation for evaluating both current and emerging diagnostic platforms, enabling researchers and clinicians to make informed decisions about method selection and implementation in diverse diagnostic contexts.
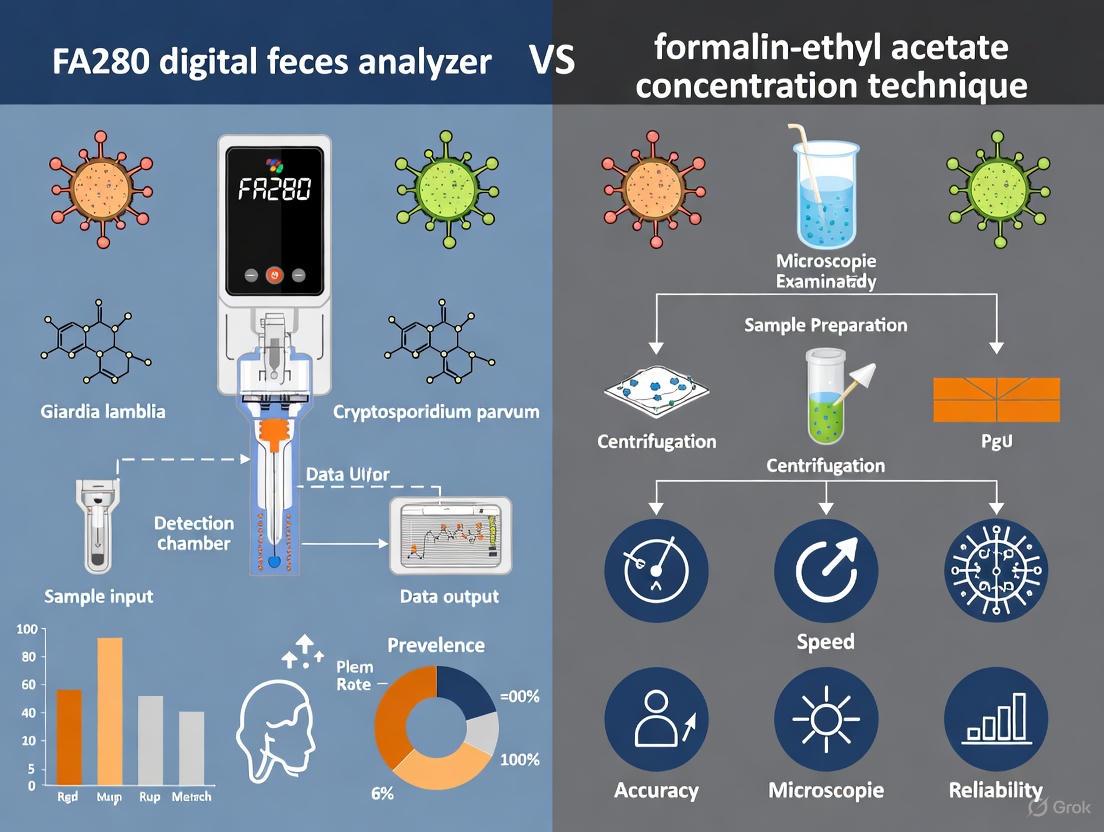
The Orienter Model FA280 represents a significant technological advancement in the diagnosis of intestinal parasitic infections. This fully automatic digital feces analyzer is engineered to address the critical limitations of traditional microscopic examination methods, which are notoriously time-consuming, labor-intensive, and heavily dependent on the expertise of trained microscopists [3]. The system integrates multiple sophisticated technologies into a unified platform, including automated sample processing, high-resolution digital imaging, and artificial intelligence (AI)-driven pattern recognition for parasite identification [11]. This architectural integration enables the FA280 to transform stool examination from a manual, subjective process into an automated, standardized, and high-throughput procedure suitable for both clinical laboratories and large-scale epidemiological surveys [2].
The clinical necessity for such a system is well-established within the medical and research communities. Intestinal parasitic infections constitute a substantial global health burden, associated with serious sequelae including malnutrition, anemia, impaired growth and cognitive development, and alterations in microbiota composition and immune responses [3]. Accurate diagnosis through stool examination remains fundamental to controlling these infections, yet traditional methods like the formalin-ethyl acetate concentration technique (FECT) and Kato-Katz (KK) method present significant operational challenges [3] [2]. The FA280 system emerges as a technological solution that balances diagnostic accuracy with operational efficiency, offering a standardized approach to parasite detection while reducing laboratory contamination and technical variability [3].
System Architecture and Technological Components
Automated Sample Processing Subsystem
The FA280 incorporates a comprehensive automated sample processing system that begins with intelligent specimen handling. The system utilizes a fully sealed design that effectively contains odors and prevents leakage, addressing a significant concern in traditional stool analysis laboratories [11]. The process initiates with intelligent sample dilution, where the system automatically determines the optimal dilution ratio based on initial specimen characteristics. This is followed by high-frequency pneumatic mixing, which ensures homogeneous suspension of stool samples without manual intervention, significantly improving the consistency of sample preparation compared to manual methods [11].
A critical innovation in the FA280's sample processing architecture is its automatic sedimentation and concentration technology [2]. When approximately 0.5 grams of fecal sample is placed in a filtered collection tube, the system employs specialized technology to concentrate parasitic elements while excluding excessive debris [2]. This process is enhanced by multi-channel counting cells that facilitate time-division sedimentation scanning and mobile window scanning, ensuring comprehensive analysis of the concentrated sample [11]. The entire process occurs within a closed system, minimizing the risk of laboratory contamination and reducing technician exposure to potentially infectious materials [3].
Digital Imaging and Microscopy Subsystem
The FA280 incorporates an advanced digital imaging system specifically engineered for parasitic morphology analysis. At the core of this subsystem is a customized optical design with an optimized light path and LED illumination source that ensures consistent imaging quality [11]. The system is equipped with a built-in 5-megapixel digital acquisition system featuring a global shutter, which captures high-resolution images without motion distortion [11]. A particularly innovative feature is the focal length memory function (protected by patent), which enables the system to automatically maintain optimal focus throughout the imaging process [11].
The FA280 employs a "Microscopic CT SCAN Method" where each field of view image is synthesized from five distinct tomographic images, effectively simulating the fine-tuning capability of a manual microscope [11]. This multi-layer imaging approach ensures that each visual field is captured with maximal clarity, which is crucial for accurate identification of parasitic structures. The system utilizes multiple objective lenses with specialized configurations: a 10X lens with adjustable field of view and three focal layers for parasite egg tracking, and a 40X lens with adjustable field of view and five focal layers for detailed analysis of other visible elements [11]. This comprehensive imaging strategy enables the system to capture morphological details equivalent to or exceeding what can be observed through conventional microscopy.
AI-Based Pattern Recognition Engine
The artificial intelligence component of the FA280 system represents a sophisticated pattern recognition engine trained to identify a wide spectrum of parasitic elements. The AI algorithm is capable of automatically detecting and classifying various helminth eggs (including Ascaris lumbricoides, hookworm, Trichuris trichiura, Taenia spp., and Clonorchis sinensis), larvae (such as Strongyloides stercoralis), and protozoan cysts and trophozoites (including Giardia lamblia, Entamoeba histolytica, and Blastocystis hominis) [11]. The system employs a locating and tracking algorithm that identifies potential parasitic structures within digital images and subjects them to further analysis to confirm identification [11].
A crucial architectural feature is the system's continuous learning capability. Through software updates, the AI engine can be upgraded to recognize additional parasitic species and improve its identification accuracy for existing targets [11]. When the AI system encounters parasites not yet incorporated into its automatic identification library, it can still capture and present images for technologist review, thus expanding its utility beyond its predefined classification capabilities [11]. This adaptive architecture ensures that the system maintains diagnostic relevance as parasitological knowledge advances and new diagnostic challenges emerge.
Comparative Performance Analysis: FA280 vs. Traditional Methods
Methodological Protocols for Performance Validation
The evaluation of the FA280 system against established manual techniques has been conducted through rigorous comparative studies employing standardized methodological protocols. In one significant study comparing the FA280 to the Formalin-Ethyl Acetate Concentration Technique (FECT), researchers analyzed 200 fresh stool samples using both methods [3]. For the FA280 arm, approximately 0.5g of stool was placed in a filtered collection tube, with the system automatically performing dilution, mixing, and sedimentation before digital imaging and AI analysis [3]. The FECT protocol utilized 2g of stool, employing formalin fixation, ethyl acetate extraction, and centrifugation before manual microscopic examination [3]. A crucial quality control measure incorporated in the FA280 protocol was the user audit, where a skilled medical technologist reviewed the AI-generated findings [3].
In a separate study focusing on clonorchiasis diagnosis, researchers employed a cross-sectional design with 1,000 participants to compare the FA280 against the Kato-Katz (KK) method [2]. For the KK method, two smears were prepared per fecal sample using 41.7mg of sieved stool per smear on a glass slide, with cellophane covers soaked in glycerol and malachite green [2]. Four experienced technicians examined the smears under a microscope, with quality control measures including re-examination of 10 stool samples from each study village by a professional staff member [2]. This systematic approach ensured comparable results between the novel automated system and established manual techniques.
Diagnostic Performance and Agreement Metrics
Table 1: Comparison of Diagnostic Performance Between FA280 and Reference Methods
| Performance Parameter | FA280 vs. FECT (Fresh Samples) | FA280 vs. FECT (Preserved Samples) | FA280 vs. Kato-Katz (Clonorchiasis) |
|---|---|---|---|
| Overall Agreement | 100% (with user audit) [3] | N/A | 96.8% [2] |
| Kappa Statistic (κ) | 1.00 (with user audit) [3] | Helminths: κ = 0.857 [3] | κ = 0.82 [2] |
| Statistical Significance | P = 1 (not significant) [3] | P < 0.001 [3] | P > 0.999 (not significant) [2] |
| AI-Only Agreement | 75.5% (κ = 0.367) [3] | N/A | N/A |
The comparative performance data reveals a complex picture of the FA280's diagnostic capabilities. When combined with expert user audit, the FA280 demonstrates perfect agreement with FECT for fresh stool samples (κ = 1.00), indicating virtually identical performance to the conventional method under optimal conditions [3]. Similarly, for clonorchiasis diagnosis, the FA280 shows no statistically significant difference from the KK method (P > 0.999) with strong agreement (κ = 0.82) [2]. However, the AI component alone shows only fair agreement (κ = 0.367) with FECT, highlighting the continued importance of human expertise in the diagnostic process [3].
For preserved stool samples, FECT detected significantly more positive samples than the FA280 with user audit (P < 0.001), a discrepancy potentially attributable to the larger stool sample size used in FECT (2g versus 0.5g in FA280) [3]. Despite this limitation, the FA280 maintained strong agreement with FECT for helminth species identification (κ = 0.857) and perfect agreement for protozoa (κ = 1.00) [3]. The system performs particularly well in detecting high-intensity infections, with significantly higher agreement rates in high infection intensity groups compared to low-intensity infections (P < 0.05) [2].
Operational Parameters and Practical Considerations
Table 2: Comparison of Operational Parameters Between Stool Examination Methods
| Operational Parameter | Direct Wet Smear | FECT | FA280 with User Audit |
|---|---|---|---|
| Sample Weight Required | 0.2g [5] | 2g [5] | 0.5-1g [5] |
| Processing Time per Sample | 2 minutes [5] | 8-10 minutes [5] | 2 minutes [5] |
| Parasite Observation Time | 5-10 minutes [5] | 5-10 minutes [5] | 3-5 minutes [5] |
| Technician Expertise Required | High [5] | High [5] | Moderate [5] |
| Result Storage Capability | No [5] | No [5] | Yes (digital images) [5] |
| Cost per Test | USD 0.25 [5] | USD 0.50 [5] | USD 2.00 [5] |
From an operational perspective, the FA280 system offers several distinct advantages over traditional methods. The significantly reduced processing time (2 minutes versus 8-10 minutes for FECT) enables higher throughput capacity, making the system particularly valuable in settings with large sample volumes [5]. The reduced observation time (3-5 minutes versus 5-10 minutes for manual methods) further enhances efficiency, though this advantage is partially offset by the need for user audit of AI-generated findings [5]. The system's digital image storage capability provides a permanent record of findings, facilitating quality control, second opinions, and retrospective analysis—features unavailable with conventional microscopy [5].
The principal operational disadvantages of the FA280 relate to cost considerations and sensitivity limitations. At USD 2.00 per test, the FA280 is substantially more expensive than both direct wet smear (USD 0.25) and FECT (USD 0.50) [5]. Additionally, the system's lower sensitivity compared to FECT, particularly for preserved samples and low-intensity infections, represents a significant diagnostic limitation [3]. The FA280 also requires technical infrastructure and training for operation and maintenance, considerations that may affect its implementation in resource-limited settings where parasitic infections are most prevalent.
Research Reagent Solutions and Essential Materials
Table 3: Key Research Reagents and Materials for FA280 and Comparative Methods
| Reagent/Material | Application | Function | Method |
|---|---|---|---|
| Filtered Sample Collection Tubes | Sample Preparation | Containment and initial filtration of stool specimens | FA280 [2] |
| Intelligent Diluent | Sample Processing | Optimal dilution for microscopic analysis | FA280 [11] |
| 10% Formalin | Sample Preservation | Fixation of parasitic elements for later analysis | FECT [3] |
| Ethyl Acetate | Parasite Concentration | Solvent extraction for separation of parasitic elements | FECT [3] |
| Glycerol-Malachite Green Solution | Slide Preparation | Clearing agent for visualization of parasitic structures | Kato-Katz [2] |
| Cellophane Covers | Slide Preparation | Membrane for uniform smear distribution | Kato-Katz [2] |
The effective implementation of stool examination methods, whether automated or manual, requires specific research reagents and materials each serving distinct functions in the diagnostic process. The FA280 system utilizes proprietary filtered sample collection tubes that serve as the primary container for specimen intake while providing initial filtration of coarse particulate matter [2]. The system's intelligent diluent is specifically formulated to achieve optimal consistency for automated digital imaging while preserving morphological features of parasitic elements [11].
For comparative manual methods, 10% formalin serves as a fixative preservative that maintains parasite morphology while reducing infectious potential [3]. Ethyl acetate functions as an extraction solvent in FECT, facilitating the separation of parasitic elements from fecal debris through centrifugation [3]. In the Kato-Katz method, glycerol-malachite green solution serves as a clearing agent that gradually transparentizes the fecal smear, enhancing visualization of parasitic structures, while cellophane covers provide a standardized medium for creating uniform smear thickness [2]. Each of these reagents requires quality control measures to ensure consistent performance across different batches and operators.
Discussion and Research Implications
The architectural analysis of the FA280 system reveals a sophisticated integration of automation, digital imaging, and artificial intelligence that collectively address significant limitations of conventional stool examination methods. The system's fully automated processing pipeline reduces technical variability and minimizes potential for laboratory contamination [3]. Its high-resolution digital imaging capabilities, particularly the multi-layer "Microscopic CT SCAN" approach, capture morphological detail comparable to manual microscopy while creating permanent digital records [11]. The AI-based pattern recognition engine provides initial screening that, while requiring expert verification, standardizes the initial detection process and reduces technologist fatigue [3] [11].
From a research perspective, the FA280 system offers particular utility in large-scale epidemiological surveys where processing efficiency and standardized results are prioritized over maximal sensitivity [2]. The system's digital archiving capability enables the creation of image libraries that can support training, quality assurance, and further refinement of AI algorithms [5]. However, the technology's reduced sensitivity for low-intensity infections and higher cost per test currently limit its application as a complete replacement for conventional methods in all scenarios [3] [5].
Future research directions should focus on enhancing AI sensitivity through expanded training datasets, particularly for low-prevalence parasites and low-intensity infections. Technical innovations addressing the sample size limitation (0.5g versus 2g in FECT) could significantly improve detection rates without compromising automation benefits [3]. Additionally, cost-reduction strategies through technological scaling and workflow optimization could enhance accessibility in resource-limited settings where parasitic infections are most prevalent. The FA280 architecture represents a significant milestone in the evolution of parasitic diagnostics, establishing a technological foundation upon which future generations of automated detection systems will undoubtedly build.
{# Introduction}
The diagnosis of intestinal parasitic infections remains a critical public health challenge, relying heavily on the microscopic examination of stool samples. For decades, the Formalin-Ethyl Acetate Concentration Technique (FECT) has been a cornerstone method in clinical and research laboratories, prized for its diagnostic accuracy [12]. However, this manual technique is labor-intensive, time-consuming, and requires significant expertise [1] [13]. The recent development of fully automated digital feces analyzers, such as the Orienter Model FA280, promises a paradigm shift by integrating artificial intelligence (AI) and automation to address these limitations [1]. This guide provides a detailed, objective comparison of the technical specifications, performance data, and infrastructure requirements of the FA280 and the traditional FECT, offering researchers and scientists a clear framework for evaluating these diagnostic approaches.
{## Technical Specifications and Performance Data}
A direct comparison of the core technical and operational characteristics of the FA280 and FECT reveals fundamental differences in their approach to parasite detection. The table below summarizes these key specifications.
{Table: Comparative Technical Specifications of FA280 and FECT}
| Feature | FA280 Fully Automatic Digital Feces Analyzer | Formalin-Ethyl Acetate Concentration Technique (FECT) |
|---|---|---|
| Basic Principle | Automated sedimentation & AI-based digital imaging [1] | Manual centrifugation & chemical concentration [6] [12] |
| Sample Throughput | High; batch of 40 samples per ~30-minute run [1] | Low; manual processing of individual samples |
| Sample Volume | ~0.5 g [1] [2] | ~0.5 g to 2 g [1] [12] |
| Degree of Automation | Fully automated from sampling to AI report generation [1] | Fully manual; requires extensive hands-on steps [6] |
| Hands-On Time | Significantly reduced | Substantial (strainin, centrifuging, decanting, microscopy) [6] |
| Key Infrastructure | Dedicated automated analyzer, proprietary software, computer [1] | Centrifuge, microscope, fume hood, consumables (tubes, formalin, ethyl acetate) [6] |
| Detection Agreement with Reference Methods | Strong to perfect for helminths with user audit (κ = 0.857-1.00) [1] [2] | Considered a reference standard; superior detection for some helminths vs. crude methods [12] |
| Relative Cost per Test | Higher cost per test [1] | Lower material cost, but higher labor cost [1] |
{## Detailed Experimental Protocols}
To ensure the reproducibility of the comparative data cited in this guide, the following sections detail the standard operating procedures for both the FECT and the FA280, as described in the literature.
{### Formalin-Ethyl Acetate Concentration Technique (FECT)}
The FECT is a sedimentation-based concentration method designed to separate parasites from fecal debris. The protocol below is adapted from established procedures [6] [1] [12].
- Sample Preparation: Approximately 0.5 to 2 grams of stool is emulsified in 10 mL of 10% formalin [1] [12].
- Filtration and Straining: The fecal suspension is strained through wetted gauze or a specialized sieve into a 15 mL conical centrifuge tube to remove large particulate matter [6] [12].
- First Centrifugation: The tube is centrifuged at 500 × g for 5-10 minutes. The supernatant is decanted [6] [1].
- Resuspension and Solvent Addition: The sediment is resuspended in 10 mL of 10% formalin. Then, 4 mL of ethyl acetate is added to the tube [6] [1].
- Vigorous Mixing: The tube is stoppered and shaken vigorously for 30 seconds to extract fats and debris into the ethyl acetate layer [6].
- Second Centrifugation: The tube is centrifuged again at 500 × g for 5-10 minutes. This step results in a four-layered structure: a plug of debris at the top (ethyl acetate and debris), a formalin layer, a sediment layer containing the concentrated parasites, and a small amount of residual formalin [6] [1].
- Supernatant Removal: The plug of debris is freed with an applicator stick, and the top layers of supernatant are carefully decanted. A cotton-tipped applicator may be used to wipe debris from the tube walls [6].
- Microscopic Examination: The final sediment is resuspended in a small volume of formalin or saline. A wet mount is prepared from this concentrate and examined under a light microscope by a trained technologist for the identification of parasites [6] [12].
{### FA280 Fully Automatic Digital Feces Analyzer Protocol}
The FA280 automates the entire process from sample preparation to analysis. The following workflow is based on manufacturer and research descriptions [1].
- Sample Loading: Approximately 0.5 g of a fecal sample is placed in a proprietary filtered sample collection tube [1] [2].
- Batch Loading and Tracking: A batch of up to 40 sample tubes is loaded into the analyzer's automatic in-sample unit, which uses a track-type carrier [1].
- Automated Processing:
- Pneumatic Mixing and Dilution: The sampling unit uses a pneumatic system to thoroughly mix the sample with a diluent [1].
- Macroscopic Imaging: A high-resolution camera captures images of the sample's character and color [1].
- Microscopic Imaging: The microscope unit, equipped with high- and low-power objectives, automatically performs multifield tomography to capture high-resolution images of the prepared sample at different magnifications [1].
- AI Analysis and Reporting: The captured digital images are automatically analyzed by the instrument's integrated AI software. The algorithm identifies and classifies parasitic elements, generating a report [1].
- User Audit (Optional): A skilled medical technologist can review the digital images and the AI's findings to audit and confirm the results, a step that has been shown to achieve near-perfect agreement with reference methods [1].
{## The Scientist's Toolkit: Essential Research Reagents and Materials}
The following table lists key materials and reagents required to perform the FECT and operate the FA280 system, which is crucial for laboratory planning and budgeting.
{Table: Essential Research Reagents and Materials}
| Item | Function/Application | Primary Technique |
|---|---|---|
| 10% Formalin Solution | Preserves parasite morphology and fixes the stool sample. | FECT [6] |
| Ethyl Acetate | Solvent that extracts fats and debris from the fecal suspension, cleaning the sample. | FECT [6] [12] |
| 0.85% Saline or Distilled Water | Used for diluting and resuspending the final sediment for microscopy. | FECT [6] |
| Conical Centrifuge Tubes | Used for the concentration steps during centrifugation. | FECT [6] |
| Gauze or Specialty Sieves | Filters out large, coarse fecal debris from the suspension. | FECT [6] [12] |
| Microscope Slides & Coverslips | For preparing wet mounts of the concentrated sediment for examination. | FECT [6] |
| Light Microscope | Essential for the visual identification of parasites in the final sample. | FECT [1] [12] |
| FA280 Proprietary Diluent | Liquid used by the analyzer to automatically mix and prepare the stool sample for imaging. | FA280 [1] |
| FA280 Filtered Sample Tubes | Proprietary collection tubes designed for use with the automated sampling system. | FA280 [1] [2] |
{## Conclusion}
The choice between the FA280 and FECT is not a simple matter of superiority but depends heavily on the context and priorities of the diagnostic or research setting. The FECT remains a highly sensitive, cost-effective (in terms of materials), and well-validated "gold standard" [12]. It is particularly suitable for low-resource environments, laboratories with low sample volume, or studies where maximizing sensitivity for a wide range of parasites is the paramount concern, even at the cost of technologist time and potential fatigue-related errors [13].
Conversely, the FA280 analyzer represents a significant advancement in laboratory automation, offering unparalleled gains in efficiency, throughput, and operator safety by minimizing exposure to hazardous chemicals and infectious materials [1]. It is ideally suited for high-volume clinical laboratories, large-scale epidemiological surveys, and facilities aiming to standardize results and reduce dependency on highly specialized microscopists [1] [2]. Its main trade-offs are a higher per-test cost and a reported lower sensitivity compared to FECT when the AI is used without expert auditing, a limitation that may be mitigated by the user audit function [1].
For the future, a hybrid approach, using the FA280 for rapid, high-throughput screening and reserving FECT for confirmation or specific diagnostic challenges, may offer an optimal path forward, leveraging the strengths of both traditional and next-generation diagnostic technologies.
The accurate diagnosis of intestinal parasitic infections (IPIs) is a cornerstone of public health initiatives, clinical management, and drug development programs, particularly in endemic regions. For decades, the Formalin-Ethyl Acetate Concentration Technique (FECT) has served as a cornerstone manual method in parasitology laboratories worldwide, prized for its ability to concentrate a wide array of parasites [14] [15]. In contrast, the FA280 (Orienter Model FA280) represents a technological evolution, employing full automation, artificial intelligence (AI), and digital imaging to modernize the diagnostic workflow [2] [3]. This guide provides an objective, data-driven comparison of the helminth and protozoan detection capabilities of these two platforms, offering researchers and scientists a clear analysis of their respective performances based on recent experimental studies. Understanding the strengths and limitations of each method is crucial for selecting the appropriate diagnostic tool for specific research or clinical objectives, whether for large-scale epidemiological surveys, high-throughput drug efficacy trials, or detailed protozoological studies.
Comparative Diagnostic Performance: Quantitative Data Analysis
The performance of any diagnostic platform is ultimately measured by its sensitivity, specificity, and agreement with established methods. The data presented below summarize key findings from recent comparative studies, providing a quantitative basis for platform evaluation.
Table 1: Comparative Detection Performance of FA280 and FECT against Reference Methods
| Parasite Type | Platform | Comparative Method | Sensitivity / Agreement | Key Findings and Context |
|---|---|---|---|---|
| Helminths (Overall) | FA280 with User Audit | FECT | Perfect agreement (κ = 1.00) [3] | Excellent for helminth species identification. |
| Helminths (Overall) | FA280 AI Report | FECT | Fair agreement (κ = 0.367) [3] | AI-alone performance was suboptimal. |
| Clonorchis sinensis | FA280 | Kato-Katz | 96.8% agreement (κ = 0.82) [2] | Strong agreement in a community-based study (n=1000). |
| Hookworm | FECT | Formalin Concentration (FC) | Superior detection (145 vs 89 positives, p<0.001) [16] | FECT demonstrates higher sensitivity for hookworm. |
| Trichuris trichiura | FECT | Formalin Concentration (FC) | Superior detection (109 vs 53 positives, p<0.001) [16] | FECT demonstrates higher sensitivity for T. trichiura. |
| Small Liver Flukes | FECT | Formalin Concentration (FC) | Superior detection (85 vs 39 positives, p<0.001) [16] | FECT demonstrates higher sensitivity for small liver flukes. |
| Protozoa | FA280 with User Audit | FECT | Perfect agreement (κ = 1.00) [3] | Excellent for protozoan species identification. |
| Protozoa | Formalin-Tween | Formalin-Ether | Higher sensitivity for protozoan cysts [7] | Alternative manual methods may outperform FECT for protozoa. |
Table 2: Platform Characteristics and Practical Considerations
| Feature | FA280 Automated Fecal Analyzer | Formalin-Ethyl Acetate Concentration Technique (FECT) |
|---|---|---|
| Core Principle | Automated sedimentation, AI-driven egg identification, digital imaging [2] [3] | Manual centrifugation, chemical concentration, visual microscopy [14] [15] |
| Throughput | High-throughput, reduced manual labor [2] [3] | Low- to medium-throughput, labor-intensive [2] |
| Sample Input | ~0.5 g of feces [2] | ~1 g of feces (can vary) [16] [17] |
| Key Advantages | Standardized operation, reduced biohazard exposure, digital records, high user acceptance [2] [3] | Lower per-test cost, well-established protocol, high sensitivity for helminths, no specialized equipment needed [16] [14] |
| Key Limitations | Higher instrument cost, lower sensitivity vs. FECT in some studies, performance varies by parasite [3] | Labor-intensive, time-consuming, requires expert microscopists, subjective results, biohazard risk [2] [3] |
| Ideal Use Case | Large-scale screening, settings with budget for automation, digital archiving | Resource-limited settings, low-volume labs, research requiring maximum sensitivity for helminths |
Experimental Protocols in Focus
A clear understanding of the methodologies used in validation studies is essential for interpreting the comparative data.
FA280 Diagnostic Protocol
The FA280 procedure is designed for minimal manual intervention. Approximately 0.5 grams of a fecal sample is placed into a filtered collection tube with a diluent [2]. The device then automates the subsequent steps: mixing, sedimentation, and concentration. A microscope within the analyzer automatically captures high-resolution images through multi-field tomography, and integrated AI software analyzes these images to identify and report on parasitic structures [2] [3]. A critical step in many studies is the "user audit," where a trained medical technologist reviews the AI-generated images and report, a process that has been shown to significantly improve diagnostic accuracy compared to the AI report alone [3].
Formalin-Ethyl Acetate Concentration Technique (FECT) Protocol
FECT is a multi-step manual procedure that requires technical expertise [14] [15].
- Emulsification: Approximately 1-2 grams of stool is emulsified in a 10% formalin solution for fixation.
- Filtration: The suspension is filtered through gauze or a sieve into a conical centrifuge tube to remove large debris.
- Centrifugation: The tube is centrifuged (e.g., at 500 × g for 5 minutes), and the supernatant is discarded.
- Solvent Addition: The sediment is re-suspended in 10% formalin, and 4 mL of ethyl acetate is added. The tube is shaken vigorously and centrifuged again. This step is crucial as ethyl acetate extracts fats and debris, forming a plug at the top of the tube and concentrating parasites in the sediment.
- Examination: The debris plug is freed, and the supernatant layers are decanted. The final sediment is used to prepare a wet mount for microscopic examination by a trained technologist [16] [14].
Workflow Visualization
The fundamental difference between the two platforms lies in their operational workflows, which directly impacts throughput, labor requirements, and objectivity.
The Scientist's Toolkit: Key Research Reagents and Materials
The execution of both diagnostic protocols relies on a set of specific reagents, each serving a critical function in the process of parasite recovery and identification.
Table 3: Essential Reagents for Parasitological Diagnosis
| Reagent/Material | Function in Protocol | Primary Platform |
|---|---|---|
| 10% Formalin | Fixes and preserves parasitic structures (eggs, larvae, cysts); kills pathogens. | Both FECT & FA280 |
| Ethyl Acetate | Solvent that dissolves fats and removes debris; concentrates parasites in sediment. | FECT |
| Diethyl Ether | Alternative solvent to ethyl acetate; similar function but more flammable and hazardous. | FECT (Alternative) |
| Surfactants (e.g., CTAB) | Reduces surface tension, improves parasite recovery from fecal matter in flotation techniques. | Advanced Methods [18] |
| Malachite Green / Methylene Blue | Staining agent in Kato-Katz solution; clears debris for better egg visibility. | Kato-Katz (Comparator) |
| Lugol's Iodine | Stains protozoan cysts (nuclei, glycogen) for easier identification under microscopy. | FECT & FA280 (slide audit) |
The choice between the FA280 and FECT is not a simple matter of declaring one superior to the other, but rather of matching the platform's strengths to the specific research or diagnostic context.
For helminth detection, the evidence indicates that FECT remains a highly sensitive and reliable method, particularly for soil-transmitted helminths and food-borne trematodes like Clonorchis sinensis [16]. The FA280 has demonstrated strong agreement with the Kato-Katz technique for C. sinensis and, when combined with a user audit, achieves perfect agreement with FECT for helminth species identification [2] [3]. This makes the FA280 a viable and efficient alternative for helminth screening, especially in high-volume settings.
For protozoan detection, both platforms can achieve accurate identification, with the FA280 plus user audit again matching FECT performance [3]. However, it is important to note that some studies suggest alternative concentration techniques, such as the Formalin-Tween method, may offer higher sensitivity for protozoan cysts than FECT [7]. The challenge of distinguishing protozoa due to their small size and morphological similarities is a limitation that AI is still overcoming, as seen in the fair agreement of the FA280's AI-only report [3].
In conclusion, the FA280 digital feces analyzer offers a transformative approach for laboratories prioritizing throughput, standardization, and digital pathology. Its automated workflow reduces labor and biohazard exposure, making it suitable for large-scale surveys and drug efficacy monitoring. In contrast, the FECT remains a robust, cost-effective, and highly sensitive manual technique, indispensable in resource-limited settings and for research demanding the highest possible detection sensitivity for helminths. The decision for researchers and drug development professionals should be guided by a careful consideration of workload, budget, target parasites, and the required balance between absolute sensitivity and operational efficiency.
Laboratory Implementation: Workflow Optimization and Practical Applications
The Formalin-Ethyl Acetate Concentration Technique (FECT) remains a cornerstone procedure in clinical parasitology laboratories for the diagnosis of intestinal parasitic infections (IPIs). As a widely adopted gold standard, it enables the detection and identification of helminth eggs, larvae, and protozoan cysts in stool samples through microscopic examination. The technique's fundamental principle relies on the differential density of parasitic elements compared to fecal debris, allowing their separation and concentration for improved diagnostic sensitivity. In contemporary laboratory practice, traditional methods like FECT face challenges from emerging automated technologies, particularly the FA280 fully automated fecal analyzer. This comparison guide provides a detailed objective analysis of the standardized FECT protocol alongside experimental data comparing its performance with the FA280 system, offering researchers and scientists comprehensive methodological insights for diagnostic decision-making.
Experimental Protocols: Methodological Approaches
Standardized FECT Protocol: Step-by-Step Methodology
The Formalin-Ethyl Acetate Concentration Technique follows a standardized procedural sequence to optimize parasite recovery and detection:
Sample Preparation: Emulsify approximately 1 gram of fresh stool specimen in 7 mL of 10% formol saline (formalin) in a clean conical centrifuge tube. The formalin serves as a fixative that preserves parasitic structures while maintaining morphological integrity for accurate identification. Following a 10-minute fixation period, the mixture is strained through three folds of gauze or a sieve (approximately 2.0 mm pore size) to remove large particulate matter and fibrous debris [19] [5].
Centrifugation Parameters: Combine the filtrate with 3-4 mL of ethyl acetate in a 15 mL conical centrifuge tube. Securely cap the tube and shake vigorously for 10-15 seconds to ensure complete mixing, taking care to periodically release pressure through the cap. Centrifuge at 1500 rpm (approximately 500 × g) for 5 minutes [19]. This standardized centrifugation force creates distinct layers: a top layer of ethyl acetate, a plug of debris at the interface, formalin below, and sediment containing parasites at the bottom.
Microscopy Techniques: Carefully decant the top three layers (ethyl acetate, debris plug, and formalin), leaving the sediment undisturbed. Prepare wet mounts from the sediment by transferring two drops to a clean glass slide and applying a coverslip. Examine systematically under the microscope, initially at 10× magnification for scanning, followed by 40× magnification for detailed morphological assessment of any detected parasitic elements [19]. Some protocols recommend examining both saline and iodine preparations to enhance differentiation of protozoan cysts.
FA280 Automated Fecal Analyzer Protocol
The FA280 system represents a technological advancement in fecal analysis with a simplified workflow:
Sample Preparation: Weigh 0.5-1 gram of stool specimen and transfer it to the proprietary sample container without the need for manual filtration or pre-mixing with solvents [5] [20].
Automated Processing: Load the sample container into the FA280 instrument, which automatically performs homogenization, filtration, and preparation of slides for digital imaging. The system requires approximately 2 minutes of hands-on technician time per sample [5].
Digital Imaging and Analysis: The instrument captures high-resolution images of the prepared sample, which are then reviewed by a trained technician on a digital interface. The system typically requires 3-5 minutes for image review and parasite identification [5].
Comparative Performance Data: FECT vs. FA280
Diagnostic Sensitivity and Detection Capabilities
Multiple studies have evaluated the diagnostic performance of FECT compared with emerging automated systems:
Table 1: Detection Performance Comparison Between Diagnostic Methods
| Parameter | Direct Wet Mount | FECT | FA280 with User Audit |
|---|---|---|---|
| Sample Weight Used | 0.2 g | 2 g | 0.5-1 g |
| Detection Rate | 41% | 75% | Comparable to reference methods |
| Process Simplicity | Less complicated | More complicated | Less complicated |
| Processing Time | 2 min/sample | 8-10 min/sample | 2 min/sample |
| Parasite Observation Time | 5-10 min | 5-10 min | 3-5 min |
| Cost per Test | USD 0.25 | USD 0.50 | USD 2.00 |
A hospital-based cross-sectional study conducted at AIIMS Gorakhpur demonstrated FECT's superior detection capability, identifying parasites in 75% of cases (82/110 samples) compared to 62% (68/110) with the formalin-ether concentration method and 41% (45/110) with direct wet mount examination [19]. The FA280 system has shown comparable detection performance to established methods, with one study reporting 96.8% agreement with the Kato-Katz technique and no statistically significant difference in positive rate detection (10.0% for both methods, P > 0.999) [20].
Technical and Operational Characteristics
Table 2: Technical and Operational Comparison of FECT and FA280
| Characteristic | FECT | FA280 Automated Analyzer |
|---|---|---|
| Technique Type | Manual | Automatic |
| Infrastructure Requirements | Centrifuge, microscope, fume hood | Dedicated instrument, power source |
| Reagent Handling | Multiple chemical reagents (formalin, ethyl acetate) | Self-contained reagent system |
| Result Documentation | Manual recording | Automated digital storage |
| Personnel Skill Requirement | Experienced laboratory technician | Trained operator |
| Throughput Capacity | Moderate (batch processing) | High (sequential processing) |
The FA280 system significantly reduces hands-on technical time (approximately 2 minutes per sample) compared to FECT (8-10 minutes per sample) while maintaining detection accuracy [5]. Qualitative assessments from medical staff indicate that the FA280 outperforms conventional methods in testing procedures, detection results, and user acceptance, though it requires a higher initial investment [20].
Research Reagent Solutions: Essential Materials for FECT
Table 3: Essential Research Reagents for FECT Protocol
| Reagent/Material | Function | Specification Notes |
|---|---|---|
| 10% Formalin (Formol saline) | Fixative and preservative | Maintains parasite morphology; 7 mL per 1 g stool |
| Ethyl Acetate | Extraction solvent | 3-4 mL per sample; creates separation interface |
| Diethyl Ether | Alternative extraction solvent | Used in formalin-ether protocol variants |
| Phosphate-Buffered Saline (PBS) | Suspension buffer | Alternative to saline for washing steps |
| Normal Saline (0.9% NaCl) | Mounting medium | For wet mount preparations |
| Iodine Solution | Staining solution | Enhances protozoan cyst visualization |
| Gauze or Sieve | Filtration media | Approximately 2.0 mm pore size for debris removal |
Workflow Visualization: FECT Procedural Sequence
Discussion: Applications in Research and Diagnostic Contexts
The continued relevance of FECT in contemporary parasitology laboratories is evidenced by its robust performance characteristics and adaptability to various research settings. While the technique demonstrates superior sensitivity compared to direct examination methods, its manual nature introduces potential variability in execution across different operators. The procedural complexity of FECT necessitates trained laboratory personnel familiar with the nuances of layer decanting and sediment preparation to minimize parasite loss [19]. The requirement for chemical reagents, particularly formalin and ethyl acetate, necessitates appropriate safety measures including fume hoods and personal protective equipment, adding to the operational considerations for laboratory directors.
Emerging automated systems like the FA280 analyzer address several limitations of manual techniques by standardizing the preparation process and reducing technical variability. The system's digital imaging component creates opportunities for image archiving, teleparasitology consultations, and the application of artificial intelligence algorithms for automated parasite detection [21] [20]. Research demonstrates that deep-learning-based approaches can achieve high accuracy in intestinal parasite identification, with models like DINOv2-large demonstrating 98.93% accuracy, 78.00% sensitivity, and 99.57% specificity [21]. Such technological advancements represent a promising direction for high-throughput diagnostic applications in large-scale research studies and public health surveillance programs.
The economic considerations between these methodologies extend beyond per-test costs to include personnel time, training requirements, and infrastructure support. While FECT remains the more economically accessible option for resource-limited settings, automated systems may offer long-term efficiencies in high-volume laboratory environments through reduced hands-on time and streamlined workflow [5] [20]. Researchers must consider these operational factors alongside diagnostic performance when selecting appropriate methodologies for specific study designs and applications.
This comparative analysis demonstrates that both FECT and automated systems like the FA280 have distinct roles in modern parasitology research and diagnostics. The standardized FECT protocol offers a cost-effective, sensitive method adaptable to various laboratory settings, while the FA280 system provides operational efficiency and standardization benefits for high-volume applications. Researchers should consider their specific diagnostic needs, available resources, and throughput requirements when selecting between these methodologies. The ongoing integration of digital imaging and artificial intelligence with conventional techniques represents a promising frontier for enhancing diagnostic accuracy and efficiency in intestinal parasite detection.
The diagnosis of intestinal parasitic infections is a cornerstone of public health, particularly in tropical and subtropical regions. For decades, the formalin-ethyl acetate concentration technique (FECT) has been a standard manual method for parasite detection, prized for its sensitivity but hampered by its labor-intensive and time-consuming nature. The advent of fully automated digital analyzers, such as the Orienter Model FA280, represents a significant technological shift, aiming to modernize parasitology diagnostics. This comparison guide objectively evaluates the operational workflow, performance, and applicability of the FA280 system against the conventional FECT, providing researchers and scientists with a detailed analysis of their respective protocols, strengths, and limitations.
The Conventional Benchmark: Formalin-Ethyl Acetate Concentration Technique (FECT)
The FECT is a well-established manual concentration method that enhances the detection of parasites in stool samples. Its principle is based on concentrating parasitic elements like eggs, larvae, and cysts by separating them from fecal debris through a process of filtration, chemical treatment, and centrifugation. The procedure requires approximately 2 grams of stool sample mixed with formalin, which is then strained and mixed with ethyl acetate before centrifugation. The resulting sediment is examined under a light microscope by a trained technologist [3] [1]. While it is considered a reference method due to its high sensitivity, its manual nature makes it tedious and variable, with processing times ranging from 8 to 10 minutes per sample and requiring significant expertise [5].
The Automated Contender: Orienter Model FA280
The FA280 is a fully automatic digital feces analyzer designed to streamline the parasitological examination of stool samples. Its operation integrates advanced robotics, high-resolution imaging, and artificial intelligence (AI). The system uses a principle of automatic sedimentation and concentration, processing approximately 0.5 grams of a fecal sample placed in a specialized collection tube [2] [1]. The core of its innovation lies in its automated workflow: the instrument handles sample mixing, preparation of microscopic slides, digital image capture via multi-field tomography, and subsequent AI-driven analysis to identify and report parasitic structures [2]. This automation significantly reduces hands-on time and technical burden.
Detailed Operational Workflow Comparison
The fundamental difference between the two methods lies in their degree of automation and the resulting workflow efficiency. The following diagrams and breakdown illustrate these distinct procedures.
Workflow of the Formalin-Ethyl Acetate Concentration Technique (FECT)
The FECT workflow is entirely manual, requiring skilled laboratory personnel at every stage. It is a multi-step process that involves chemical processing and physical concentration of the sample.
Manual FECT Experimental Protocol [1] [5]:
- Sample Preparation: Weigh approximately 2 grams of stool and emulsify it in 10 ml of 10% formalin in a conical tube.
- Filtration: Strain the mixture through a two-layer gauze into a new 15-ml conical centrifuge tube to remove large particulate matter.
- Solvent Addition: Add 3 ml of ethyl acetate to the filtered suspension. Close the tube tightly.
- Vigorous Mixing: Shake the tube vigorously in an inverted position for one minute.
- Centrifugation: Centrifuge the tube at 2500 rpm for 2 minutes. This results in four layers: a thin plug of debris at the top, a layer of ethyl acetate, a formalin layer, and the sediment at the bottom.
- Sediment Isolation: Free the debris plug by ringing the tube with an applicator stick. Decant the top three layers. Use a cotton-tipped applicator to wipe the inner walls of the tube to remove residual debris.
- Slide Preparation: Using a pipette, transfer a portion of the remaining sediment onto a clean glass slide.
- Microscopy: Examine the slide under a light microscope at appropriate magnifications. A skilled laboratory technician must identify and count the parasitic structures, which typically takes 5-10 minutes of observation.
Workflow of the FA280 Fully Automatic Digital Feces Analyzer
The FA280 automates the entire process from sample loading to result analysis, minimizing human intervention. The workflow is streamlined and integrated within the instrument.
Automated FA280 Experimental Protocol [2] [1]:
- Sample Loading: Approximately 0.5 grams of a stool sample is collected in a proprietary filtered sample collection tube. A batch of up to 40 tubes can be loaded into the instrument's sample carrier track.
- Automated Processing:
- Sampling & Mixing: The instrument's sampling unit uses a pneumatic mixing system to thoroughly homogenize the sample with a diluent.
- Macroscopic Analysis: A high-resolution camera captures images of the sample's macroscopic attributes, such as color and consistency.
- Sedimentation & Concentration: The analyzer employs an automatic sedimentation and concentration technology to prepare the sample for microscopy.
- Digital Imaging: The prepared sample is presented to the microscope unit, which uses high- and low-power objective lenses to automatically capture high-resolution images through multi-field tomography, ensuring comprehensive coverage.
- Image Analysis: The captured digital images are automatically evaluated by the instrument's proprietary AI software, which is trained to identify and classify parasitic structures based on their morphology.
- Result Verification & Reporting: The AI generates a diagnostic report. In many research and clinical settings, a crucial final step involves a "user audit," where a skilled medical technologist reviews the AI-flagged images to verify the results, adding a layer of quality control. The entire process, from loading to reporting, takes about 30 minutes for a batch of 40 samples [2] [1].
Performance Data and Comparative Analysis
Diagnostic Agreement and Sensitivity
Independent studies have directly compared the diagnostic performance of the FA280 and FECT. The data indicates that while the FA280 offers excellent agreement for certain parasites, its sensitivity can be lower than FECT, partly due to the smaller stool sample used.
Table 1: Comparative Diagnostic Performance of FA280 (with User Audit) vs. FECT
| Parameter | FA280 with User Audit | FECT (Reference) | Notes and Context |
|---|---|---|---|
| Overall Parasite Detection | Lower detection rate [3] | Significantly higher detection rate [3] | Disparity attributed to FECT's larger sample size (2g vs 0.5g). |
| Helminth Species Identification | Strong Agreement (κ = 0.857) [3] | Reference Standard | High level of agreement for worm parasites. |
| Protozoan Species Identification | Perfect Agreement (κ = 1.00) [3] | Reference Standard | Perfect agreement for protozoan cysts. |
| AI-Only vs. FECT Agreement | Fair Agreement (κ = 0.367) [3] [1] | Reference Standard | Highlights critical importance of user audit for accurate AI results. |
| Comparison with Kato-Katz | 96.8% Agreement (κ = 0.82) [2] | Not Applicable | Study on Clonorchis sinensis detection, showing strong agreement with another common method. |
Operational and Practical Metrics
From a laboratory management perspective, operational metrics such as throughput, cost, and required expertise are critical for evaluating a new technology.
Table 2: Operational and Practical Comparison: FA280 vs. FECT
| Operational Metric | FA280 Automated Analyzer | Traditional FECT |
|---|---|---|
| Sample Processing Time | ~2 minutes per sample (batch of 40 in ~30 min) [5] | 8-10 minutes per sample [5] |
| Observation Time | 3-5 minutes (technologist review) [5] | 5-10 minutes (manual microscopy) [5] |
| Sample Weight | 0.5 g [2] [1] | 2.0 g [1] [5] |
| Skill Dependency | High for setup and user audit; low for routine operation | Consistently high for all steps |
| Result Storage & Traceability | Yes, digital images and reports are saved [5] | No, typically no permanent record |
| Throughput | High-throughput, suitable for batch processing [3] | Low-throughput, limited by technician time |
| Cost per Test | ~USD 2.00 [5] | ~USD 0.50 [5] |
| Laboratory Safety | Reduced contamination risk [3] | Higher exposure risk to chemicals and samples |
The Scientist's Toolkit: Key Research Reagents and Materials
The following table details essential materials and reagents used in the experimental protocols for both FECT and the FA280 analyzer, as cited in the research literature.
Table 3: Essential Research Materials for Parasitological Stool Analysis
| Item | Function/Description | Application in Protocol |
|---|---|---|
| 10% Formalin Solution | A fixative and preservative that hardens the stool and kills most pathogenic organisms, ensuring sample stability. | Used in FECT to emulsify and preserve the stool sample [1]. |
| Ethyl Acetate | An organic solvent that extracts fat and debris from the fecal suspension, concentrating parasitic elements in the sediment. | Added to the formalin-sample mixture in FECT before centrifugation [1]. |
| Hexadecyltrimethylammonium Bromide (CTAB) | A cationic surfactant that modifies surface charges, aiding in the separation and recovery of parasites from fecal debris. | Used in advanced processing techniques like Dissolved Air Flotation (DAF) integrated with automated analysis [18]. |
| FA280 Filtered Sample Collection Tube | A proprietary tube containing a filter, designed for specific use with the FA280 analyzer. | Used to contain and filter the ~0.5g stool sample for automated loading and processing [2] [1]. |
| Lugol's Iodine Solution | A staining solution that enhances the visualization of protozoan cysts by staining their nuclei and glycogen. | Often used to stain slides prepared by both FECT and other concentration methods for microscopic examination [18]. |
The comparative analysis between the Orienter Model FA280 and the formalin-ethyl acetate concentration technique reveals a clear trade-off that researchers and laboratory managers must consider.
The FA280 analyzer demonstrates superior efficiency, significantly reducing hands-on time and technician workload through automation. It offers excellent agreement with conventional methods for species identification, particularly for helminths and protozoa when combined with a user audit. Its ability to digitally archive images also enhances traceability and quality control. However, its primary limitations are a lower absolute sensitivity compared to FECT, likely due to the smaller sample size, and a higher per-test cost.
Conversely, the FECT remains a highly sensitive method and is more cost-effective per test. Its status as a benchmark is justified. Its drawbacks are its reliance on highly skilled technicians, low throughput, and increased physical burden on staff.
For high-volume laboratories and public health surveillance programs where workflow efficiency and staff safety are paramount, the FA280 represents a transformative technology. For resource-limited settings or for diagnosing infections with very low parasite loads, the FECT retains its vital role. The integration of a mandatory user audit is currently essential to bridge the performance gap between the AI's capability and expert human analysis. Future developments in AI algorithms and sample processing protocols will be key to further closing the sensitivity gap between automated and traditional methods.
The diagnosis of intestinal parasitic infections remains a cornerstone of public health, particularly in tropical regions. While the formalin-ethyl acetate concentration technique (FECT) has long been a standard laboratory method, its labor-intensive and time-consuming nature has prompted the development of automated alternatives such as the Orienter Model FA280 digital feces analyzer. This guide provides a comprehensive, data-driven comparison of the throughput efficiency of these two methods, analyzing processing time, personnel requirements, and batch processing capabilities to inform researchers and laboratory directors in their diagnostic selection process.
The accurate and efficient diagnosis of intestinal parasites is fundamental to epidemiological surveys, drug efficacy evaluations, and clinical management. The formalin-ethyl acetate concentration technique (FECT) represents a manual concentration method that enhances parasite recovery through centrifugation and chemical separation [1]. In contrast, the FA280 digital feces analyzer represents an emerging technology that automates the entire process from sample preparation to digital imaging and AI-assisted analysis [3] [1]. Understanding the relative throughput efficiency of these methods is crucial for laboratories balancing diagnostic accuracy with operational capacity.
Comparative Performance Data
The following tables summarize the key performance metrics derived from experimental comparisons between the FA280 and FECT methods.
Table 1: Throughput Efficiency Comparison Between FA280 and FECT
| Performance Parameter | FA280 Autoanalyzer | FECT (Manual) |
|---|---|---|
| Sample Weight Used | 0.5 g [1] | 2 g [1] |
| Total Processing Time | ~2 minutes/sample [5] | 8-10 minutes/sample [5] |
| Observation Time | 3-5 minutes [5] | 5-10 minutes [5] |
| Batch Processing Capability | 40 samples/run [1] | Individual sample processing |
| Technician Expertise Required | Required for user audit [5] | Required [5] |
| Result Recording & Storage | Automated digital storage [5] | Manual recording [5] |
Table 2: Cost and Operational Considerations
| Consideration | FA280 Autoanalyzer | FECT (Manual) |
|---|---|---|
| Cost Per Test | USD 2.00 [5] | USD 0.50 [5] |
| Technician Hands-on Time | Significantly reduced | Extensive |
| Process Simplicity | Less complicated [5] | More complicated [5] |
| Laboratory Contamination Risk | Reduced [3] | Higher |
Experimental Protocols for Efficiency Assessment
FA280 Digital Feces Analyzer Protocol
The FA280 employs a fully automated workflow based on simple sedimentation principles [1]. The system consists of an automatic in-sample unit, sampling unit with pneumatic mixing, sample character and color photographing unit, microscope unit with high- and low-power objective lenses, and a test kit unit [1].
Methodology:
- Sample Preparation: Approximately 0.5 g of stool is placed in a filtered sample collection tube [1].
- Automated Processing: The sample is automatically mixed with diluent using a high-frequency pneumatic mixing system [1].
- Digital Imaging: High-resolution cameras capture images of sample attributes and color. The microscope unit automatically captures multifield tomography images at different magnifications [1].
- AI Analysis: Images are automatically analyzed by artificial intelligence software to generate preliminary reports [3].
- User Audit: A skilled medical technologist reviews and confirms the AI-generated findings [3].
- Batch Processing: The system processes up to 40 samples in a single run, requiring approximately 30 minutes total run time [1].
Formalin-Ethyl Acetate Concentration Technique (FECT) Protocol
The FECT method operates on centrifugal-sedimentation principles to separate parasitic elements from fecal debris [9] [1].
Methodology:
- Sample Preparation: 2 g of stool sample is mixed with 10 ml of 10% formalin [1].
- Filtration: The fecal suspension is strained through a 2-layer gauze into a 15-ml conical centrifuge tube [1].
- Solvent Addition: 3 ml of ethyl acetate is added to the mixture [1].
- Mixing: The tube is tightly closed and vigorously shaken in an inverted position for 1 minute [1].
- Centrifugation: The tube is centrifuged at 2500 rpm for 2 minutes [1].
- Debris Removal: The plug of debris at the top of the tube is freed, the supernatant is decanted, and debris on the tube sides is removed with a cotton-tipped applicator [1].
- Microscopy: The sediment is pipetted onto a clean glass slide and examined under a light microscope for ova and parasites [1].
Workflow Visualization
The contrasting workflows between the automated FA280 and manual FECT methods can be visualized through the following diagram:
Diagram 1: Comparative workflow analysis between FA280 automated analyzer and manual FECT method
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for Parasitological Diagnosis
| Reagent/Material | Function/Application | Method |
|---|---|---|
| 10% Buffered Formalin | Sample preservation and fixation | FECT [1] |
| Ethyl Acetate | Solvent for extraction of fats and debris | FECT [1] |
| Alcorfix | Alcohol-based fixative in closed concentration systems | Alternative to formalin [8] |
| PROTO-FIX | Formalin-free collecting/preserving solution | Alternative concentration methods [22] |
| CONSED Solution | Specialized solution for sedimentation method | CONSED sedimentation technique [22] |
| Orienter FA280 Diluent | Proprietary solution for sample dilution and mixing | FA280 Autoanalyzer [1] |
| Gauze (2-layer) | Filtration of coarse fecal particles | FECT [1] |
| Parasep SF Tubes | Closed concentration system with integrated filtration | Alternative concentration method [8] |
Discussion and Research Implications
Throughput Efficiency Analysis
The FA280 demonstrates superior throughput efficiency, processing samples in approximately 2 minutes compared to 8-10 minutes for FECT [5]. This 75-80% reduction in processing time represents a significant advantage for high-volume laboratories. Furthermore, the FA280's batch processing capability of 40 samples per run enables substantial scalability, while FECT requires individual sample handling throughout the entire process [1].
Personnel and Training Considerations
While both methods require skilled technicians, the distribution of labor differs substantially. FECT demands continuous expert involvement throughout the multi-step process, whereas the FA280 primarily requires technician expertise for the user audit function, reducing hands-on time by approximately 50-70% based on processing time metrics [3] [5].
Limitations and Research Applications
The higher per-test cost of the FA280 (USD 2.00 vs USD 0.50 for FECT) may constrain implementation in resource-limited settings [5]. Additionally, FECT demonstrated higher sensitivity in some studies, potentially due to the larger stool sample processed (2g vs 0.5g) [3]. Researchers must balance these factors against throughput requirements when selecting diagnostic methodologies for specific study designs.
This efficiency analysis demonstrates a clear trade-off between the operational advantages of the FA280 autoanalyzer and the established sensitivity of traditional FECT. The FA280 offers substantial benefits in processing speed, batch capacity, and reduced technician hands-on time, making it particularly suitable for high-throughput screening applications. Conversely, FECT remains a cost-effective option for laboratories with lower sample volumes or those prioritizing maximum sensitivity through larger sample processing. Future research should focus on optimizing AI algorithms to improve sensitivity while maintaining the throughput advantages of automated systems.
The accurate diagnosis of intestinal parasitic infections is a cornerstone of public health, particularly in tropical and developing regions. The reliability of this diagnosis, however, is profoundly influenced by the methods of sample preservation and preparation prior to analysis. Within the context of evaluating automated diagnostic systems like the FA280 digital feces analyzer against the reference formalin-ethyl acetate concentration technique (FECT), the choice between using fresh or formalin-preserved specimens presents a critical variable. This guide provides a systematic comparison of these two preservation states, synthesizing current experimental data to elucidate their respective impacts on parasite detection efficacy, operational workflow, and diagnostic performance. Understanding these distinctions is essential for researchers and laboratory professionals aiming to optimize testing protocols, interpret comparative study results accurately, and implement these methods effectively in both clinical and research settings.
Experimental Protocols and Methodologies
Sample Collection and Processing Workflow
The comparative evaluation of fresh versus formalin-preserved specimens follows a standardized pathway, branching at the point of preservation. Figure 1 illustrates the generalized experimental workflow, from sample collection to final analysis.
Figure 1: Workflow for the processing of fresh and formalin-preserved stool samples.
Key Experimental Protocols
The following protocols are central to the comparative studies cited in this guide.
FA280 Automated Analysis Protocol: The process begins with the homogenization of either fresh or formalin-preserved stool samples. The FA280 analyzer then automatically prepares smears, generates high-resolution digital images, and analyzes them using a built-in artificial intelligence (AI) program. A critical step in the validation process is the "user audit," where a skilled medical technologist reviews the digital images to confirm or correct the AI's findings [3] [23]. The sample size used by the FA280 is typically 0.5-1.0 grams [5].
Formalin-Ethyl Acetate Concentration Technique (FECT) Protocol: For fresh samples, approximately 2 grams of stool is emulsified in a 10% formalin solution. For preserved samples, the fixed material is used directly. The suspension is filtered to remove large debris, and ethyl-acetate is added as a solvent. The mixture is centrifuged, creating a layered solution where fats and debris are extracted into the ethyl-acetate layer. This layer is discarded, and the concentrated sediment at the bottom of the tube, containing the parasites, is resuspended for microscopic examination [12]. The process is manual, requiring experienced technicians to examine the wet mounts under a microscope [3].
Comparative Performance Data
Detection Sensitivity and Agreement
The performance of a diagnostic method is fundamentally gauged by its sensitivity and its agreement with established reference techniques. The data reveals significant differences between the analysis of fresh and preserved samples.
Table 1: Comparison of detection performance for fresh vs. formalin-preserved specimens.
| Parameter | Fresh Stool Samples | Formalin-Preserved Stool Samples |
|---|---|---|
| FA280 (AI) vs. FECT Agreement | Fair agreement (Overall agreement = 75.5%, κ = 0.37) [3] | Data not available in search results |
| FA280 (User Audit) vs. FECT Agreement | Perfect agreement (Overall agreement = 100%, κ = 1.00) [3] | Strong to perfect agreement for species ID (Helminths κ = 0.86, Protozoa κ = 1.00) [3] |
| Relative Detection Rate (FA280 vs. FECT) | No statistically significant difference [3] | FECT detected significantly more positive samples than FA280 (P < 0.001) [3] |
| Positivity Rate in Clonorchiasis | Data not available | 10.0% positive rate for both FA280 and Kato-Katz (KK), 96.8% agreement [20] |
Operational and Practical Parameters
Beyond pure detection capability, practical considerations such as processing time, cost, and laboratory safety are critical for the adoption of any diagnostic method.
Table 2: Comparison of operational parameters for different methods and sample types.
| Parameter | Direct Wet Smear | FECT | FA280 Analyzer |
|---|---|---|---|
| Stool Sample Weight | 0.2 g [5] | 2 g [5] | 0.5-1 g [5] |
| Technique | Manual [5] | Manual [5] | Automatic [5] |
| Process Simplicity | Less complicated [5] | More complicated [5] | Less complicated [5] |
| Processing Time | ~2 min/sample [5] | 8-10 min/sample [5] | ~2 min/sample [5] |
| Observation Time | 5-10 min [5] | 5-10 min [5] | 3-5 min [5] |
| Result Storage | No [5] | No [5] | Yes (Digital images) [5] |
| Cost per Test | USD 0.25 [5] | USD 0.50 [5] | USD 2.00 [5] |
Impact of Sample Number on Detection
The number of samples collected from a patient is another variable that significantly impacts detection sensitivity, independent of the preservation or analysis method. A retrospective study found that the diagnostic yield for intestinal parasites increases with each additional sample. While some parasites like hookworms were easily detected in the first sample, more than half of the patients infected with Trichuris trichiura and all patients infected with Isospora belli were missed if only one stool specimen was examined [24]. The cumulative detection rate reached 100% after three specimens, underscoring the importance of multiple sampling for accurate diagnosis, particularly in immunocompetent hosts [24].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful parasitological diagnosis relies on a suite of specific reagents and materials. The following table details key items used in the methodologies discussed in this guide.
Table 3: Key research reagents and materials for stool parasite examination.
| Reagent/Material | Function/Application |
|---|---|
| 10% Formalin Solution | Primary fixative and preservative for stool samples; kills pathogens and stabilizes parasite morphology for long-term storage and delayed processing [3] [12]. |
| Ethyl-Acetate Solvent | Used in the FECT method as a less hazardous substitute for ether; extracts fats and debris from the fecal suspension, concentrating parasites in the sediment [12] [25]. |
| Merthiolate-Iodine-Formalin (MIF) | A combined fixative and staining solution used for the preservation and simultaneous staining of protozoan cysts, enhancing their visibility under microscopy [26]. |
| Formalin-Ether Solution | A classic concentration technique solvent; ether is highly effective at removing debris but presents a significant flammability hazard [26] [25]. |
| Conical Centrifuge Tubes | Essential for concentration techniques like FECT; their shape facilitates the formation of distinct layers during centrifugation and the efficient recovery of the parasite-containing sediment [12]. |
| Moulded Fecal Strainers | Used to filter coarse fecal debris from the liquid suspension prior to centrifugation, preventing clogging and allowing for a cleaner microscopic examination [12]. |
The choice between fresh and formalin-preserved specimens presents a clear trade-off that researchers and laboratory managers must navigate. For the FA280 analyzer, fresh samples, when coupled with a user audit, demonstrate exemplary agreement with the reference FECT method. However, the practical advantages of formalin preservation—specifically, enhanced laboratory safety and logistical flexibility for sample transport and batch testing—are substantial. The data indicates that while preservation may lead to a slight reduction in absolute sensitivity for the FA280, possibly due to the smaller sample size processed compared to FECT, it still maintains strong agreement on species identification. Therefore, the decision is context-dependent. In high-throughput, centralized laboratories where workflow efficiency and technician safety are paramount, the use of formalin-preserved specimens with the FA280 represents a robust and viable approach. In settings where maximizing detection sensitivity for low-intensity infections is the absolute priority, and where logistical constraints can be managed, the analysis of fresh samples remains the gold-standard approach.
Intestinal parasitic infections remain a significant global health burden, necessitating accurate and efficient diagnostic methods for epidemiological studies, clinical trials, and drug efficacy monitoring. For decades, the formalin-ethyl acetate concentration technique (FECT) has served as a standard microscopic method for parasite detection. Recently, the fully automated FA280 digital feces analyzer has emerged as a potential alternative, promising increased throughput and reduced labor. This guide provides a comprehensive, evidence-based comparison of these two methodologies, evaluating their performance characteristics, operational requirements, and suitability for various research contexts. We synthesize data from recent comparative studies to objectively assess the strengths and limitations of each system, providing researchers with critical information for methodological selection in parasitic disease research and control programs.
The formalin-ethyl acetate concentration technique (FECT) is a well-established sedimentation method that concentrates parasitic elements by separating them from fecal debris through specific gravity differentiation and chemical treatment. This manual technique involves multiple steps including formalin fixation, ethyl acetate extraction, centrifugation, and microscopic examination by trained personnel [6] [27]. Its widespread adoption in research settings stems from proven sensitivity and cost-effectiveness, particularly in resource-limited environments where parasitic infections are endemic.
The FA280 digital feces analyzer represents technological innovation in parasitological diagnosis, automating the entire process from sample preparation to detection. This system employs intelligent dilution, high-frequency pneumatic mixing, digital imaging, and artificial intelligence (AI)-based pattern recognition to identify and classify parasitic structures in stool samples [3] [2]. By minimizing direct human involvement in the analytical phase, it addresses challenges related to technician expertise and workload capacity while generating digital records for future reference and quality control.
Performance Comparison in Research Settings
Detection Sensitivity and Agreement
Table 1: Comparative detection performance of FA280 and FECT across parasite types
| Parasite Category | Sample Size | FA280 Performance | FECT Performance | Agreement (κ statistic) | Study Reference |
|---|---|---|---|---|---|
| Overall Helminths | 800 preserved samples | Significantly lower detection (P < 0.001) | Higher detection rate | κ = 0.857 (strong) | [3] |
| Protozoa | 800 preserved samples | Comparable detection | Comparable detection | κ = 1.00 (perfect) | [3] |
| Clonorchis sinensis | 1000 community samples | 10.0% positive rate | 10.0% positive rate | κ = 0.82 (strong) | [2] |
| Mixed Parasites (AI vs FECT) | 200 fresh samples | Lower detection (P < 0.001) | Higher detection rate | κ = 0.367 (fair) | [3] |
| Mixed Parasites (User Audit vs FECT) | 200 fresh samples | Comparable detection (P = 1) | Comparable detection | κ = 1.00 (perfect) | [3] |
The FA280 demonstrates variable performance depending on parasite species and operational mode. For helminth detection, particularly in preserved samples, FECT showed significantly higher sensitivity, potentially attributable to its larger sample size (2g versus 0.5-1g for FA280) which increases the probability of detecting low-intensity infections [3]. This difference is crucial for research studies measuring infection prevalence or drug efficacy where missing low-burden infections can significantly impact outcomes.
For protozoan parasites, the FA280 with user audit achieved perfect agreement with FECT, indicating comparable detection capabilities for these organisms [3]. The AI-based detection component alone showed only fair agreement with FECT, suggesting that while the automated classification shows promise, it still requires human verification for optimal performance in research settings.
In community-based screening for Clonorchis sinensis, both methods demonstrated identical positive rates (10.0%) with strong agreement (κ = 0.82), indicating comparable performance for this specific food-borne trematode in field conditions [2]. The agreement was significantly higher in high-intensity infections, suggesting that both methods are reliable for detecting moderate to heavy infections but may differ in sensitivity for low-intensity cases.
Operational Characteristics and Workflow Efficiency
Table 2: Procedural and efficiency comparison between diagnostic methods
| Parameter | Direct Wet Smear | FECT | FA280 with User Audit |
|---|---|---|---|
| Sample Weight | 0.2 g | 2 g | 0.5-1 g |
| Technique Type | Manual | Manual | Automatic |
| Process Simplicity | Less complicated | More complicated | Less complicated |
| Processing Time | 2 min/sample | 8-10 min/sample | 2 min/sample |
| Parasite Observation Time | 5-10 min | 5-10 min | 3-5 min |
| Experienced Technician Required | Yes | Yes | Yes (for audit) |
| Result Recorded and Stored | No | No | Yes |
| Cost per Test | USD 0.25 | USD 0.50 | USD 2.00 |
The FA280 significantly reduces hands-on technical time (2 minutes per sample versus 8-10 minutes for FECT) and decreases parasite observation time (3-5 minutes versus 5-10 minutes for conventional methods) [5]. This operational efficiency becomes particularly valuable in large-scale studies screening thousands of samples, such as national epidemiological surveys or multi-center clinical trials.
However, the substantial cost difference (USD 2.00 per test for FA280 versus USD 0.50 for FECT) must be considered in research budget planning [5]. While the automated system reduces labor requirements, the per-test cost may be prohibitive for large-scale studies in resource-limited settings where parasitic infections are most prevalent.
The FA280's digital image storage capability provides significant advantages for research quality control, allowing re-evaluation of results and data verification – features not available with conventional microscopic techniques [3] [5]. This is particularly valuable in multi-center trials where standardized assessment is crucial and in training programs for field personnel.
Experimental Protocols for Method Comparison
Standard FECT Protocol for Research Studies
The FECT procedure follows a standardized protocol to ensure consistent results across research settings [6]:
Sample Preparation: Approximately 2g of stool is emulsified in 10mL of 10% formalin to preserve parasitic structures.
Filtration: The suspension is strained through gauze or a specialized sieve (500μm pore size) into a 15mL conical centrifuge tube to remove large particulate matter.
Centrifugation: Tubes are centrifuged at 500 × g for 10 minutes to sediment parasitic elements.
Solvent Extraction: The supernatant is decanted, and the sediment is resuspended in 10mL of 10% formalin. Then, 4mL of ethyl acetate is added, the tube is stoppered and shaken vigorously for 30 seconds.
Final Concentration: Tubes are recentrifuged at 500 × g for 10 minutes, resulting in four layers: ethyl acetate, debris plug, formalin, and sediment. The top three layers are discarded, and the sediment is examined microscopically.
This protocol's effectiveness stems from the ethyl acetate extraction of fats and debris, which improves visualization of parasites in the sediment. The larger sample size (2g compared to FA280's 0.5g) potentially enhances detection sensitivity for low-intensity infections [3] [27].
FA280 Automated Protocol
The FA280 system automates the diagnostic process through standardized steps [3] [2]:
Sample Loading: Approximately 0.5g of fecal sample is placed in a filtered collection tube containing a specific diluent.
Automated Processing: The instrument performs intelligent dilution and high-frequency pneumatic mixing to homogenize the sample.
Digital Imaging: The system automatically captures high-resolution images through multi-field tomography.
AI Analysis: Software analyzes images based on morphological characteristics (size, shape, texture) to identify and classify parasitic elements.
Result Verification: A user audit step allows trained technicians to review and verify AI-generated findings, enhancing accuracy.
The entire process requires minimal technical intervention, with the system processing samples in approximately 2 minutes each [5]. The standardized workflow reduces inter-technician variability, a significant advantage in multi-site research studies.
Comparative Study Design Methodology
Recent rigorous comparisons between these methods employed systematic approaches [3] [2]:
Sample Collection: Fresh stool samples collected from target populations, with portions allocated to each method simultaneously.
Methodological Application: Both FECT and FA280 processing performed according to standardized protocols.
Blinded Assessment: Technicians examining results from one method blinded to the results of the other method.
Statistical Analysis: Agreement measured using kappa statistics, with McNemar's test applied to compare positive detection rates.
Discrepancy Resolution: Samples with discordant results re-examined or subjected to additional testing to resolve differences.
This methodological rigor ensures unbiased comparison, providing researchers with reliable data for selecting appropriate diagnostic approaches for specific study designs.
Workflow Comparison Diagram
Research Reagent Solutions and Essential Materials
Table 3: Key reagents and materials for parasitic detection methods
| Item | Application | Function in Protocol | Method |
|---|---|---|---|
| 10% Formalin Solution | Sample preservation | Fixes parasitic structures, prevents degradation | FECT |
| Ethyl Acetate | Solvent extraction | Extracts fats and debris, cleans sample | FECT |
| Filtered Collection Tubes | Sample preparation | Contains specific diluent for automated processing | FA280 |
| Centrifuge Tubes (15mL) | Sample processing | Holds sample during concentration steps | FECT |
| Gauze/Strainer (500μm) | Filtration | Removes large particulate matter | FECT |
| Microscope Slides | Microscopy | Platform for sample examination | FECT |
| Lugol's Iodine Solution | Staining | Enhances visualization of parasitic structures | Both |
| AI Classification Software | Digital analysis | Automates parasite identification and classification | FA280 |
Applications in Specific Research Contexts
Epidemiological Studies
For large-scale prevalence studies, the choice between FECT and FA280 involves trade-offs between cost, sensitivity, and throughput. FECT remains advantageous for community-based surveys in resource-limited settings due to lower per-test costs and established protocols [27]. However, the FA280's higher throughput and reduced technical time make it suitable for rapid assessment in time-sensitive situations or when trained microscopists are scarce.
The FA280's digital archiving capability provides significant benefits for longitudinal studies and retrospective analyses, allowing re-evaluation of samples as research questions evolve [3]. This feature is particularly valuable for monitoring temporal trends in parasitic infections or reanalyzing data when new parasite species are identified in a region.
Clinical Trials and Drug Efficacy Monitoring
In drug efficacy trials where detection of low-intensity infections is crucial, FECT's higher sensitivity for helminths, attributable to larger sample size, may provide more accurate assessment of treatment outcomes [3]. The FA280's lower sensitivity for low-burden infections could underestimate cure rates, particularly for soil-transmitted helminths where egg reduction rate is a primary endpoint.
For protozoan parasites, the FA280 with user audit shows comparable performance to FECT, potentially offering time savings in trials focusing on organisms like Giardia or Entamoeba [3]. The system's consistency reduces inter-observer variability, a common challenge in multi-center trials where standardized endpoint assessment is critical.
Community-Based Interventions
The strong agreement between FA280 and KK method (a similar concentration technique) for Clonorchis sinensis detection (κ = 0.82) supports its use in food-borne trematode surveillance programs [2]. Qualitative research with healthcare staff indicates higher acceptance of the automated system compared to traditional methods, potentially improving compliance with screening protocols [2].
The comparative analysis reveals that both FECT and FA280 have distinct advantages suited to different research scenarios. FECT remains the preferred method for studies requiring maximum sensitivity for helminth detection, particularly in low-intensity infections, and when research budgets are constrained. The FA280 offers significant operational advantages for high-throughput screening, studies with limited technical staff, and research requiring digital archiving for quality control or secondary analysis.
For optimal research outcomes, a tiered approach may be most effective: using FA280 for initial rapid screening in large studies, with FECT confirmation of positive samples and all negative samples in high-risk populations. This hybrid approach balances efficiency with sensitivity, addressing the limitations of each method while leveraging their respective strengths.
Future research should focus on refining the FA280's AI algorithms to improve sensitivity, particularly for low-burden helminth infections, and developing protocols for using larger sample sizes to enhance detection rates. Such advancements could establish automated systems as the new gold standard for parasitological research across diverse settings.
Performance Optimization: Addressing Technical Limitations and Enhancing Diagnostic Accuracy
The diagnosis of intestinal parasitic infections relies heavily on the microscopic examination of stool samples. However, the sensitivity of these diagnostic methods is intrinsically linked to the volume of stool processed, creating a critical methodological variable that directly impacts detection capabilities. This guide examines how sample volume influences parasite detection rates, with a specific focus on comparing the traditional Formalin-Ethyl Acetate Concentration Technique (FECT) against the automated FA280 Digital Feces Analyzer. Understanding this relationship is paramount for researchers and clinicians in selecting appropriate diagnostic approaches, particularly for low-intensity infections and drug efficacy studies where detection sensitivity directly influences outcomes and public health interventions.
Comparative Performance Data: FA280 vs. FECT
The following tables summarize key quantitative findings from comparative studies, highlighting the impact of sample volume and methodological differences on diagnostic performance.
Table 1: Comparison of Methodological Parameters and Overall Performance
| Parameter | FA280 Autoanalyzer | Formalin-Ethyl Acetate Concentration Technique (FECT) |
|---|---|---|
| Stool Sample Volume | 0.5 grams [3] [1] | 2 grams [1] |
| Automation Level | Fully automatic [3] [1] | Manual [1] |
| Processing Time | ~2 minutes per sample [5] | 8-10 minutes per sample [5] |
| Cost Per Test | ~USD $2.00 [5] | ~USD $0.50 [5] |
| Key Advantage | Rapid, convenient, safe; reduced laboratory contamination [3] [1] | Higher sensitivity due to larger sample volume processed [3] [1] |
Table 2: Diagnostic Agreement in Species Identification (FA280 with User Audit vs. FECT)
| Parasite Group | Kappa (κ) Agreement Value | Agreement Strength |
|---|---|---|
| Protozoa | κ = 1.00 (95% CI: 1.00–1.00) [3] [1] | Perfect Agreement |
| Helminths | κ = 0.857 (95% CI: 0.82–0.894) [3] [1] | Strong Agreement |
Table 3: Performance in a Community-Based Study of Clonorchiasis (FA280 vs. Kato-Katz)
| Performance Metric | FA280 Result | Kato-Katz (KK) Result |
|---|---|---|
| Positive Detection Rate | 10.0% [2] | 10.0% [2] |
| Overall Agreement | 96.8% [2] | 96.8% [2] |
| Statistical Kappa (κ) | κ = 0.82 (95% CI: 0.76–0.88) [2] | κ = 0.82 (95% CI: 0.76–0.88) [2] |
| Agreement in High-Intensity Infections | Significantly Higher [2] | Significantly Higher [2] |
Detailed Experimental Protocols
To critically evaluate the data presented, an understanding of the underlying experimental methodologies is essential.
Protocol: Comparative Performance of FA280 vs. FECT
This study employed a two-part design to validate the FA280 against the FECT [3] [1].
- Sample Sets: The investigation utilized two distinct sets of samples. The first consisted of 200 fresh stool samples, and the second comprised 800 samples preserved in 10% formalin [3] [1].
- FECT Procedure: For the FECT, approximately 2 grams of stool were emulsified in 10% formalin. The suspension was strained through gauze into a conical tube, to which 3 ml of ethyl acetate was added. After vigorous shaking and centrifugation, the sediment was examined microscopically for ova and parasites [1].
- FA280 Procedure: For the FA280, approximately 0.5 grams of stool was placed in a filtered collection tube. The analyzer then automatically performed sample dilution, high-frequency pneumatic mixing, and sedimentation. It captured high-resolution digital images via multi-field tomography, which were analyzed by an AI program to generate a report [3] [1].
- Data Analysis: A skilled medical technologist conducted a user audit of the FA280's AI-generated findings. The agreement between methods was statistically analyzed using McNemar's test and Kappa (κ) statistics [3] [1].
Protocol: Community-Based Evaluation of FA280 for Clonorchiasis
This mixed-methods study assessed the FA280's performance in a field setting for detecting Clonorchis sinensis [2].
- Study Design and Population: A cross-sectional survey was conducted among 1000 participants in Xinhui District, Guangdong, China, a region endemic for clonorchiasis [2].
- Sample Collection and Processing: Each participant provided a single stool sample, which was examined in parallel by the FA280 and the Kato-Katz (KK) method, the latter being a common standard in epidemiological surveys [2].
- KK Method: For the KK method, two smears were prepared from each sample, each using 41.7 mg of sieved stool. Experienced technicians examined the slides under a microscope and counted C. sinensis eggs [2].
- Data Analysis: The positive rates and agreement between the FA280 and KK methods were evaluated using McNemar's test and the Kappa statistic. A qualitative component involving interviews with medical staff provided insights into the FA280's applicability and user acceptance [2].
Impact of Sample Volume on Detection Sensitivity: A Workflow
The following diagram illustrates the fundamental trade-off between sample volume and diagnostic sensitivity identified in the comparative research, which is a central finding of the analyzed studies.
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential materials and reagents used in the featured experiments, with their specific functions in the diagnostic process.
Table 4: Essential Reagents and Materials for Parasitological Diagnosis
| Reagent/Material | Primary Function in Diagnostic Protocol |
|---|---|
| 10% Buffered Formalin | Preserves parasite morphology and fixes stool samples for safe handling and processing [3] [1]. |
| Ethyl Acetate | Acts as an organic solvent in concentration techniques; it extracts fat and debris from the fecal suspension, concentrating parasites in the sediment [1]. |
| Orienter Model FA280 Analyzer | A fully automated system that performs intelligent dilution, mixing, sedimentation, and AI-based digital image analysis for high-throughput parasite detection [3] [2] [1]. |
| Kato-Katz Cellophane Slides | Pre-treated with glycerol and malachite green; used in the KK method to clear debris for microscopic visualization and quantitation of helminth eggs [2]. |
| CTAB Surfactant | A cationic surfactant used in advanced processing protocols to modify surface charges, improving parasite recovery efficiency during flotation concentration [18]. |
The body of evidence demonstrates that sample volume is a fundamental determinant of sensitivity in parasite detection. The traditional FECT method, by processing a larger stool mass (2 g), achieves a higher detection rate, making it a more sensitive tool for identifying low-intensity infections. The FA280 autoanalyzer, while offering transformative benefits in speed, standardization, and safety through automation, processes a smaller sample (0.5 g), which inherently limits its absolute sensitivity. The choice between these methods is not a matter of superiority but of strategic application. Researchers and drug development professionals must prioritize their needs: the maximal sensitivity of manual techniques for definitive diagnosis and prevalence studies in low-transmission settings, versus the high-throughput efficiency and workflow advantages of automation for large-scale screening and monitoring programs.
The diagnosis of intestinal parasitic infections, a global public health concern, has long relied on traditional microscopic techniques such as the formalin-ethyl acetate concentration technique (FECT). While considered a standard, FECT is labor-intensive, time-consuming, and highly dependent on technician expertise [3]. The advent of fully automated digital feces analyzers like the Orienter Model FA280 represents a paradigm shift, offering a potential solution to these limitations through automation and artificial intelligence (AI). These systems leverage AI algorithms to identify parasites and ova in stool samples, promising increased throughput and reduced manual labor [3] [2]. However, the deployment of AI in clinical diagnostics necessitates rigorous validation to ensure its accuracy and reliability. This article explores the critical role of user audits—where skilled laboratory personnel review the AI's findings—in bridging the performance gap between raw AI output and established diagnostic methods, ultimately improving diagnostic accuracy to a clinically acceptable standard.
Comparative Performance: FA280 vs. FECT
The diagnostic performance of the FA280 has been directly compared to FECT in multiple studies, with the results highlighting a clear distinction between the AI's initial analysis and its performance after expert human review.
Key Performance Metrics
A study comparing three methods on 200 fresh stool samples revealed stark differences in agreement with FECT [3] [23].
Table 1: Agreement with FECT for Parasite Detection in Fresh Stool Samples (n=200)
| Method | Overall Agreement with FECT | Kappa (κ) Statistic | Strength of Agreement |
|---|---|---|---|
| FA280 (AI Report Only) | 75.5% | κ = 0.367 (95% CI: 0.248-0.486) | Fair |
| FA280 (With User Audit) | 100% | κ = 1.00 (95% CI: 1.00-1.00) | Perfect |
The transformation from "fair" to "perfect" agreement following a user audit underscores the indispensable role of human expertise in the diagnostic workflow. The same study, on a larger set of preserved samples (n=800), found that the FA280 with a user audit maintained strong agreement with FECT for species identification, particularly for helminths (κ = 0.857) and protozoa (κ = 1.00) [3].
Operational and Practical Comparison
Beyond pure accuracy, the choice of a diagnostic method involves practical considerations of workflow, cost, and biosafety.
Table 2: Operational Comparison of FECT and FA280
| Parameter | Formalin-Ethyl Acetate Concentration Technique (FECT) | FA280 Feces Analyzer with User Audit |
|---|---|---|
| Stool Sample Weight | 2 g [5] | 0.5-1 g [5] |
| Technique | Manual [3] | Automatic [3] |
| Total Processing Time | 8-10 minutes per sample [5] | ~2 minutes per sample [5] |
| Parasite Observation Time | 5-10 minutes [5] | 3-5 minutes [5] |
| Biosafety | Higher risk of contamination [3] | Reduced contamination risk; enclosed system [3] [28] |
| Data Management | Results not automatically stored [5] | Digital images and results recorded and stored [5] |
| Cost per Test | Approximately USD 0.50 [5] | Approximately USD 2.00 [5] |
| Technician Dependency | High (required for entire process) [3] | Medium (required for audit, not initial analysis) [3] |
The FA280 offers significant advantages in speed, simplicity, and laboratory safety. However, the higher cost per test and its reliance on a user audit to achieve maximum accuracy are important factors for laboratories to consider [3] [5].
Experimental Protocols and Workflow
Understanding the experimental protocols is essential for interpreting the comparative data.
Detailed Methodology for FA280 and FECT Comparison
The following workflow was used in a key study to validate the FA280 against the FECT method [3]:
Sample Collection and Processing: The study utilized two distinct sets of samples to ensure robustness. For the FA280, approximately 0.5-1 g of stool was placed in a filtered collection tube. The instrument then automatically performed sample dilution, mixing, and filtration. It captured high-resolution digital images through multi-field tomography, which were analyzed by its proprietary AI software to generate an initial report [3] [2].
The User Audit: A critical step followed the AI analysis. A skilled medical technologist, blinded to the FECT results, reviewed the digital microscope images generated by the FA280. This audit involved verifying the AI's identifications and correcting any misclassifications before the final result was issued [3].
Reference Method (FECT): For the FECT, a larger 2 g stool sample was processed using standard centrifugation and concentration steps. The final sediment was examined by trained microscopists [3].
Statistical Analysis: The agreement between the FA280 (both AI-only and user-audited) and the FECT was statistically evaluated using McNemar's test and the kappa (κ) statistic [3] [2].
The User Audit Process
The user audit is a decisive quality control step. The diagram below details its logical flow within the FA280's diagnostic pathway.
The Scientist's Toolkit: Essential Research Reagents and Materials
For researchers seeking to replicate or build upon these validation studies, the following key materials and reagents are essential.
Table 3: Key Research Reagent Solutions for Fecal Parasitology Validation
| Item | Function/Description | Application in Protocol |
|---|---|---|
| Orienter Model FA280 Analyzer | A fully automated digital feces analyzer that uses AI for initial parasite identification. | The primary device under evaluation; handles sample prep, imaging, and initial AI analysis. |
| Formalin (10%) | A preservative and fixative solution for stool specimens. | Used for preserving stool samples for later batch testing with FECT or FA280. |
| Ethyl Acetate | An organic solvent used in concentration techniques. | A key reagent in FECT for extracting fats and debris from the fecal sample, concentrating parasites in the sediment. |
| Hexadecyltrimethylammonium Bromide (CTAB) | A cationic surfactant. | Used in advanced processing protocols like Dissolved Air Flotation (DAF) to improve parasite recovery by modifying surface charges [29]. |
| Lugol's Iodine Solution | A staining solution that highlights nuclear and cytoplasmic details. | Used for staining protozoan cysts and oocysts to facilitate microscopic identification in manual methods. |
| Filtered Sample Collection Tubes | Specialized tubes with built-in filters for automated analyzers. | Used with the FA280 to remove large particulate matter that could interfere with automated analysis. |
Discussion and Future Directions
The experimental data consistently demonstrates that while the FA280's AI algorithm provides a rapid and useful first-pass analysis, it is the integration of a human user audit that elevates its diagnostic accuracy to a level comparable with, and in some cases indistinguishable from, the traditional FECT [3] [2]. This human-in-the-loop model effectively marries the efficiency and consistency of automation with the nuanced expertise of a trained technologist. The audit is crucial for mitigating the AI's limitations, which include lower sensitivity in samples with low parasite load, potentially due to the smaller stool sample size used by the instrument compared to FECT [3].
Future developments in this field will likely focus on improving the underlying AI models. Continuous training with vast and diverse datasets of validated parasite images, potentially incorporating techniques from human-powered AI validation that leverage domain expertise for data quality assurance and complex problem-solving, is essential [30]. As these models evolve, the need for extensive user audits may diminish, but the principle of human oversight will remain a cornerstone of responsible and reliable AI-assisted diagnostic medicine.
The diagnosis of intestinal parasitic infections remains a critical component of public health initiatives, particularly in tropical regions where these infections affect billions of people, causing malnutrition, anemia, and impaired cognitive development [3] [1]. For decades, the formalin-ethyl acetate concentration technique (FECT) has served as a cornerstone method in parasitology laboratories, providing enhanced sensitivity compared to direct smear methods [1]. Despite its widespread use and recognition, FECT presents significant technical challenges that can compromise diagnostic accuracy and efficiency.
This article examines three fundamental technical limitations of FECT: interference from fecal debris, preservation of parasite morphology, and technician variability. Concurrently, we evaluate the FA280 fully automatic digital feces analyzer as a technological advancement designed to address these challenges through automation, digital imaging, and artificial intelligence (AI). Within the broader context of comparative research between traditional and automated diagnostic approaches, we present experimental data from recent studies to objectively assess the performance characteristics of both methods.
Technical Challenges of the FECT Method
Fecal Debris Interference and Sample Processing Limitations
The FECT method involves a complex concentration process where fecal samples are mixed with formalin and ethyl acetate, then centrifuged to separate parasitic elements from debris [1]. A primary limitation of this technique stems from the incomplete removal of fecal debris, which can obscure the visualization of parasites during microscopic examination. This interference is particularly problematic in samples with high fat content or fibrous material, where debris can mimic or hide parasitic structures, potentially leading to false-negative results or misidentification.
The sample size discrepancy between FECT and emerging automated systems presents another significant challenge. FECT typically utilizes 2 grams of stool sample, compared to the approximately 0.5 grams used by the FA280 system [3] [1]. While larger sample size theoretically improves detection sensitivity, it also amplifies the amount of debris in the final concentrate, creating a paradoxical situation where both target organisms and interfering substances are concentrated simultaneously.
Parasite Morphology Preservation Issues
The chemical reagents used in FECT, particularly formalin and ethyl acetate, can adversely affect parasite morphology preservation. Formalin, while excellent for preserving overall structural integrity, can alter the refractive index of parasitic elements, making them more difficult to identify under standard bright-field microscopy. Ethyl acetate, used as an extractant for fats and debris, may damage delicate structures such as protozoan trophozoites, leading to identification challenges.
These morphological alterations are especially problematic for less experienced technicians who rely heavily on textbook appearances for accurate identification. The preservation artifacts introduced by the chemical processing can lead to misidentification of species, particularly among protozoa where morphological differences between pathogenic and non-pathogenic species are subtle and require optimal preservation for discrimination.
Technician Variability and Operational Complexities
The FECT method is notoriously dependent on technician expertise and consistency. The multiple processing steps—including mixing, straining, centrifugation, and slide preparation—introduce numerous variables that can affect final results. Studies have demonstrated significant inter-technician variability in both parasite detection and species identification, particularly in samples with low parasite loads or mixed infections [1].
The operational complexity of FECT extends beyond technical skill to encompass time efficiency and workflow limitations. The manual processing requires approximately 8-10 minutes per sample of active technician time, not including the microscopic examination period [5]. This intensive labor requirement creates bottlenecks in high-volume settings and contributes to technician fatigue, which further compounds variability issues in slide interpretation.
The FA280 System: Technological Approach to FECT Limitations
The FA280 fully automatic digital feces analyzer represents a technological paradigm shift in parasitological diagnosis, transforming a traditionally manual and operator-dependent process into a standardized, automated procedure [31]. The system integrates several technological innovations including automated sample preparation, high-resolution digital microscopy, and artificial intelligence-driven image analysis.
The operational workflow begins with a patented sealed cartridge that eliminates direct technician exposure to biological samples [31]. The system automatically performs sample dilution and mixing through a high-frequency pneumatic system that ensures homogeneity without damaging delicate parasitic structures. The automated sedimentation and concentration technology then prepares the sample for microscopic analysis, with the entire process requiring minimal operator intervention.
AI-Driven Image Analysis and Reduction of Technical Variability
A cornerstone of the FA280 system is its artificial intelligence algorithm for morphological recognition of parasitic elements [31]. The system utilizes a double-lens optical system (low and high power) to capture digital images through multi-field tomography, creating a comprehensive digital representation of the sample concentrate.
The AI software automatically identifies and classifies parasitic structures including eggs, larvae, cysts, and trophozoites based on trained morphological parameters. In cases of uncertain identification, the system flags high-definition images for technician review, creating a collaborative human-machine diagnostic workflow [31]. This approach maintains the interpretive wisdom of experienced parasitologists while leveraging the consistency and objectivity of computerized analysis.
Comparative Experimental Data: FECT vs. FA280
Methodology of Key Comparative Studies
Recent studies have employed rigorous methodological approaches to compare the diagnostic performance of FECT and the FA280 system. One significant study utilized 200 fresh stool samples and 800 formalin-preserved samples to evaluate the agreement between methods [3] [1]. The experimental design included both AI-generated reports and user-audited results from the FA280, compared against FECT as the reference method.
The FECT procedure followed established protocols: 2g of stool was mixed with 10ml of 10% formalin, strained through gauze, mixed with ethyl acetate, centrifuged, and examined microscopically [1]. The FA280 protocol utilized approximately 0.5g of stool in a sealed cartridge, with automated processing and digital imaging [31]. Statistical analysis included McNemar's test for pairwise comparison and kappa (κ) statistics for agreement assessment [3] [1].
Detection Sensitivity and Agreement Metrics
The comparative performance data reveals nuanced differences between the two methods:
Table 1: Comparison of Parasite Detection Between FECT and FA280
| Sample Type | Comparison Method | Overall Agreement | Kappa (κ) Value | Statistical Significance |
|---|---|---|---|---|
| Fresh samples (n=200) | FA280 (User Audit) vs. FECT | 100% | 1.00 (95% CI: 1.00-1.00) | P = 1 (exact binomial test) |
| Fresh samples (n=200) | FA280 (AI Report) vs. FECT | 75.5% | 0.367 (95% CI: 0.248-0.486) | P < 0.001 (McNemar's test) |
| Preserved samples (n=800) | FA280 (User Audit) vs. FECT | N/A | Helminths: κ = 0.857 (95% CI: 0.82-0.894)Protozoa: κ = 1.00 (95% CI: 1.00-1.00) | P < 0.001 (McNemar's test) |
For species identification, the FA280 with user audit demonstrated perfect agreement (κ = 1.00) with FECT for protozoa and strong agreement for helminths (κ = 0.857) in preserved samples [3]. The disparity in detection rates for preserved samples (with FECT detecting significantly more positive samples) was attributed to the larger stool sample size processed by FECT (2g vs. 0.5g), potentially increasing probability of detection in low-burden infections [3] [1].
Operational Efficiency and Practical Considerations
Beyond diagnostic accuracy, practical operational factors significantly impact laboratory workflow:
Table 2: Operational Comparison Between Diagnostic Methods
| Parameter | Direct Wet Smear | FECT | FA280 with User Audit |
|---|---|---|---|
| Weight of stool used | 0.2 g | 2 g | 0.5-1 g |
| Technique | Manual | Manual | Automatic |
| Process simplicity | Less complicated | More complicated | Less complicated |
| Processing time | 2 min/sample | 8-10 min/sample | 2 min/sample |
| Parasite observing time | 5-10 min | 5-10 min | 3-5 min |
| Result recording and storage | No | No | Yes |
| Cost per test | USD 0.25 | USD 0.50 | USD 2.00 |
The FA280 system significantly reduces hands-on technical time and standardizes the pre-analytical phase, minimizing inter-operator variability [5]. However, this operational efficiency comes at a higher cost per test (USD 2.00 vs. USD 0.50 for FECT), an important consideration for resource-limited settings [5].
Research Reagent Solutions and Essential Materials
The effective implementation of either FECT or automated systems requires specific reagents and materials:
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function | Application in Protocol |
|---|---|---|
| 10% Formalin | Fixation and preservation | Preserves parasitic structures in FECT; used in sample preservation for both methods |
| Ethyl Acetate | Solvent extraction | Extracts fats and debris in FECT protocol |
| Hexadecyltrimethylammonium bromide (CTAB) | Surfactant | Enhances parasite recovery in dissolved air flotation (DAF) protocols [29] |
| Lugol's Solution | Staining | Enhances visualization of parasitic structures in microscopic examination |
| Patented Sealed Cartridge | Sample containment | Provides closed system for FA280, ensuring safety and standardization [31] |
| Dissolved Air Flotation System | Parasite recovery | Alternative processing method showing 94% sensitivity with automated analysis [29] |
The technical challenges inherent in the FECT method—particularly debris interference, morphology preservation issues, and technician variability—represent significant limitations in contemporary parasitological diagnosis. The FA280 automated digital feces analyzer addresses many of these challenges through standardized automation, digital imaging, and AI-assisted interpretation, demonstrating substantial agreement with conventional methods while offering operational efficiencies.
However, the choice between methods requires careful consideration of context-specific factors. The FA280 system offers advantages in settings prioritizing workflow efficiency, technician safety, and standardization, particularly where cost considerations are secondary. Conversely, FECT remains a viable option in resource-limited environments where per-test cost outweighs efficiency benefits, and where technical expertise remains available.
Future developments in automated parasitology systems would benefit from addressing the current limitations of reduced sensitivity in low-burden infections through improved concentration methods and AI algorithm refinement. The integration of automated systems like the FA280 with emerging molecular techniques may ultimately provide the optimal diagnostic paradigm, combining morphological preservation with genetic confirmation for challenging diagnostic scenarios.
Experimental Workflow and Technical Diagrams
Diagram 1: Comparative Workflow of FECT and FA280 Methods. The diagram illustrates the procedural differences between the manual FECT protocol and the automated FA280 system, highlighting key technical challenges and advantages at critical process stages.
The diagnosis of intestinal parasitic infections remains a cornerstone of public health initiatives and clinical management in many regions. For decades, the formalin-ethyl acetate concentration technique (FECT) has served as a fundamental diagnostic method, prized for its sensitivity and reliability [3] [32]. However, this manual microscopic technique is characterized by significant labor demands, time consumption, and operational challenges [3] [32]. The advent of fully automated digital feces analyzers, exemplified by the Orienter Model FA280, promises to transform parasitology diagnostics by integrating artificial intelligence (AI) with high-throughput automation [3] [33]. This analysis provides a systematic cost-benefit comparison between the FA280 and FECT, examining reagent costs, capital investment, and labor efficiency to inform decision-making for researchers, laboratory managers, and public health officials engaged in parasite detection and drug development research.
Performance Metrics and Detection Capabilities
A comprehensive understanding of the cost-benefit relationship requires initial examination of the fundamental performance characteristics of each diagnostic approach. The table below summarizes key performance metrics derived from comparative studies.
Table 1: Performance and Operational Comparison Between FECT and FA280
| Parameter | FECT (Reference) | FA280 with User Audit | FA280 with AI Report Only |
|---|---|---|---|
| Sample Weight Used | 2 g [32] [5] | 0.5-1 g [32] [5] | 0.5-1 g [32] |
| Process Simplicity | More complicated [5] | Less complicated [5] | Less complicated [32] |
| Total Processing Time | 8-10 min/sample [5] | ~2 min/sample [5] | ~2 min/sample [32] |
| Parasite Observation Time | 5-10 min [5] | 3-5 min [5] | Automated |
| Agreement with FECT (κ statistic) | Self (N/A) | Perfect (κ=1.00) for fresh samples [3] [32] | Fair (κ=0.367) [3] [32] |
| Helminth Species Identification Agreement | Self (N/A) | Strong (κ=0.857) [3] [32] | Not Reported |
The diagnostic performance of the FA280 is notably enhanced by a user audit, where a skilled technologist reviews the AI-generated findings. One study on fresh stool samples found perfect agreement (κ=1.00) between the FA280 with user audit and FECT [3] [32] [23]. For preserved samples, the agreement for helminth species identification remained strong (κ=0.857) [3] [32]. However, the fully automated AI report alone showed only fair agreement (κ=0.367) with FECT, underscoring the current necessity of expert verification for the most accurate results [3] [32]. It is crucial to note that FECT has demonstrated the ability to detect a higher number of positive samples in some studies, a factor potentially attributable to its use of a larger stool sample (2 g for FECT vs. 0.5 g for FA280), which increases the probability of detecting low-intensity infections [3] [32].
Detailed Experimental Protocols
A clear understanding of the underlying methodologies is essential for a nuanced cost-benefit analysis.
Formalin-Ethyl Acetate Concentration Technique (FECT) Protocol
The FECT method is a manual centrifugation procedure designed to concentrate parasitic elements [32]. The standardized protocol is as follows:
- Sample Preparation: A 2 g stool sample is emulsified in 10 mL of 10% formalin [32].
- Filtration: The resulting suspension is filtered through a two-layer gauze into a 15-mL conical centrifuge tube to remove large debris [32].
- Solvent Addition: 3 mL of ethyl acetate is added to the tube. The tube is sealed and shaken vigorously for one minute [32].
- Centrifugation: The tube is centrifuged at 2500 rpm for 2 minutes. This step creates a layered structure: a plug of debris at the top, a layer of solvent, and parasitic elements concentrated in the sediment at the bottom [32].
- Sediment Examination: The debris plug is loosened and the top layers are decanted. The final sediment is pipetted onto a glass slide for microscopic examination by a trained technologist [32].
FA280 Automated Digital Analysis Protocol
The FA280 system automates the process using AI and digital imaging [32] [33]:
- Sample Loading: Approximately 0.5 g of a fecal sample is placed into a filtered collection tube [2] [32].
- Automated Processing: The instrument uses intelligent dilution and high-frequency pneumatic mixing to standardize the sample consistency [32] [33].
- Imaging and Analysis: The analyzer employs high-resolution CMOS microscopy and multi-field tomography to capture digital images. An integrated AI program automatically identifies and classifies parasite eggs based on learned morphological features [32] [33].
- Audit and Reporting: The results, including captured images, are compiled into a report. In the recommended workflow, these findings are audited by a skilled medical technologist before the final report is issued [3] [32].
The following diagram illustrates the core workflow and technological foundation of the FA280 analyzer.
Financial and Operational Cost-Benefit Analysis
Direct and Indirect Cost Comparison
The financial implications of adopting either method extend beyond simple reagent costs to include equipment investment and personnel time. The following table provides a detailed breakdown.
Table 2: Comprehensive Cost-Benefit Analysis: FECT vs. FA280
| Cost & Efficiency Factor | FECT | FA280 |
|---|---|---|
| Reagent Cost per Test | USD 0.50 [5] | Not Explicitly Stated |
| Total Direct Cost per Test | ~USD 0.50 [5] | ~USD 2.00 [5] |
| Equipment Investment | Microscope (Low) | High-Capital Instrument [3] |
| Laboratory Technician Time | 13-20 min/sample [5] | 3-5 min/sample (Audit) [5] |
| Labor Intensity | High (Manual, multi-step) [3] | Low (Automated) [3] |
| Throughput | Low | High (40 samples/batch in ~30 min) [32] |
| Biosafety & Contamination Risk | Higher (Open handling) [28] | Lower (Fully sealed system) [3] [33] |
| Data Management | Manual recording, no image storage | Automated result and high-resolution image storage [5] |
| Key Operational Advantage | High sensitivity, low reagent cost [3] | Speed, standardization, and safety [3] |
| Key Operational Disadvantage | Time-consuming and laborious [3] | Higher per-test cost and lower sensitivity for low-intensity infections [3] |
The direct cost analysis reveals a stark contrast: FECT costs approximately USD 0.50 per test, while the FA280 costs about USD 2.00 per test [5]. This four-fold difference is significant in resource-constrained settings. However, this direct cost is offset by substantial savings in labor, a major operational expenditure. The FA280 reduces hands-on technologist time to just 3-5 minutes per sample for the audit, compared to 13-20 minutes per sample for the fully manual FECT process [5]. This translates into a 60-75% reduction in active labor time per test, allowing highly trained staff to focus on result verification and other high-value tasks [5].
Equipment and Research Reagent Solutions
The core materials and instruments required for each method differ significantly, impacting initial setup costs and operational workflows.
Table 3: Key Research Reagent Solutions and Equipment
| Item | Function/Application | Method |
|---|---|---|
| 10% Formalin | Preserves parasitic structures in stool samples. | FECT [32] |
| Ethyl Acetate | Organic solvent used to extract fats and debris during concentration. | FECT [32] |
| Centrifuge | Essential for sedimenting parasitic elements in the concentration step. | FECT [32] |
| Conical Centrifuge Tubes | Vessels for the concentration procedure. | FECT [32] |
| Light Microscope | Primary tool for visual identification of parasites. | FECT [32] |
| Orienter Model FA280 Analyzer | Integrated system for automated sample processing, imaging, and AI analysis. | FA280 [3] [32] |
| FA280 Sample Collection Tubes | Proprietary filtered tubes for standardized sample introduction. | FA280 [2] |
| Intelligent Diluent | Reagent for standardizing various stool consistencies for automated imaging. | FA280 [32] [33] |
The capital investment for FECT is relatively low, centered around a standard laboratory centrifuge and microscope. In contrast, the FA280 represents a high-cost capital instrument [3]. This initial investment must be justified by the anticipated volume of testing and the value assigned to increased throughput, labor savings, and improved standardization.
The choice between the traditional FECT and the automated FA280 digital feces analyzer involves a direct trade-off between cost and efficiency. FECT remains the most cost-effective method in terms of direct reagent expense and is capable of high sensitivity, making it suitable for low-volume settings or those with severe budget constraints [3] [5]. However, the FA280 offers compelling advantages in high-throughput environments by dramatically reducing labor requirements, minimizing biosafety risks through a sealed system, and standardizing the diagnostic process with digital archiving capabilities [3] [5] [33].
For researchers and drug development professionals, the FA280's ability to rapidly process batches of samples with digital record-keeping can significantly accelerate experimental workflows and data analysis. The integration of artificial intelligence, while still requiring expert oversight, points toward a future of increasingly automated and precise diagnostic parasitology [3] [26]. The decision ultimately hinges on a laboratory's specific priorities: maximizing diagnostic sensitivity with minimal reagent cost (favoring FECT) versus maximizing throughput, operational efficiency, and standardization (favoring FA280).
Within clinical and research laboratories, the accuracy of intestinal parasite diagnosis has significant implications for patient treatment, public health policies, and drug efficacy studies. The gold standard for diagnosis has traditionally relied on manual microscopic techniques, primarily the formalin-ethyl acetate concentration technique (FECT), which, despite its proven sensitivity, is labor-intensive, time-consuming, and subject to human error [3] [12]. The introduction of fully automated digital analyzers, such as the Orienter Model FA280, represents a paradigm shift in diagnostic parasitology, promising high-throughput testing with reduced manual labor [3] [1].
This guide provides an objective comparison between the automated FA280 digital feces analyzer and the conventional FECT, framing the evaluation within the critical context of quality assurance. The standardization of procedures and methodologies for cross-verification is essential for laboratories considering the integration of such automated systems into their diagnostic or research workflows. We synthesize recent experimental data to compare the performance, operational characteristics, and practical applicability of both methods, offering a robust framework for verification and quality control.
Performance Comparison: FA280 vs. Conventional Techniques
Evaluating the FA280 involves comparing its performance against established manual methods like FECT and the Kato-Katz (KK) technique. Key performance metrics include detection sensitivity, species identification agreement, and operational efficiency. The following tables summarize quantitative data from recent comparative studies.
Table 1: Diagnostic Performance Agreement in Comparative Studies
| Study Focus | Comparison Methods | Sample Size | Statistical Agreement (κ statistic) | Overall Agreement (%) | Key Finding |
|---|---|---|---|---|---|
| General Parasite Detection [3] | FA280 (User Audit) vs. FECT | 200 (fresh) | κ = 1.00 | 100% | Perfect agreement for fresh samples. |
| General Parasite Detection [3] | FA280 (AI Report) vs. FECT | 200 (fresh) | κ = 0.367 | 75.5% | Fair agreement; human audit crucial. |
| General Parasite Detection [3] | FA280 (User Audit) vs. FECT | 800 (preserved) | Helminths: κ = 0.857; Protozoa: κ = 1.00 | N/R | Strong to perfect species identification. |
| Clonorchis sinensis Detection [2] | FA280 vs. Kato-Katz | 1,000 | κ = 0.82 | 96.8% | Strong agreement for a specific food-borne trematode. |
Table 2: Operational and Practical Characteristics
| Parameter | Direct Wet Smear | FECT | FA280 with User Audit |
|---|---|---|---|
| Stool Sample Weight | 0.2 g [5] | 2 g [3] [5] | 0.5–1 g [3] [5] |
| Technique | Manual [5] | Manual [5] | Automatic [5] |
| Process Simplicity | Less complicated [5] | More complicated [5] | Less complicated [5] |
| Total Processing Time | ~7-12 min/sample [5] | ~13-20 min/sample [5] | ~5 min/sample [5] |
| Cost per Test | USD 0.25 [5] | USD 0.50 [5] | USD 2.00 [5] |
| Result Storage | No [5] | No [5] | Yes (Digital Images) [5] |
Key Insights from Performance Data
- Sensitivity and the Sample Volume Factor: While the FA280 shows high agreement with gold standards in species identification, one study noted that FECT detected significantly more positive samples in a set of 800 preserved specimens [3]. This disparity is potentially attributed to the larger stool sample used in FECT (2 g) compared to the FA280 (0.5-1 g), giving FECT a theoretical advantage in detecting low-intensity infections [3] [1].
- The Critical Role of the User Audit: The performance of the FA280 is significantly enhanced by a "user audit," where a skilled technologist reviews the digital images analyzed by the AI. The agreement with FECT jumped from fair (κ = 0.367) with the AI alone to perfect (κ = 1.00) with a user audit, underscoring that the current technology serves best as a powerful assistive tool rather than a fully autonomous diagnostician [3].
- Operational Efficiency vs. Cost: The primary advantages of the FA280 are its simplicity, shorter processing time, reduced hands-on labor, and minimized risk of laboratory contamination [3] [5]. However, this comes at a higher direct cost per test ($2.00) compared to FECT ($0.50) [5]. The value proposition lies in its high-throughput capability, which can free up highly trained personnel for more complex tasks.
Experimental Protocols for Cross-Verification
For a laboratory to validate the FA280 against its existing standard method, a rigorous cross-verification study must be designed. The following outlines a standard protocol based on the methodologies cited in the literature.
Protocol 1: Comparison with FECT for General Parasite Detection
This protocol is adapted from the study by Boonyong et al. (2024) [3] [1].
- Sample Collection and Preparation: Collect fresh stool samples. Partition each sample for parallel processing by FECT and the FA280. A portion of the sample for FECT should be preserved in 10% formalin if testing is delayed.
- FECT Procedure (Reference Method):
- Emulsify 2 g of stool in 10 mL of 10% formalin [1].
- Filter the suspension through two layers of wet gauze into a 15 mL conical centrifuge tube [6] [1].
- Add 3-4 mL of ethyl acetate to the filtered suspension, stopper the tube, and shake vigorously for 30-60 seconds [6] [1].
- Centrifuge at 500 × g for 2-10 minutes [6] [1].
- Ring the plug of debris, decant the supernatant, and use a cotton-tipped applicator to clean the tube walls [6].
- Examine the resuspended sediment under a light microscope for ova and parasites [6] [1].
- FA280 Procedure (Test Method):
- Place approximately 0.5 g of stool into the designated sample collection tube [3] [1].
- Load the tube into the FA280 analyzer. The instrument automatically handles dilution, mixing, and sample transfer to a test kit [1].
- The built-in microscope captures high-resolution, multi-field images which are analyzed by the AI software to generate a preliminary report [1].
- A crucial quality assurance step: a skilled medical technologist must perform a user audit by reviewing all captured digital images to confirm or correct the AI's findings [3].
- Data Analysis: Compare the results from the FA280 (post-user audit) and FECT for each sample. Use McNemar’s test to check for significant differences in detection rates and Cohen’s kappa (κ) statistic to measure agreement in species identification beyond chance [3] [2].
Protocol 2: Comparison with Kato-Katz for Clonorchiasis Screening
This protocol is adapted from the study published in 2025 focusing on Clonorchis sinensis [2].
- Study Design and Sampling: A community-based cross-sectional survey using a multi-stage cluster sampling method is ideal for assessing performance in a real-world screening context [2].
- Kato-Katz Procedure (Reference Method):
- Prepare a smear using a plastic template that holds 41.7 mg of sieved stool on a glass slide [2].
- Cover the sample with a cellophane strip soaked in glycerol-malachite green (or methylene blue) solution [2] [14].
- Allow the slide to clear for a period before microscopic examination.
- Have experienced technicians examine the slides under a microscope and count the number of C. sinensis eggs to calculate eggs per gram (EPG) [2].
- FA280 Procedure (Test Method): The procedure is as described in Protocol 1. The study noted that the agreement between FA280 and KK was significantly higher in high-infection intensity groups [2].
- Data Analysis: Compare positive rates using McNemar’s test and calculate kappa agreement. Additionally, use Pearson’s Chi-square test to analyze the consistency of positive results across different EPG-based infection intensity groups [2].
The following workflow diagram visualizes the key steps in a cross-verification study:
Cross Verification Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful implementation and verification of the FA280 require specific laboratory materials and reagents. The following table details these essential components and their functions in the experimental workflow.
Table 3: Research Reagent Solutions and Essential Materials
| Item Name | Function/Application | Key Characteristics |
|---|---|---|
| 10% Formalin | Preservative for stool samples; component of FECT and FA280 diluent [3] [6]. | Fixes parasites and prevents degradation of morphology. |
| Ethyl Acetate | Solvent in FECT for extracting fats and debris from fecal suspension [6] [12]. | Less flammable and safer alternative to diethyl ether. |
| Ethyl Acetate | Solvent in FECT for extracting fats and debris from fecal suspension [6] [12]. | Less flammable and safer alternative to diethyl ether. |
| Orienter FA280 Analyzer | Fully automated digital feces analyzer [3] [1]. | Integrates automated sampling, imaging, and AI-based analysis. |
| FA280 Test Kits & Diluent | Proprietary consumables for the FA280 system [1]. | Designed for automated dilution and concentration of stool samples. |
| Gauze or Fecal Concentrator | Filtering step in FECT to remove large debris [6] [12]. | Cheesecloth-type gauze or commercial concentrator devices (e.g., Evergreen Scientific). |
| Kato-Katz Template & Cellophane | For quantitative Kato-Katz technique [2] [14]. | Template standardizes stool amount; cellophane is soaked in glycerol-malachite green. |
The FA280 digital feces analyzer presents a compelling alternative to traditional manual methods, offering significant gains in standardization, throughput, and operator safety. The experimental data confirms that with a mandatory user audit, its diagnostic accuracy for species identification is excellent. However, quality assurance protocols must acknowledge its current limitations: a potentially lower absolute sensitivity due to smaller sample volume and a higher per-test cost.
The choice between FA280 and FECT is not a simple binary decision but a strategic one based on context. For high-volume laboratories and large-scale screening programs where efficiency, digital archiving, and technician time are priorities, the FA280 is a transformative tool. For settings with extremely low parasite prevalence, where maximizing sensitivity is the paramount concern, or where budgets are highly constrained, the FECT remains a robust and reliable option. Ultimately, the integration of automated systems like the FA280 should be guided by rigorous, locally-validated cross-verification studies as detailed in this guide, ensuring that quality assurance and diagnostic accuracy remain the highest priorities.
Performance Validation: Statistical Comparison and Diagnostic Agreement Metrics
This comparison guide provides an objective evaluation of the diagnostic performance between the fully automated FA280 digital feces analyzer and the manual formalin-ethyl acetate concentration technique (FECT) for intestinal parasite detection. Through systematic analysis of comparative studies, we assess agreement metrics (kappa statistics), sensitivity, specificity, and operational characteristics of both methods. The FA280 demonstrates strong species identification agreement with FECT when combined with expert audit, though variations in absolute sensitivity occur due to stool sample size differences. This analysis provides researchers and clinical laboratory professionals with evidence-based data to inform diagnostic protocol selection for parasitic infection detection and surveillance.
Intestinal parasitic infections remain a significant global health burden, causing malnutrition, anemia, and impaired cognitive development [1]. Accurate diagnosis is fundamental for treatment, yet many regions rely on traditional microscopic methods that are labor-intensive and operator-dependent [1] [34]. The formalin-ethyl acetate concentration technique (FECT) has been widely regarded as a sensitive manual method for detecting parasites in stool samples [34]. Recently, automated solutions like the Orienter Model FA280 digital feces analyzer have emerged to address limitations of conventional microscopy [3] [1].
This guide provides a systematic comparison between FA280 and FECT within the broader research context of diagnostic method validation. We present quantitative performance data including kappa statistics for inter-method agreement, sensitivity, specificity, and detailed experimental protocols from recent studies. The analysis aims to support researchers, scientists, and drug development professionals in evaluating these diagnostic platforms for clinical studies and public health interventions.
Methodological Approaches
Formalin-Ethyl Acetate Concentration Technique (FECT)
The FECT procedure follows standardized parasitological methods [1]. Approximately 2 grams of stool sample is emulsified in 10 mL of 10% formalin. The suspension is filtered through gauze into a 15-mL conical centrifuge tube. Next, 3 mL of ethyl acetate is added, the tube is tightly capped and vigorously shaken for 1 minute. After centrifugation at 500-2500 × g for 2-5 minutes, the debris plug formed at the tube's top is freed using an applicator stick, and the supernatant layers are decanted [16] [1]. Residual debris is removed from the tube walls with a cotton-tipped applicator, and the sediment is examined microscopically for parasites [1].
FA280 Fully Automatic Digital Feces Analyzer
The FA280 system automates stool processing and analysis through several integrated units [1]. The system uses approximately 0.5-1.0 grams of stool placed in a filtered collection tube. An automatic in-sample unit with track-type carrier handles sample loading. A pneumatic mixing system ensures homogenization with diluent. A high-resolution camera captures sample characteristics and color. The microscope unit employs high- and low-power objectives with multifield tomography imaging to capture detailed sectional images. Captured digital images are automatically evaluated by an artificial intelligence (AI) program, with results subject to optional audit by skilled technicians [3] [1]. The process is based on a simple sedimentation technique, with a throughput of approximately 40 samples per 30-minute run [1].
Experimental Designs in Comparative Studies
Recent comparative studies have employed varied experimental designs. One mixed-method study evaluated FA280 for clonorchiasis diagnosis using 1,000 participants, comparing results with the Kato-Katz method and incorporating qualitative interviews with healthcare professionals [20]. Another major comparison study utilized two sample sets: 200 fresh stool samples and 800 formalin-preserved samples, with both FA280 (AI and expert-audited results) and FECT performed on all samples [3] [1]. Performance was assessed through statistical measures including McNemar's test for pairwise agreement and kappa statistics for species identification concordance [3] [1].
Comparative Performance Metrics
Diagnostic Agreement and Kappa Statistics
Kappa statistics measure inter-method agreement beyond chance, with values ranging from 0 (no agreement) to 1 (perfect agreement). Studies demonstrate varying agreement levels between FA280 and FECT based on analysis type and parasite species.
Table 1: Kappa Agreement Values Between FA280 and FECT for Parasite Detection
| Sample Type | Analysis Method | Parasite Category | Kappa Value (κ) | Agreement Interpretation |
|---|---|---|---|---|
| Fresh stool | FA280 AI vs. FECT | All parasites | 0.367 (95% CI: 0.248-0.486) | Fair agreement |
| Fresh stool | FA280 with user audit vs. FECT | All parasites | 1.00 (95% CI: 1.00-1.00) | Perfect agreement |
| Preserved stool | FA280 with user audit vs. FECT | Helminths | 0.857 (95% CI: 0.82-0.894) | Strong agreement |
| Preserved stool | FA280 with user audit vs. FECT | Protozoa | 1.00 (95% CI: 1.00-1.00) | Perfect agreement |
| Preserved stool | FA280 vs. Kato-Katz (clonorchiasis) | Clonorchis sinensis | 0.82 (95% CI: 0.76-0.88) | Strong agreement [20] |
The diagnostic agreement between methods shows significant improvement with expert intervention. While the FA280's AI-based analysis alone showed only fair agreement with FECT (κ = 0.367), the implementation of a user audit by skilled technicians improved agreement to perfect levels (κ = 1.00) for fresh stool samples [3] [1]. For preserved stool samples, species-specific agreement remained strong to perfect, particularly for protozoa (κ = 1.00) and helminths (κ = 0.857) [1].
Sensitivity and Specificity Profiles
Sensitivity and specificity metrics vary between FA280 and FECT, influenced by sample processing characteristics and detection methodologies.
Table 2: Sensitivity and Detection Rates of FA280 Versus FECT
| Performance Parameter | FA280 with User Audit | FECT | Notes |
|---|---|---|---|
| Positive detection (preserved samples) | Significantly lower (P < 0.001) | Significantly higher | 800 preserved samples [1] |
| Positive detection (fresh samples) | No significant difference (P = 1) | No significant difference | 200 fresh samples [1] |
| Clonorchis sinensis detection | 10.0% positive rate | 10.0% positive rate (Kato-Katz) | 96.8% agreement [20] |
| Hookworm detection | 145 positive | 89 positive | FECT superior (P < 0.001) [16] |
| Trichuris trichiura detection | 109 positive | 53 positive | FECT superior (P < 0.001) [16] |
| Small liver flukes detection | 85 positive | 39 positive | FECT superior (P < 0.001) [16] |
| Ascaris lumbricoides detection | 50 positive | 57 positive | No significant difference (P = 0.546) [16] |
The disparity in detection rates between methods is potentially attributed to differences in stool sample size, with FECT utilizing approximately 2 grams compared to FA280's 0.5-1.0 grams [5] [1]. This quantitative advantage in sample processing likely enhances FECT's sensitivity for certain parasites, particularly in preserved samples [1]. For helminths with high egg density like Ascaris lumbricoides, both methods show comparable detection capability [16].
Operational Characteristics and Practical Implementation
Beyond diagnostic performance, operational factors significantly impact method selection for different laboratory settings.
Table 3: Operational Comparison Between Diagnostic Methods
| Operational Parameter | Direct Wet Smear | FECT | FA280 with User Audit |
|---|---|---|---|
| Stool sample weight | 0.2 g [5] | 2 g [5] | 0.5-1 g [5] |
| Technique type | Manual [5] | Manual [5] | Automatic [5] |
| Process complexity | Less complicated [5] | More complicated [5] | Less complicated [5] |
| Processing time per sample | 2 minutes [5] | 8-10 minutes [5] | 2 minutes [5] |
| Parasite observation time | 5-10 minutes [5] | 5-10 minutes [5] | 3-5 minutes [5] |
| Observation tool | Microscope [5] | Microscope [5] | High-resolution images [5] |
| Required expertise | Experienced technician [5] | Experienced technician [5] | Experienced technician [5] |
| Data storage | No [5] | No [5] | Yes [5] |
| Cost per test | USD 0.25 [5] | USD 0.50 [5] | USD 2.00 [5] |
The FA280 system offers significant advantages in processing efficiency, reduced hands-on time, and automated data storage [5]. However, these benefits come at a higher per-test cost compared to conventional methods [5]. Qualitative assessments from healthcare professionals indicate that FA280 outperforms FECT in testing procedures, detection results, and user acceptance, though challenges in cost and implementation scalability were noted [20].
Research Reagent Solutions and Essential Materials
Table 4: Key Research Reagents and Materials for Parasitological Diagnosis
| Reagent/Material | Application | Function | Method Compatibility |
|---|---|---|---|
| 10% formalin | Sample preservation | Preserves parasite morphology, inhibits further development of helminth ova and larvae | FECT, FA280 (preserved samples) |
| Ethyl acetate | Solvent extraction | Extracts fats and debris, concentrates parasitic elements in sediment | FECT |
| Hexadecyltrimethylammonium bromide (CTAB) | Surfactant | Enhances parasite recovery in flotation techniques | Dissolved air flotation (DAF) [29] |
| Polyvinyl alcohol (PVA) | Sample preservation | Preserves stool samples for permanent stained smears | Conventional microscopy [34] |
| Lugol's iodine solution | Staining | Enhances contrast for microscopic identification of protozoal cysts | Direct wet mount, FA280 [29] |
| Trichrome stain | Permanent staining | Provides polychromatic contrast for protozoan identification | Permanent stained smears [34] |
| Modified acid-fast stain | Special staining | Identifies coccidian parasites (Cryptosporidium, Cyclospora) | Specialized detection [34] |
| Gauze or strainer (400μm, 200μm) | Filtration | Removes large debris while allowing parasite passage | FECT, DAF [16] [29] |
| Conical centrifuge tubes (15mL) | Sample processing | Facilitates concentration and sediment formation | FECT, DAF |
The comparative analysis between FA280 and FECT reveals a nuanced performance profile. The FA280 system with expert audit demonstrates strong to perfect agreement with FECT for species identification, particularly for protozoa and helminths, while offering advantages in automation, processing time, and reduced laboratory contamination [1]. However, FECT maintains higher absolute sensitivity for parasite detection overall, attributable to its larger stool sample size [1]. This sensitivity advantage makes FECT particularly valuable in low-prevalence settings or for low-intensity infections.
For research applications, the choice between methods depends on specific study requirements. The FA280 offers high-throughput, standardized analysis with digital archiving capabilities beneficial for large-scale surveillance studies and clinical trials requiring documentation. FECT remains a cost-effective, highly sensitive option for field studies and resource-limited settings. Future methodological improvements for FA280 should focus on enhancing AI algorithm performance, optimizing sample preparation to increase sensitivity, and reducing operational costs to improve accessibility for broader research implementation.
The diagnosis of intestinal parasitic infections remains a cornerstone of public health, particularly in tropical and subtropical regions where these infections contribute significantly to morbidity, including malnutrition, anemia, and impaired cognitive development [1] [19]. For decades, the formalin-ethyl acetate concentration technique (FECT) has served as a fundamental microscopic method for concentrating and detecting parasites in stool specimens, balancing sensitivity with practical feasibility [6]. However, this traditional technique is labor-intensive, time-consuming, and highly dependent on the expertise of trained microscopists [1]. The recent introduction of fully automated digital feces analyzers, such as the Orienter Model FA280, promises a paradigm shift in diagnostic parasitology. These systems leverage artificial intelligence (AI) and digital imaging to automate stool examination, offering the potential for high-throughput testing and reduced operator dependency [1] [35]. This guide provides a systematic, data-driven comparison of the species-specific detection efficacy of the FA280 digital feces analyzer versus the well-established FECT, providing researchers and scientists with an objective analysis of their performance characteristics for both helminth and protozoan parasites.
Experimental Protocols and Methodologies
A clear understanding of the underlying procedures is essential for interpreting performance data. The following sections detail the standard protocols for the two techniques under comparison.
Formalin-Ethyl Acetate Concentration Technique (FECT)
The FECT is a sedimentation concentration method that uses formalin to fix specimens and ethyl acetate to separate debris from parasitic elements [6]. The procedure, as standardized by institutions like the CDC, is as follows [6]:
- Emulsification: Approximately 1-2 grams of stool is emulsified in 10 mL of 10% formalin.
- Filtration: The suspension is strained through a cheesecloth-type gauze (or a specialized sieve like the Parasep concentrator with a ~425μm pore size) into a 15 mL conical centrifuge tube to remove large particulate matter [6] [36].
- Solvent Addition: The filtrate is supplemented with 3-4 mL of ethyl acetate (or diethyl ether), the tube is stoppered, and shaken vigorously to form an emulsion.
- Centrifugation: The tube is centrifuged at a recommended 500 × g for 5-10 minutes. This step creates four layers: an ethyl acetate layer, a debris plug, a formalin layer, and a sediment containing the parasites [6] [37].
- Sediment Examination: The top three layers are discarded, and the sediment is resuspended in a small volume of formalin or saline. The final sediment is examined under a microscope for ova, cysts, and larvae [6].
The method's effectiveness is attributed to the concentration of parasites from a larger stool sample, thereby increasing the probability of detection.
FA280 Fully Automatic Digital Feces Analyzer
The FA280 system automates the entire process of fecal analysis, from sample preparation to AI-powered detection [1] [2]. Its workflow integrates several advanced technological units:
- Automatic In-Sample Unit: A track-type carrier ensures consistent and accurate loading of samples.
- Sampling and Mixing Unit: A pneumatic system thoroughly mixes the sample with a diluent.
- Imaging Units: High-resolution cameras capture the sample's macroscopic characteristics (color, consistency) and microscopic details via a microscope unit with high- and low-power objective lenses. This unit employs multifield tomography to capture detailed sectional images.
- AI Analysis: Digital microscope images are automatically analyzed by a convolutional neural network (CNN) model trained to detect and classify a wide range of parasitic organisms [1] [35]. The system can process a batch of 40 samples in approximately 30 minutes [1].
- User Audit: A critical feature allows a skilled medical technologist to review the digital images and the AI's findings, which has been shown to significantly improve accuracy [1] [23].
The FA280 operates on a simple sedimentation principle, though it uses a smaller sample size (approximately 0.5 g) compared to the standard FECT [1] [2].
Comparative Performance Data
Direct comparative studies reveal distinct performance profiles for the FA280 and FECT, which vary significantly by parasite species and the use of AI versus human audit.
The following table summarizes the overall detection rates of the two methods from key studies.
| Methodology | Sample Type (n) | Overall Detection Rate | Key Study Findings | Citation |
|---|---|---|---|---|
| FECT | 200 fresh samples | Benchmark | Considered the reference standard in the study. | [1] |
| FA280 (AI Report) | 200 fresh samples | Significantly lower | Statistically significant difference vs. FECT (P < 0.001). | [1] |
| FA280 (User Audit) | 200 fresh samples | No significant difference | No statistically significant difference from FECT (P = 1). | [1] |
| FECT | 800 preserved samples | Significantly higher | Detected more positives than FA280 with user audit (P < 0.001); attributed to larger sample size. | [1] |
| FA280 (vs. Kato-Katz) | 1000 community samples | No significant difference | 96.8% agreement for C. sinensis detection (κ = 0.82). | [2] |
Species-Specific Agreement
The agreement between methods is not uniform across all parasites. The kappa (κ) statistic, which measures agreement beyond chance, highlights these species-specific variations. A κ value of 0.81-1.00 is considered almost perfect agreement, 0.61-0.80 substantial, 0.41-0.60 moderate, and 0.21-0.40 fair.
Table: Agreement in Species Identification between FA280 (with User Audit) and FECT [1]
| Parasite Group | Kappa (κ) Value | Agreement Level |
|---|---|---|
| Helminths | 0.857 (95% CI: 0.82–0.894) | Strong / Almost Perfect |
| Protozoa | 1.00 (95% CI: 1.00–1.00) | Perfect |
The perfect agreement for protozoa and strong agreement for helminths with a user audit demonstrates that the image quality generated by the FA280 is sufficient for accurate identification by a trained technologist. However, the AI's autonomous performance requires further refinement, as initial agreement with FECT was only fair (Overall agreement = 75.5%, κ = 0.367) [1].
The Scientist's Toolkit: Key Research Reagents and Materials
Successful implementation of these diagnostic techniques relies on specific materials and reagents. The following table outlines essential components for the featured methods.
Table: Essential Research Reagents and Materials for FECT and FA280 Protocols
| Item | Function/Description | Application |
|---|---|---|
| 10% Formalin | Fixative solution that preserves parasitic morphology. | Used in both FECT and as a preservative for samples prior to FA280 analysis. [1] [6] |
| Ethyl Acetate / Diethyl Ether | Organic solvent that dissolves fats and debris, forming a separate layer during centrifugation to clean the sample. | Critical for the FECT protocol. Ethyl acetate is preferred over ether due to lower flammability. [6] [7] |
| Parasep Faecal Parasite Concentrator | An enclosed, disposable filtration and concentration system. Includes a filter thimble (e.g., 425μm pore size) and sedimentation cone. | Reduces laboratory contamination and standardizes the FECT process, improving safety and reproducibility. [36] |
| FA280 Test Kit & Diluent | Proprietary consumables for the automated analyzer. | Contains the filtered sample collection tubes and specific diluents required for the FA280's pneumatic mixing and imaging process. [1] |
| Convolutional Neural Network (CNN) Model | AI software trained on diverse parasite image libraries for automated detection and classification. | The core of the FA280's analytical capability, enabling high-throughput screening of digital slide images. [35] |
Workflow and Logical Process Diagrams
The fundamental difference between the two methods lies in their workflow: one is a manual laboratory procedure, while the other is an integrated automated process. The following diagram illustrates the operational and logical workflow of the FA280 system, which consolidates multiple manual steps into a single automated instrument.
To contextualize the performance findings, it is critical to understand the technological backbone of the FA280, particularly its use of a pre-trained Convolutional Neural Network (CNN). The following diagram outlines the architecture and training process of the AI model that enables its detection capabilities.
The experimental data reveals a nuanced performance landscape. The formalin-ethyl acetate concentration technique (FECT) maintains high sensitivity, particularly due to its use of a larger stool sample (1-2 g), which provides a greater opportunity to detect low-intensity infections [1]. Its strengths are well-established protocol, high sensitivity with adequate sampling, and lower cost per test. However, its limitations are significant and include being labor-intensive, time-consuming, subject to inter-technician variability, and involving potential exposure to hazardous chemicals [1] [6].
The FA280 automated analyzer addresses several of FECT's limitations. Its primary advantages are high-throughput capacity (40 samples in ~30 min), reduced manual labor and technical dependency, minimized operator exposure to specimens, and generation of digital records for audit and re-evaluation [1] [2]. Its limitations include a higher cost per test, a potentially lower sensitivity attributed to the smaller sample size (~0.5 g), and the current necessity of a user audit to achieve optimal, reliable agreement with reference methods [1] [23].
In conclusion, the choice between the FA280 and FECT is context-dependent. For high-volume laboratories with sufficient funding, where throughput, workflow efficiency, and staff safety are priorities, the FA280 with a mandatory user audit presents a compelling modern alternative. Its species-specific performance for common helminths and protozoa, once verified by a technologist, is on par with FECT [1] [2]. For resource-limited settings, large-scale epidemiological surveys where cost is a primary constraint, or for detecting very low-intensity infections, the traditional FECT remains a highly sensitive and cost-effective benchmark. The evolution of the FA280's AI algorithms, particularly with training on larger and more diverse datasets, is poised to further bridge the current sensitivity gap and solidify the role of automation in the future of diagnostic parasitology.
Within the field of clinical parasitology, the accurate diagnosis of intestinal helminth infections is a cornerstone of effective public health interventions and patient care. The diagnostic sensitivity of any method is not absolute but is significantly influenced by the intensity of the infection, commonly measured by the number of eggs per gram (EPG) of stool. This analytical comparison examines the performance correlation between a novel, fully automated digital feces analyzer, the Orienter Model FA280, and the long-established reference method, the Formalin-Ethyl Acetate Concentration Technique (FECT). The focus is on their respective diagnostic efficiencies in low versus high egg burden scenarios, a critical distinction for epidemiological surveys, drug efficacy evaluations, and the management of neglected tropical diseases [32] [2] [12]. The automation and artificial intelligence (AI)-driven analysis of the FA280 present a potential paradigm shift in laboratory diagnostics, promising high-throughput testing while reducing operational burdens. However, its operational parameters, particularly the smaller stool sample size it processes, necessitate a rigorous, context-dependent evaluation of its capabilities, especially when compared to the robust, albeit labor-intensive, FECT [32] [2].
Methodology
Formalin-Ethyl Acetate Concentration Technique (FECT)
The FECT is a sedimentation-based concentration method widely regarded as a standard for parasitic diagnosis due to its high sensitivity. The procedure, as outlined by the CDC and employed in comparative studies, is as follows [6] [32] [12]:
- Sample Preparation: Approximately 2 to 5 grams of stool is emulsified in 10 mL of 10% formalin. The mixture is then filtered through a sieve or gauze (pore size ~450-500μm) into a 15 mL conical centrifuge tube to remove large particulate debris.
- Solvent Addition and Centrifugation: To the filtered suspension, 3 to 4 mL of ethyl acetate is added. The tube is sealed and shaken vigorously for 30 seconds to 1 minute to facilitate the extraction of fats and debris into the organic solvent layer. The tube is then centrifuged at 500 × g for 5-10 minutes.
- Post-Centrifugation Processing: After centrifugation, the tube contains four distinct layers: a pellet of sediment (containing parasites) at the bottom, a layer of formalin, a plug of debris, and an upper layer of ethyl acetate. The debris plug is freed by ringing the tube with an applicator stick, and the top three layers are decanted.
- Microscopic Examination: The remaining sediment is resuspended in a small volume of formalin or saline. The suspension is examined microscopically for the presence of parasites, cysts, and ova using saline and iodine wet mounts. The entire process is manual and requires significant expertise from a trained microscopist [6] [32].
FA280 Fully Automatic Digital Feces Analyzer
The FA280 system automates the process of fecal analysis, integrating AI to minimize manual labor. Its methodology is based on a simplified sedimentation technique and operates as follows [32] [2]:
- Automated Sample Processing: A smaller stool sample (approximately 0.5 g) is placed in a proprietary collection tube. The instrument automatically performs pneumatic mixing with a diluent, character and color photographing of the sample, and preparation of the sample for microscopic analysis.
- Digital Imaging and AI Analysis: The analyzer uses high-resolution microscopy with multifield tomography to capture digital images of the prepared sample. These images are then automatically evaluated by a built-in AI program to identify and classify parasitic structures.
- User Audit Function: The system allows for a crucial secondary step: a skilled medical technologist can review the digital images generated by the instrument to audit and verify the AI-generated report, enhancing diagnostic accuracy.
- Throughput: The FA280 processes batches of up to 40 samples in a single run, which is completed in approximately 30 minutes, offering a significant advantage in throughput [32].
Table 1: Core Methodological Differences Between FECT and the FA280 Analyzer
| Parameter | Formalin-Ethyl Acetate (FECT) | FA280 Automated Analyzer |
|---|---|---|
| Sample Size | 2 - 5 g [32] [12] | ~0.5 g [32] [2] |
| Concentration Principle | Formalin-ethyl acetate sedimentation [6] | Automated sedimentation [32] |
| Parasite Detection | Manual microscopy by technologist | Automated digital imaging & AI analysis [32] |
| Key Procedural Steps | Filtration, solvent addition, centrifugation, decanting [6] | Automated mixing, imaging, and software analysis [32] |
| Throughput | Laborious; ~10-15 minutes per sample [8] | High; 40 samples in ~30 minutes [32] |
| Expertise Dependency | High (requires trained microscopist) [32] | Lower (after initial setup; user audit advised) [32] |
Experimental Workflow for Comparative Studies
The following diagram illustrates the typical workflow used in studies that directly compare the diagnostic performance of FECT and the FA280 analyzer, highlighting the points of comparison relevant to infection intensity.
Results & Performance Comparison
Initial studies indicate that the overall agreement between the FA280 and established methods like FECT and Kato-Katz (KK) can be strong, but this is highly dependent on protocol and the inclusion of a human expert in the loop.
- A study comparing the FA280 with FECT on 200 fresh stool samples found that the AI report alone showed only fair agreement (κ = 0.367) with FECT. However, when the same samples were subjected to a user audit of the FA280's digital images, agreement with FECT became perfect (κ = 1.00) [32].
- In a community-based study of 1000 participants for Clonorchis sinensis detection, the FA280 demonstrated strong agreement (κ = 0.82) with the KK method, with no significant difference in positive rate (10.0% for both) [2].
Performance Stratified by Infection Intensity
The critical differentiator in performance emerges when results are stratified by the intensity of infection, as measured by EPG.
- High Infection Intensity: In high-burden scenarios, the performance gap between methods narrows. The FA280 demonstrates high agreement with reference methods. For example, in Clonorchis detection, agreement between FA280 and KK was significantly higher in high infection intensity groups compared to low-intensity groups [2]. Similarly, for Ascaris lumbricoides, which often presents with high egg counts, one study found no significant difference in detection between FECT and a simple formalin concentration method, suggesting high burdens are easily detected even by less sensitive techniques [12].
- Low Infection Intensity: This is where the FA280's smaller sample size becomes a significant limitation. A large study on 800 preserved samples found that FECT detected significantly more positive samples for parasites than the FA280 with a user audit (McNemar’s test, P < 0.001). The authors concluded this disparity was likely because FECT uses a larger stool sample (2g vs 0.5g), thereby increasing the probability of detecting parasites when they are scarce [32]. This is consistent with the finding that the KK method, which also quantifies eggs, can detect low-intensity infections that might be missed by other methods [2].
Table 2: Quantitative Performance Data in Low vs. High Intensity Infections
| Infection / Organism | Comparison Method | Performance in High Intensity | Performance in Low Intensity |
|---|---|---|---|
| Clonorchis sinensis [2] | Kato-Katz (KK) | High Agreement with KK (P < 0.05) | Lower agreement with KK; KK potentially more sensitive |
| Soil-transmitted helminths (Hookworm, T. trichiura) [12] | Formalin Concentration (FC) | Data not specifically stated for high intensity | FECT superior to FC for detection |
| General Parasite Detection [32] | FECT | Data not specifically stated for high intensity | FECT detected significantly more positives than FA280 (P < 0.001) |
| Ascaris lumbricoides [12] | Formalin Concentration (FC) | No significant difference between FECT and FC | Data not specifically stated for low intensity |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Materials and Reagents for Parasitology Diagnostics
| Item | Function / Description |
|---|---|
| 10% Formalin [6] [32] | A universal preservative for stool specimens; fixes parasites and prevents degradation of morphology. |
| Ethyl Acetate [6] [12] | A solvent used in FECT to extract fats, oils, and debris from the fecal suspension, resulting in a cleaner sediment for microscopic examination. It is preferred over ether due to lower flammability. |
| Conical Centrifuge Tubes [6] [36] | Tubes (typically 15 mL) used for the concentration steps in both FECT and commercial concentrator systems. |
| Parasep SF Faecal Concentrator [8] [36] | A commercial, enclosed filtration and concentration system that simplifies the FECT process, improves safety, and reduces processing time. |
| Alcorfix / PROTO-FIX [8] [22] | Alcohol-based or proprietary fixatives used as alternatives to formalin in some commercial systems. They eliminate the use of formalin and may better preserve trophozoite morphology [22]. |
| Modified Ziehl-Neelsen (MZN) Stain [38] | A special stain used to identify acid-fast oocysts of coccidian parasites like Cryptosporidium spp., Cyclospora cayetanensis, and Cystoisospora belli in concentrated stool sediments. |
The comparative analysis between the FA280 and FECT reveals a nuanced picture where the optimal diagnostic tool is dictated by the specific clinical or research context, particularly the expected parasite burden.
The FA280 analyzer offers transformative advantages in settings where throughput, speed, and operator safety are priorities. Its ability to process batches automatically reduces hands-on time, minimizes exposure to infectious samples, and standardizes the initial screening process [32]. The digital image audit function is a critical feature, bridging the gap between automated screening and expert confirmation. However, its primary limitation is a lower sensitivity in low-intensity infections, a direct consequence of the smaller sample volume analyzed [32]. Therefore, its application is best suited for routine clinical laboratories with moderate to high sample loads or in settings where high-intensity infections are prevalent.
Conversely, the FECT method remains the benchmark for maximum sensitivity, especially in low-burden scenarios and for epidemiological studies where detecting every positive case is paramount [32] [12]. Its use of a larger sample size provides a greater chance of detecting scant infections. The drawbacks of FECT are its manual, time-consuming nature, higher skill requirement, and increased risk of laboratory contamination [32] [8].
In conclusion, the correlation between infection intensity and diagnostic performance is clear: while the FA280 represents a significant step forward in laboratory automation for parasitology, the traditional FECT retains superior sensitivity for low egg burden scenarios. The choice between them should be informed by a clear understanding of the target population's infection prevalence, the purpose of the testing (e.g., clinical diagnosis vs. surveillance for elimination), and the available laboratory resources. For the most critical applications, particularly in low-prevalence settings or elimination campaigns, FECT or a method using a comparable sample size remains an essential reference standard.
Within the field of diagnostic parasitology, the accurate and consistent detection of intestinal parasites is fundamental to public health initiatives, patient care, and pharmaceutical development. For decades, the formalin-ethyl acetate concentration technique (FECT) has served as a cornerstone manual method for stool examination, despite its labor-intensive and time-consuming nature [3]. The recent introduction of fully automated digital analyzers, such as the Orienter Model FA280, promises to revolutionize this field by integrating artificial intelligence (AI) and high-throughput automation [33]. This guide objectively compares the performance of the FA280 against the traditional FECT, with a specific focus on a critical metric for adoption in research and development: consistency across diverse laboratory settings and varying levels of technician expertise. The synthesis of multi-center validation data provides crucial insights for scientists and drug development professionals evaluating these technologies for use in clinical trials and large-scale surveillance studies.
Experimental Protocols & Methodologies
A clear understanding of the underlying methodologies is essential for interpreting the comparative data.
The FA280 Automated Digital Feces Analyzer
The FA280 system automates the entire process of stool analysis. It utilizes intelligent sample dilution and high-frequency pneumatic mixing to standardize various sample consistencies. Following preparation, the system employs a CMOS microscope to capture high-resolution, multi-field tomographic images [33]. These images are then analyzed by a proprietary AI program to automatically identify and classify parasite eggs and cysts. The system features cyclic loading, allowing for the batch processing of up to 50 samples, and its test kit system supports batches of up to 300 samples, significantly enhancing throughput [33]. A key feature is the "user audit," where a skilled medical technologist can review the digital images generated by the FA280 to verify or correct the AI's findings [3].
The Formalin-Ethyl Acetate Concentration Technique (FECT)
FECT is a manual sedimentation concentration method designed to separate parasites from fecal debris. The standard protocol, as outlined by the CDC, involves straining a formalin-preserved stool sample through gauze into a conical tube [6]. After centrifugation, the supernatant is decanted, and the sediment is resuspended in formalin. Ethyl acetate is then added, the tube is shaken vigorously, and a second centrifugation step is performed. This process concentrates parasitic elements in the sediment, which is then examined under a microscope by a technician for identification and counting [6]. This method is technically robust but relies heavily on the skill and experience of the laboratory personnel.
Comparative Performance Data
The following tables summarize key quantitative findings from recent studies directly comparing the FA280 and FECT.
Detection Agreement and Operational Comparison
Table 1: Agreement in Parasite Detection Between FA280 and FECT
| Sample Set & Method | Statistical Agreement with FECT | Key Findings |
|---|---|---|
| 200 Fresh Samples: FA280 with User Audit | Perfect agreement (κ = 1.00) [3] | No significant difference in positive sample detection [3]. |
| 200 Fresh Samples: FA280 with AI Report Only | Fair agreement (κ = 0.367, Overall agreement = 75.5%) [3] | Statistically significant difference in detection rates vs. FECT [3]. |
| 800 Preserved Samples: FA280 with User Audit | FECT detected significantly more positives (P < 0.001) [3] | Disparity attributed to FECT's use of larger stool samples (2g vs. 0.5g) [3]. |
| 1000 Samples: FA280 vs. Kato-Katz (KK) | Strong agreement (κ = 0.82) [2] | No significant difference in positive rate for Clonorchis sinensis detection (P > 0.999) [2]. |
Table 2: Operational and Practical Comparison
| Parameter | Formalin-Ethyl Acetate Technique (FECT) | FA280 Feces Analyzer |
|---|---|---|
| Stool Sample Weight | 2 g [5] | 0.5 - 1 g [5] |
| Technique | Manual [3] | Fully Automatic [3] |
| Process Simplicity | More complicated [5] | Less complicated [5] |
| Total Processing Time | 8-10 min/sample [5] | ~2 min/sample [5] |
| Parasite Observation Time | 5-10 min [5] | 3-5 min [5] |
| Result Storage | No [5] | Yes, digital images [5] |
| Estimated Cost per Test | USD 0.50 [5] | USD 2.00 [5] |
Consistency Across Technician Expertise
A critical finding from the validation studies is the impact of human expertise on the FA280's performance. When the FA280's AI software operated without human oversight, its agreement with the FECT reference method was only fair (κ = 0.367) [3]. However, when the same digital images were subjected to a user audit by a skilled technologist, the agreement with FECT reached perfect levels (κ = 1.00) for fresh samples [3]. This demonstrates that the current optimal use of the FA280 involves a hybrid model where the AI serves as a high-throughput pre-screener, and expert technicians provide final validation, ensuring results are consistent with those from highly trained specialists using traditional methods.
The Scientist's Toolkit
Table 3: Essential Research Reagents and Materials for Stool Parasitology
| Item | Function in Protocol |
|---|---|
| 10% Formalin | A universal preservative that fixes stool specimens, maintaining the morphology of parasites for later examination [6]. |
| Ethyl Acetate | An organic solvent used as a lipid-dissolving agent in the FECT procedure to clear debris from the fecal concentrate [6]. |
| Ethyl Acetate Sedimentation Tubes | Specialized centrifuge tubes used in the FECT protocol for the concentration of parasitic elements [6]. |
| Orienter FA280 Test Kits | Proprietary kits containing filtered sample collection tubes and diluents designed for use with the automated FA280 system [2]. |
| Alcorfix | An alcohol-based fixative used in some commercial concentrator tubes (e.g., Parasep) as a formalin-free alternative for sample preservation [8]. |
Workflow and Signaling Pathways
The fundamental difference between the two methods lies in their operational workflows, from sample preparation to result generation. The following diagram illustrates these distinct pathways.
The multi-center validation data reveals a nuanced picture of the FA280's performance relative to the established FECT method. The FA280 demonstrates strong operational advantages in speed, throughput, and laboratory safety due to its automated, sealed system [3] [5]. For specific parasites like Clonorchis sinensis, it shows statistically equivalent detection performance to the Kato-Katz method [2]. However, its absolute sensitivity can be lower than FECT when the latter uses larger stool samples, highlighting the impact of sample size on detection limits [3].
Regarding consistency across laboratory environments—the core of this analysis—the data indicates that the FA280 excels at standardizing output. It reduces hands-on time and minimizes the direct impact of a technician's skill on the mechanical processing of samples. The hybrid AI-plus-audit model effectively transfers the requirement for expert knowledge from the microscope to the digital screen, potentially making high-quality diagnostics more accessible in settings where specialist microscopists are scarce. In conclusion, the FA280 represents a significant step forward in diagnostic parasitology. For researchers and drug developers, it offers a reliable, high-throughput tool for large-scale studies, provided its operational parameters—particularly the use of a expert user audit and consideration of sample size—are incorporated into the study design to ensure data consistency and reliability.
The diagnosis of intestinal parasitic infections (IPIs) remains a critical public health issue, particularly in tropical and subtropical regions where these infections are highly prevalent. The gold standard for diagnosis involves the microscopic detection of parasites in stool samples, yet no single method is universally accepted as the definitive standard [2]. Conventional techniques, primarily the Kato-Katz (KK) method and the formalin-ethyl acetate concentration technique (FECT), have been widely used for decades. While these methods are considered reference standards in various settings, they are labor-intensive, time-consuming, and heavily reliant on the expertise of trained microscopists [2] [3]. The FA280 digital feces analyzer (Sichuan Orienter Bioengineering Co., Ltd.) represents a technological advancement, automating the process through intelligent sample dilution, high-frequency pneumatic mixing, and artificial intelligence (AI)-driven parasite egg identification via high-resolution imaging [2]. This guide provides a comparative analysis of the FA280's performance against the KK method and FECT, synthesizing current experimental data to inform researchers, scientists, and drug development professionals.
Methodological Protocols
A direct comparison of diagnostic techniques requires a clear understanding of their respective experimental protocols. The methodologies for the KK method, FECT, and the FA280 analyzer differ significantly in their procedural steps, sample requirements, and technological integration.
Kato-Katz (KK) Method Protocol
The KK technique is a standardized, quantitative method widely used in epidemiological surveys for soil-transmitted helminths [2]. The detailed protocol is as follows:
- Sample Preparation: A plastic template with a 6-mm diameter hole (holding approximately 41.7 mg of sieved stool) is placed on a glass slide.
- Smear Preparation: The template is filled with sieved stool, and the excess is scraped off. The template is then removed, leaving a precise amount of feces on the slide.
- Cellophane Covering: A glycerin-soaked or malachite green-soaked cellophane strip is placed over the sample to clear the debris and enable visualization of parasite eggs.
- Microscopic Examination: After a clearing period (typically 30 minutes to several hours), the slide is examined under a microscope by experienced technicians who identify and count the eggs. The count is used to calculate the eggs per gram (EPG) of feces to estimate infection intensity [2].
Formalin-Ethyl Acetate Concentration Technique (FECT) Protocol
FECT is a concentration method that improves detection sensitivity by removing debris and concentrating parasitic elements [12].
- Sample Emulsification: Approximately 2 grams of stool is emulsified in a 10% formalin solution to preserve parasitic structures.
- Filtration and Concentration: The suspension is filtered through a strainer (e.g., with 0.6 mm x 0.6 mm sieve openings) to remove large debris.
- Ethyl-Acetate Addition: The filtered sample is transferred to a conical tube, and 4 mL of ethyl-acetate is added. The tube is vigorously shaken for 30 seconds to extract fat and debris into the organic solvent layer.
- Centrifugation and Examination: The tube is centrifuged (e.g., at 500 g for 5 minutes), resulting in a layered solution. The top layers of ethyl-acetate, debris, and formalin are decanted. The remaining sediment, containing concentrated parasites, is resuspended in saline, and a wet mount is prepared for microscopic examination [12].
FA280 Automated Feces Analyzer Protocol
The FA280 automates the staining and examination process, reducing manual intervention [2] [3].
- Sample Loading: A filtered sample collection tube containing approximately 0.5-1 gram of stool is loaded into the device.
- Automated Processing: The instrument automatically adds a diluent, performs high-frequency pneumatic mixing to homogenize the sample, and uses sedimentation and concentration technology.
- Imaging and AI Analysis: The device's microscope automatically focuses and captures high-resolution images through multi-field tomography. The built-in AI software analyzes these images in real-time based on the color, shape, and consistency of parasitic structures to generate a report.
- User Audit: The system allows for a "user audit," where a skilled medical technologist can review the acquired digital images to confirm or override the AI-generated findings, enhancing diagnostic accuracy [3].
The workflow below illustrates the key steps involved in the FA280 diagnostic process and its comparative position alongside traditional methods:
Comparative Performance Data
The diagnostic performance of the FA280 has been evaluated against both the KK method and FECT in multiple studies. The following tables summarize key quantitative comparisons, including agreement statistics, operational characteristics, and performance across different infection intensities.
Table 1: Comparative Agreement of FA280 with Reference Standard Methods
| Comparison | Study Population (n) | Agreement Metric | Result | Interpretation | Citation |
|---|---|---|---|---|---|
| FA280 vs. Kato-Katz | Community-based, China (1000) | Overall Agreement / Kappa (κ) | 96.8% / κ = 0.82 (95% CI: 0.76–0.88) | Strong Agreement | [2] |
| FA280 (AI only) vs. FECT | Fresh Stool Samples (200) | Overall Agreement / Kappa (κ) | 75.5% / κ = 0.367 (95% CI: 0.248–0.486) | Fair Agreement | [3] [4] |
| FA280 (User Audit) vs. FECT | Fresh Stool Samples (200) | Overall Agreement / Kappa (κ) | 100% / κ = 1.00 (95% CI: 1.00–1.00) | Perfect Agreement | [3] [4] |
| FA280 (User Audit) vs. FECT | Preserved Stool Samples (800) | Helminth Species ID / Kappa (κ) | κ = 0.857 (95% CI: 0.82–0.894) | Strong Agreement | [3] [4] |
| FA280 vs. Normal Saline Staining | Hospital Patients, Thailand (350) | Positive Predictive Value (PPV) | 16.13% | High False-Positive Rate | [39] |
Table 2: Operational and Practical Comparison of Diagnostic Methods
| Parameter | Direct Wet Smear | FECT | FA280 with User Audit | |
|---|---|---|---|---|
| Stool Sample Weight | 0.2 g | 2 g | 0.5–1 g | [5] |
| Technique | Manual | Manual | Automatic | [5] |
| Process Simplicity | Less complicated | More complicated | Less complicated | [5] |
| Total Processing Time | ~7–12 min/sample | ~13–20 min/sample | ~5–7 min/sample | [5] |
| Parasite Observing Time | 5–10 min | 5–10 min | 3–5 min | [5] |
| Result Storage | No | No | Yes (Digital Images) | [5] |
| Cost per Test | USD 0.25 | USD 0.50 | USD 2.00 | [5] |
| Dependence on Experienced Technician | Required | Required | Required (for user audit) | [5] |
A critical factor influencing the agreement between the FA280 and the KK method is the intensity of the infection, as measured by eggs per gram (EPG). The FA280 demonstrates higher agreement with the KK method in samples with moderate to high infection intensities.
Table 3: Agreement Between FA280 and Kato-Katz Across Infection Intensity Groups
| Infection Intensity (EPG from KK) | Number of Consistently Positive Cases | Number of Positive Individuals | Agreement Rate (%) | P-value | |
|---|---|---|---|---|---|
| < 24 | 8 | 28 | 28.6 | < 0.001 | [40] |
| ≥ 24 | 76 | 88 | 86.4 | < 0.001 | [40] |
| < 48 | 32 | 54 | 59.3 | 0.003 | [40] |
| ≥ 48 | 52 | 62 | 83.9 | 0.003 | [40] |
| ≥ 192 | 16 | 16 | 100.0 | 0.018 | [40] |
Essential Research Reagent Solutions
The experiments and methodologies cited in the comparative studies rely on a set of specific reagents and materials. The following table details key research reagent solutions and their functions in the context of stool examination for parasitic infections.
Table 4: Key Research Reagents and Materials for Parasitological Diagnosis
| Reagent/Material | Function in Diagnostic Protocol | Application in Methods | |
|---|---|---|---|
| 10% Formalin Solution | Preserves parasitic structures (eggs, larvae, cysts) and fixes the stool sample to prevent microbial growth and degradation. | FECT, FC, Sample Preservation | [12] |
| Ethyl-Acetate | An organic solvent used as a fat and debris extractor. It creates a layer in the concentration tube, trapping debris and leaving concentrated parasites in the sediment. | FECT | [12] |
| Glycerol/Malachite Green (Cellophane) | Used in the KK method to clear debris on the slide, making parasite eggs more visible and transparent for microscopy. | Kato-Katz | [2] |
| Hexadecyltrimethylammonium Bromide (CTAB) | A cationic surfactant used in dissolved air flotation (DAF) and other processing protocols to modify surface charges, enhancing parasite recovery from the fecal matrix. | DAF, Advanced Processing | [29] |
| Lugol's Iodine Solution | A staining solution that enhances the contrast of protozoan cysts (e.g., nuclei and cytoplasmic inclusions) for easier identification under the microscope. | Wet Mounts, FECT, Staining | [29] |
| Merthiolate-Iodine-Formalin (MIF) | A combined fixative and staining solution used for the preservation and direct examination of stools, particularly effective for protozoa. | MIF Staining | [21] |
Discussion and Synthesis
The synthesized data indicate that the FA280 analyzer shows strong, statistically significant agreement with the Kato-Katz method for detecting Clonorchis sinensis in community-based settings (κ=0.82) [2]. Similarly, when combined with a user audit, its agreement with FECT for species identification, particularly for helminths and protozoa, can be near-perfect [3]. However, the performance is not uniform. The stand-alone AI analysis shows only fair agreement with FECT (κ=0.367), underscoring the continued importance of human expert review for optimal accuracy [3] [4]. Furthermore, the FA280's sensitivity is dependent on infection intensity, with significantly lower agreement in low-intensity infections (EPG < 24), a common challenge for many diagnostic methods [40].
The operational advantages of the FA280 are substantial. It significantly reduces processing and observation time per sample (2 minutes for processing, 3-5 minutes for observation) compared to FECT (8-10 minutes for processing, 5-10 minutes for observation) and standard wet smears [5]. This, coupled with its automated workflow and digital archiving capabilities, makes it a high-throughput tool suitable for processing large sample volumes while reducing technician fatigue and potential biohazard exposure. The primary trade-off is the higher cost per test (USD 2.00) compared to traditional methods (USD 0.25 - USD 0.50) [5].
In conclusion, the FA280 digital feces analyzer represents a significant step toward modernizing parasitological diagnosis. It serves as a robust, high-throughput screening tool that, when used with a user audit, can perform on par with established reference standards like the KK method and FECT. Its utility is greatest in settings with moderate to high infection intensities and where workflow efficiency is a priority. Researchers should be mindful of its lower sensitivity in very low-burden infections and the need for confirmatory testing in cases of ambiguity. Future developments in its AI algorithms are likely to further bridge the current performance gaps.
Conclusion
The comparative analysis between the FA280 digital feces analyzer and formalin-ethyl acetate concentration technique reveals a nuanced landscape where automation efficiency must be balanced against diagnostic sensitivity. The FA280 represents a significant advancement in parasitology diagnostics, offering substantial benefits in processing speed, technician workload reduction, and standardized reporting through its automated digital platform. However, its lower sensitivity compared to FECT, particularly in low-intensity infections and when using smaller sample volumes, necessitates careful consideration of application context. For research and drug development requiring high-throughput screening with moderate sensitivity, the FA280 offers compelling advantages. For studies demanding maximum detection sensitivity, particularly in low-prevalence settings or eradication monitoring, FECT remains essential. Future developments should focus on optimizing FA280's AI algorithms, increasing processed sample volume, and reducing operational costs. The integration of both methods in a tiered diagnostic approach may represent the most effective strategy, leveraging the FA280's efficiency for initial screening while reserving FECT for confirmation and low-intensity infection detection. This evolution in parasitic disease diagnostics will significantly impact global health initiatives, drug development pipelines, and epidemiological research methodologies.
References
- 1. High-throughput detection of parasites and ova in stool using ... [https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-023-06108-1]
- 2. Assessment of the application of the FA280—a fully ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11702166/]
- 3. High-throughput detection of parasites and ova in stool ... [https://pubmed.ncbi.nlm.nih.gov/38185634/]
- 4. High-throughput detection of parasites and ova in stool ... [https://read.qxmd.com/read/38185634/high-throughput-detection-of-parasites-and-ova-in-stool-using-the-fully-automatic-digital-feces-analyzer-orienter-model-fa280]
- 5. High-throughput detection of parasites and ova in stool using ... [https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-023-06108-1/tables/3]
- 6. CDC - DPDx - Diagnostic Procedures - Stool Specimens [https://www.cdc.gov/dpdx/diagnosticprocedures/stool/specimenproc.html]
- 7. Comparing the efficiency of four diagnostic concentration ... [https://www.sciencedirect.com/science/article/pii/S2090506817302580]
- 8. A comparative study of formalin-ethyl acetate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5991041/]
- 9. A historical review of the techniques of recovery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7269538/]
- 10. Comparison of the Quantitative Formalin Ethyl Acetate ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC1081356/]
- 11. FA-280 (AI+Feces Analyzer) [https://orbtionbiomedical.com/products/fa-280-aifeces-analyzer]
- 12. Improved Detection of Intestinal Helminth Infections with a ... [https://www.mdpi.com/2414-6366/6/2/51]
- 13. Practical Guidance for Clinical Microbiology Laboratories [https://pmc.ncbi.nlm.nih.gov/articles/PMC5740970/]
- 14. Practical guide for the diagnosis of helminth ova in stools [https://www.acm.or.kr/2702-03/]
- 15. Practical guide for the diagnosis of helminth ova ... [https://synapse.koreamed.org/articles/1516087545]
- 16. Improved Detection of Intestinal Helminth Infections with a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8167623/]
- 17. Low Sensitivity of the Formol-Ethyl Acetate Sedimentation ... [https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0000386]
- 18. Laboratory validation of the automated diagnosis of intestinal ... [https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-024-06434-y]
- 19. Evaluation of different concentration techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11922389/]
- 20. Assessment of the application of the FA280-a fully ... [https://pubmed.ncbi.nlm.nih.gov/39757228/]
- 21. Performance validation of deep-learning-based approach in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12317604/]
- 22. Comparison of CONSED™ and Formalin-Ethyl Acetate ... [https://www.alphatecsystems.com/comparison-of-consedtm-and-formalin-ethyl-acetate-methods-for-concentrating-intestinal-parasites-and-eggs/]
- 23. High-throughput detection of parasites and ova in stool ... [https://mahidol.elsevierpure.com/en/publications/high-throughput-detection-of-parasites-and-ova-in-stool-using-the]
- 24. Factors associated with detecting pathogenic intestinal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12549424/]
- 25. Advantages and limitations of diagnostic procedures for ... [https://www.cambridge.org/core/journals/journal-of-helminthology/article/advantages-and-limitations-of-diagnostic-procedures-for-zoonotic-helminth-infections-in-feces/DBA5DB59C9ADF7BD7FFDC53832D95DB3]
- 26. Performance validation of deep-learning-based approach in ... [https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-025-06878-w]
- 27. Improved Detection of Intestinal Helminth Infections with a ... [https://pubmed.ncbi.nlm.nih.gov/33921041/]
- 28. The clinical application value of the KU-F40 fully automatic ... [https://www.nature.com/articles/s41598-025-09935-7]
- 29. Laboratory validation of the automated diagnosis of intestinal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11363502/]
- 30. Ensuring Accuracy and Efficiency through AI Model Validation [https://macgence.com/blog/ensuring-accuracy-and-efficiency-through-ai-model-validation/]
- 31. FA280 Feces Analyzer - Orienter [https://liondx.com/en/fa280-feces-analyzer-orienter-2/]
- 32. High-throughput detection of parasites and ova in stool ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10771706/]
- 33. Introducing Orienter FA280: AI-Powered Feces Analyzer for ... [https://www.linkedin.com/posts/dcllabs_innovation-youronestoplaboratoryequipmentprovider-activity-7359195862465396740-WCqw]
- 34. Stool Ova and Parasite Test [https://emedicine.medscape.com/article/2117799-overview]
- 35. Detection of protozoan and helminth parasites in concentrated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12607898/]
- 36. Concentration method [https://bio-protocol.org/exchange/minidetail?id=2176385&type=30]
- 37. Evaluation of Modified Formalin-Ether Concentration ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4446584/]
- 38. Performance of formalin ethyl acetate, percoll sedimentation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8254688/]
- 39. Evaluation of the Performance of the Orienter FA280 Feces ... [https://li01.tci-thaijo.org/index.php/SRIMEDJ/article/view/265584]
- 40. Table 3 The consistency of positive results between the FA280 ... [https://idpjournal.biomedcentral.com/articles/10.1186/s40249-024-01271-8/tables/3]